Abstract
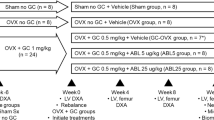
The objective of this study is to evaluate the efficacy of ibandronate (IBN) in prevention and treatment of glucocorticoid-induced osteoporosis in rabbits. A total of 60 New Zealand white rabbits were randomly allocated into six groups. Twenty rabbits in the control group were injected with saline. Forty rabbits received dexamethasone (DX) treatment at a dose of 3 mg/kg twice weekly during the first 6 weeks, while 10 of these rabbits (group IBN&DX) were injected additionally with IBN at a dose of 2 mg/kg before DX treatment. At week 6, the rabbits from IBN&DX group, 10 rabbits from control group (group CNTR-1) and 10 rabbits treated with DX (group DX6) were killed. Half (10) of the remaining rabbits in DX group were continued for DX treatment at a dose of 3 mg/kg once weekly (group DX12), while the other half (10) rabbits (group DX&IBN) additionally received IBN injection (2 mg/kg) once before continuing DX treatment. The remaining rabbits (10) in an additional of control group (group CNTR-2) continuously received saline. At week 12, all rabbits were killed for bone biomechanical analysis and histological examination. At week 6, the analysis of bone biomechanical and histological results of group CNTR-1 and DX6 showed that GIOP rabbit models were successfully established. Compared with group DX6, bone volume/tissue volume (BV/TV), trabecular number (Tb.N) and trabecular thickness (Tb.Th) of lumbar spine in group IBN&DX were increased by 100, 45.74 and 40.55%, respectively (P < 0.01). Meanwhile, BV/TV and Tb.N of femoral neck were increased by 30.29 and 16.86%, respectively (P < 0.01). The maximum compressive load, the maximum bending stress and the maximum torque were increased by 24.19, 29.91 and 37.24%, respectively (P < 0.01). At week 12, in comparison of the results between group DX12 and group DX6, the histomorphometric and mechanical analysis demonstrated that prolonged DX treatment could lead to further loss of bone mass and strength. Compared with group DX12, BV/TV, Tb.N and Tb.Th of lumbar spine in group DX&IBN were increased by 73.34, 39.02 and 23.87%, respectively (P < 0.05), the parameters of femoral neck were increased by 88.75, 31.29 and 42.01%, respectively (P < 0.01), and the biomechanical parameters were increased by 54.36, 21.38 and 105.75%, respectively (P < 0.05). IBN could effectively prevent and treat high-dosing glucocorticoid-induced loss of bone mass and strength in rabbits.

Similar content being viewed by others
References
Baxter JD, Rousseau GG (1979) Glucocorticoid hormone action: an overview. Monogr Endocrinol 12:1–24
Van Staa TP, Leufkens HG, Abenhaim L, Zhang B, Cooper C (2000) Use of oral corticosteroids and risk of fractures. J Bone Miner Res 15:993–1000
Steinbuch M, Youket TE, Cohen S (2004) Oral glucocorticoid use is associated with an increased risk of fracture. Osteoporos Int 15:323–328
Mazziotti G, Angeli A, Bilezikian JP, Canalisd E, Giustinaa A (2006) Glucocorticoid-induced osteoporosis: an update. Trends Endocrinol Metab 7:144–149
Van Staa TP, Laan RF, Barton IP, Cohen S, Reid DM, Cooper C (2003) Bone density threshold and other predictors of vertebral fracture in patients receiving oral glucocorticoid therapy. Arthritis Rheum 48:3224–3229
Van Staa TP, Leufkens HG, Cooper C (2002) The epidemiology of corticosteroid-induced osteoporosis. Osteoporos Int 13:777–787
Weinstein RS, Jilka RL, Parfitt AM, Manolagas SC (1998) Inhibition of osteoblastogenesis and promotion of apoptosis of osteoblasts and osteocytes by glucocorticoids potential mechanisms of their deleterious effects on bone. J Clin Invest 102:274–282
Canalis E (2005) Mechanisms of glucocorticoid action in bone. Curr Osteoporos 3:98–102
Compston J (2010) Management of glucocorticoid-induced osteoporosis. Nat Rev Rheumatol 6:82–88
Amin S, Lavalley MP, Simms RW, Felson DT (2002) The comparative efficacy of drug therapies used for the management of corticosteroid-induced osteoporosis: a meta-regression. J Bone Miner Res 17:1512–1526
Cramer JA, Lynch NO, Gaudin AF, Walker M, Cowell W (2006) The effect of dosing frequency on compliance and persistence with bisphosphonate therapy in postmenopausal women: a comparison of studies in the United States, the United Kingdom and France. Clin Ther 28:1686–1694
Cramer JA, Gold DT, Silverman SL, Lewiecki EM (2007) A systematic review of persistence and compliance with bisphosphonates for osteoporosis. Osteoporos Int 18:1023–1031
Miller PD, McClung MR, Macovei L, Stakkestad JA, Luckey M, Bonvoisin B, Reginster JY, Recker RR, Hughes C, Lewiecki EM, Felsenberg D, Delmas PD, Kendler DL, Bolognese MA, Mairon N, Cooper C (2005) Monthly oral ibandronate therapy in postmenopausal osteoporosis: 1-year results from the MOBILE study. J Bone Miner Res 20:1315–1322
Stakkestad JA, Benevolenskaya LI, Stepan JJ, Skag A, Nordby A, Oefjord E, Burdeska A, Jonkanski I, Mahoney P (2003) Intravenous ibandronate injections given every three months: a new treatment option to prevent bone loss in postmenopausal women. Ann Rheum Dis 62:969–975
Adami S, Felsenberg D, Christiansen C, Robinson J, Lorenc RS, Mahoney P, Coutant K, Schimmer RC, Delmas PD (2004) Efficacy and safety of ibandronate given by intravenous injection once every 3 months. Bone 34:881–889
Eisman JA, Civitelli R, Adami S, Czerwinski E, Recknor C, Prince R, Reginster JY, Zaidi M, Felsenberg D, Hughes C, Mairon N, Masanauskaite D, Reid DM, Delmas PD, Recker RR (2008) Efficacy and tolerability of intravenous ibandronate injections in postmenopausal osteoporosis: 2-year results from the DIVA study. J Rheumatol 35:488–497
Turner CH, Burr DB (1993) Basic biomechanical measurements of bone: a tutorial. Bone 14:595–608
Turner AS (2001) Animal models of osteoporosis–necessity and limitations. Eur Cell Mater 1:66–81
Castañeda S, Calvo E, Largo R, González-González R, de la Piedra C, Díaz-Curiel M, Herrero-Beaumont G (2008) Characterization of a new experimental model of osteoporosis in rabbits. J Bone Miner Metab 26:53–59
Wen Q, Ma L, Chen YP, Yang L, Luo W, Wang XN (2008) A rabbit model of hormone-induced early avascular necrosis of the femoral head. Biomed Environ Sci 21:398–403
Takahashi M, Saha PK, Wehrli FW (2006) Skeletal effects of short-term exposure to dexamethasone and response to risedronate treatment studied in vivo in rabbits by magnetic resonance micro-imaging and spectroscopy. J Bone Miner Metab 24:467–475
Pecherstorfer M, Steinhauer EU, Rizzoli R, Wetterwald M, Bergström B (2003) Efficacy and safety of ibandronate in the treatment of hypercalcemia of malignancy: a randomized multicentric comparison to pamidronate. Support Care Cancer 1:539–547
Heras P, Kritikos K, Hatzopoulos A, Georgopoulou AP (2009) Efficacy of ibandronate for the treatment of skeletal events in patients with metastatic breast cancer. Eur J Cancer Care 18:653–656
Delmas PD, Adami S, Strugala C, Stakkestad JA, Reginster JY, Felsenberg D, Christiansen C, Civitelli R, Drezner MK, Recker RR, Bolognese M, Hughes C, Masanauskaite D, Ward P, Sambrook P, Reid DM (2006) Intravenous ibandronate injections in postmenopausal women with osteoporosis: one-year results from the dosing intravenous administration study. Arthritis Rheum 54:1838–1846
Glüer CC, Scholz-Ahrens KE, Helfenstein A, Delling G, Timm W, Açil Y, Barkmann R, Hassenpflug J, Stampa B, Bauss F, Schrezenmeir J (2007) Ibandronate treatment reverses glucocorticoid-induced loss of bone mineral density and strength in mini pigs. Bone 40:645–655
Ringe JD, Dorst A, Faber H, Ibach K, Sorenson F (2003) Intermittent intravenous ibandronate injections reduce vertebral fracture risk in corticosteroid-induced osteoporosis: results from a long-term comparative study. Osteoporos Int 14:801–807
Guidelines Working Group for the Bone and Tooth Society, National Osteoporosis Society and Royal College of Physicians (2002) Glucocorticoid-induced osteoporosis: guidelines for prevention and treatment. Royal College of Physicians, London
Kikuchi Y, Imakiire T, Yamada M, Saigusa T, Hyodo T, Kushiyama T, Higashi K, Hyodo N, Yamamoto K, Suzuki S, Miura S (2007) Effect of risedronate on high-dose corticosteroid-induced bone loss in patients with glomerular disease. Nephrol Dial Transplant 22:1593–1600
Nakayamada S, Okada Y, Saito K, Tanaka Y (2004) Etidronate prevents high dose glucocorticoid induced bone loss in premenopausal individuals with systemic autoimmune diseases. J Rheumatol 31:163–166
Meikle AW, Tyler FH (1977) Potency and duration of action of glucocorticoids Effects of hydrocortisone, prednisone and dexamethasone on human pituitary-adrenal function. Am J Med 63:200–207
Webb R, Singer M (2005) Oxford handbook of critical care. Oxford University Press, New York
Monier-Faugere MC, Geng Z, Paschalis EP, Qi Q, Arnala I, Bauss F, Boskey AL, Malluche HH (1999) Intermittent and continuous administration of the bisphosphonate ibandronate in ovariohysterectomized beagle dogs: effects on bone morphometry and mineral properties. J Bone Miner Res 14:1768–1778
Bauss F, Wagner M, Hothorn LH (2002) Total administered dose of ibandronate determines its effects on bone mass and architecture in ovariectomized aged rats. J Rheumatol 29:990–998
Müller R, Hannan M, Smith SY, Bauss F (2004) Intermittent ibandronate preserves bone quality and bone strength in the lumbar spine after 16 months of treatment in the ovariectomized cynomolgus monkey. J Bone Miner Res 19:1787–1796
Li M, Xing XP, Zhang ZL, Liu JL, Zhang ZL, Liu DG, Xia WB, Meng XW (2010) Infusion of ibandronate once every 3 months effectively decreases bone resorption markers and increases bone mineral density in Chinese postmenopausal osteoporotic women: a 1-year study. J Bone Miner Metab 28:299–305
Bergner R, Henrich D, Hoffmann M, Schmidt-Gayk H, Lenz T, Upperkamp M (2008) Treatment of reduced bone density with ibandronate in dialysis patients. J Nephrol 21:510–516
Acknowledgments
This work was supported by Key Issues of Medical Sciences Research of Hebei Provincial Health Bureau (No. 08114).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Kj., Zhang, J., Kang, Zk. et al. Ibandronate for prevention and treatment of glucocorticoid-induced osteoporosis in rabbits. Rheumatol Int 32, 3405–3411 (2012). https://doi.org/10.1007/s00296-011-2074-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-011-2074-9