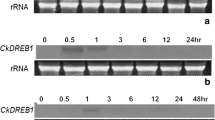
Abstract
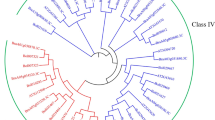
Salicylic acid is a messenger molecule in the activation of defense responses in plants. In this study, we isolated four cDNA clones representing salicylic acid-induced genes in Chinese cabbage (Brassica rapa subsp. pekinensis) by subtractive hybridization. Of the four clones, the BC5-2 clone encodes a putative glucosyltransferase protein. The BC5-3 clone is highly similar to an Arabidopsis gene encoding a putative metal-binding farnesylated protein. The BC6-1 clone is a chitinase gene with similarities to a rapeseed class IV chitinase. Class IV chitinases have deletions in the chitin-binding and catalytic domains and the BC6-1 chitinase has an additional deletion in the catalytic domain. The BCP8-1 clone is most homologous to an Arabidopsis gene that contains a tandem array of two thiJ-like sequences. These four cabbage genes were barely expressed in healthy leaves, but were strongly induced by salicylic acid and benzothiadiazole. Expression of the three genes represented by the BC5-2, BC5-3 and BCP8-1 clones were also induced by Pseudomonas syringae pv. tomato, a nonhost pathogen that elicits a hypersensitive response in Chinese cabbage. None of these four genes, however, was strongly induced by methyl jasmonate or by ethylene.





Similar content being viewed by others
Abbreviations
- BTH :
-
Benzothiadiazole
- DIG :
-
Digoxigenin
- GAPD :
-
Glyceraldehyde 3-phosphate dehydrogenase
- HMP :
-
Hydroxymethylpyrimidine
- HR :
-
Hypersensitive response
- Me-JA :
-
Methyl jasmonate
- Pst :
-
Pseudomonas syringae pv. tomato
- SAR :
-
Systemic acquired resistance
References
Bak S, Nielsen HL, Halkier BA (1998) The presence of CYP79 homologues in glucosinolate-producing plants shows evolutionary conservation of the enzymes in the conversion of amino acid to aldoxime in the biosynthesis of cyanogenic glucosides and glucosinolates. Plant Mol Biol 38:725–734
Bent AF (1996) Plant disease resistance genes: function meets structure. Plant Cell 8:1757–1771
Chong J, Baltz R, Schmitt C, Beffa R, Fritig B, Saindrenan P (2002) Downregulation of a pathogen-responsive tobacco UDP-Glc:phenylpropanoid glucosyltransferase reduces scopoletin glucoside accumulation, enhances oxidative stress, and weakens virus resistance. Plant Cell 14:1093–1107
Dangl JL, Jones JDG (2001) Plant pathogens and integrated defence responses to infection. Nature 411:826–833
De Vries S, Hoge H, Bisseling T (1988) Isolation of total and polysomal RNA from plant tissues. In: Gelvin SB, Schilperoot RA (eds) Plant molecular biology. Kluwer, Dordrecht, pp B6/1–5
Dykema PE, Sipes PR, Marie A, Biermann BJ, Crowell DN, Randall SK (1999) A new class of proteins capable of binding transition metals. Plant Mol Biol 41:139–150
Goerlach J, Volrath S, Knauf-Beiter G, Hengy G, Beckhove U, Kogel K-H, Oostendorp M, Staub T, Ward E, Kessmann H, Ryals J (1996) Benzothiadiazole, a novel class of inducers of systemic acquired resistance, activates gene expression and disease resistance in wheat. Plant Cell 8:629–643
Halio SB, Blumenthals II, Short SA, Merrill BM, Kelly RM (1996) Sequence, expression in Escheria coli, and analysis of the gene encoding a novel intracellular protease (PfpI) from the hyperthermophilic archaeon Pyrococcus furiosus. J Bacteriol 178:2605–2612
Hammond-Kosak KE, Jones JDG (1996) Resistance gene-dependent plant defense responses. Plant Cell 8:1773–1791
Himelblau E, Mira H, Lin SJ, Culotta VC, Penarrubia L, Amasino RM (1998) Identification of a functional homolog of the yeast copper homeostasis gene ATX1 from Arabidopsis. Plant Physiol 117:1227–1234
Jones PR, Moeller BL, Hoej PB (1999) The UDP-glucose:hydroxymandelonitrile-O-glucosyltransferase that catalyzes the last step in synthesis of the cyanogenic glucoside dhurrin in Sorghum bicolor. J Biol Chem 274:35483–35491
Lam E, Kato N, Lawton M (2001) Programmed cell death, mitochondria and the plant hypersensitive response. Nature 411:848–853
Lamb C, Dixon RA (1997) The oxidative burst in plant disease resistance. Annu Rev Plant Physiol Plant Mol Biol 48:251–275
Lange J, Mohr U, Wiemken A, Boller T, Vogeli-Lange R (1996) Proteolytic processing of class IV chitinase in the compatible interaction of bean roots with Fusarium solani. Plant Physiol 111:1135–1144
Li Y, Baldauf S, Lim E-K, Bowles DJ (2001) Phylogenetic analysis of the UDP-glycosyltransferase multigene family of Arabidopsis thaliana. J Biol Chem 276:4338–4343
Lin S-J, Culotta VC (1995) The ATX1 gene of Saccharomyces cerevisiae encodes a small metal homeostasis factor that protects cells against reactive oxygen toxicity. Proc Natl Acad Sci USA 92:3784–3788
Martin RC, Mok MC, Mok DWS (1999) A gene encoding the cytokinin enzyme zeatin O-xylosyltransferase of Phaseolus vulgaris. Plant Physiol 120:553–557
McConn M, Creelman RA, Bell E, Mullet JE, Browse J (1997) Jasmonate is essential for insect defense in Arabidopsis. Proc Natl Acad Sci USA 94:5473–5477
McDowell JM, Dangl JL (2000) Signal transduction in the plant immune response. Trends Biochem Sci 25:79–82
Mizote T, Tsuda M, Smith DDS, Nakayama H, Nakazawa T (1999) Cloning and characterization of the thiD/J gene of Escherichia coli encoding a thiamin-synthesizing bifunctional enzyme, hydroxymethylpyrimidine kinase/phosphomethylpyrimidine kinase. Microbiology 145:495–501
Nagakubo D, Taira T, Kitaura H, Ikeda M, Tamai K, Iguchi-Ariga SMM, Ariga H (1997) DJ-1, a novel oncogene which transforms mouse NIH3T3 cells in cooperation with ras. Biochem Biophys Res Commun 231:509–513
Nambara E, McCourt P (2000) Protein farnesylation in plants: a greasy tale. Curr Opin Plant Biol 2:388–392
Neuhaus J-M (1999) Plant chitinase (PR-3, PR-4, PR-8, PR-11). In: Datta SK, Muthukrishnan S (eds) Pathogenesis-related proteins in plants, CRC Press, Boca Raton, pp 77–105
Nuernberger T, Scheel D (2001) Signal transmission in the plant immune response. Trends Plant Sci 6:372–379
O'Donnell PJ, Calvert C, Atzorn R, Wastemack C, Leyser HMO, Bowles DJ (1996) Ethylene as a signal mediating the wound response of tomato plants. Science 274:1914–1917
Pieterse CMJ, van Loon LC (1999) Salicylic acid-independent plant defence pathways. Trends Plant Sci 4:52–58
Rad U von, Huttl R, Lottspeich F, Gierl A, Frey M (2001) Two glucosyltransferases are involved in detoxification of benzoxazinoids in maize. Plant J 28:633–642
Rasmussen U, Bojsen K, Collinge DB (1992) Cloning and characterization of a pathogen-induced chitinase in Brassica napus. Plant Mol Biol 20:277–287
Ryals JA, Neuenschwander UH, Willits MG, Molina A, Steiner HY, Hunt MD (1996) Systemic acquired resistance. Plant Cell 8:1809–1819
Ryang S-H, Chung S-Y, Lee S-H, Cha J-S, Kim HY, Cho T-J (2002) Isolation of pathogen-induced Chinese cabbage genes by subtractive hybridization employing selective adaptor ligation. Biochem Biophys Res Commun 299:352–359
Szerszen JB, Szczyglowski K, Bandurski RS (1994) iaglu, a gene from Zea mays involved in conjugation of growth hormone indole-3-acetic acid. Science 265:1699–1701
Takasaki T, Hatakeyama K, Suzuki G, Watanabe M, Isogai A, Hinata K (2000) The S receptor kinase determines self-incompatibility in Brassica stigma. Nature 403:913–916
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res 22:4673–4680
Vogt T, Jones P (2000) Glycosyltransferase in plant natural product synthesis: characterization of a supergene family. Trends Plant Sci 5:380–386
Wagenfeld A, Gromoll J, Cooper TG (1998) Molecular cloning of rat contraception associated protein 1 (CAP1), a protein putatively involved in fertilization. Biochem Biophys Res Commun 251:545–549
Zhang FL, Casey PJ (1996) Protein prenylation: molecular mechanisms and functional consequences. Annu Rev Biochem 65:241–269
Acknowledgements
This work was supported by a grant from the Technology Development Program of the Ministry of Agriculture and Forestry, Republic of Korea (grant no. 299045-3).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by I.S. Chung
Rights and permissions
About this article
Cite this article
Park, YS., Min, HJ., Ryang, SH. et al. Characterization of salicylic acid-induced genes in Chinese cabbage. Plant Cell Rep 21, 1027–1034 (2003). https://doi.org/10.1007/s00299-003-0606-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-003-0606-9