Abstract
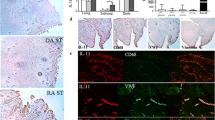
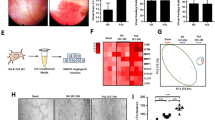
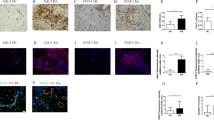
Angiogenesis in synovia is a characteristic of RA patients. We examined whether IL-6 or TNF-α induce tubule formation in a co-culture system of fibroblast-like synovial cells from RA patients (RA-FLS) and human umbilical vein endothelial cells (HUVEC). The effects of IL-6 and TNF-α on the expression of angiogenic factors in RA-FLS and HUVEC, and the proliferation of HUVEC were also studied. IL-6 + sIL-6R induced tubule formation, whereas IL-6 alone did not. IL-6/sIL-6R-induced tubule formation was completely suppressed by the addition of either anti-IL-6R or anti-VEGF antibody. TNF-α did not induce tubule formation. On the contrary, it decreased CD31-positive area compared with the control. IL-6 + sIL-6R augmented VEGF production in RA-FLS, whereas IL-6 alone did not. Anti-IL-6R antibody suppressed IL-6/sIL-6R-induced VEGF production, but not spontaneous VEGF production. In contrast, TNF-α did not induce VEGF production from RA-FLS and HUVEC. IL-6 + sIL-6R stimulation of RA-FLS strongly induced mRNA expression of VEGF, but not of other angiogenic factors, such as EGF, bFGF, TGF-β, IL-1, TNF-α and IL-8. Neither IL-6 nor IL-6/sIL-6R promoted HUVEC proliferation, whereas TNF-α significantly inhibited VEGF-induced HUVEC proliferation. In conclusion, IL-6/sIL-6R complex showed angiogenic activity via the production of VEGF from RA-FLS, but TNF-α was anti-angiogenic in our experimental system.




Similar content being viewed by others
References
Koch AE (2000) The role of angiogenesis in rheumatoid arthritis: recent developments. Ann Rheum Dis 59(suppl 1):65–71. doi:10.1136/ard.59.suppl_1.i65
Bainbridge J, Sivakumar B, Paleolog E (2006) Angiogenesis as a therapeutic target in arthritis: lessons from oncology. Curr Pharm Des 12:2631–2644. doi:10.2174/138161206777698747
Maruotti N, Cantatore FP, Crivellato E, Vacca A, Ribatti D (2006) Angiogenesis in rheumatoid arthritis. Histol Histopathol 21:557–566
Fava RA, Olsen NJ, Spencer-Green G, Yeo KT, Yeo TK, Berse B et al (1994) Vascular permeability factor/endothelial growth factor (VPF/VEGF): accumulation and expression in human synovial fluids and rheumatoid synovial tissue. J Exp Med 180:341–346. doi:10.1084/jem.180.1.341
Harada M, Mitsuyama K, Yoshida H, Sakisaka S, Taniguchi E, Kawaguchi T et al (1998) Vascular endothelial growth factor in patients with rheumatoid arthritis. Scand J Rheumatol 27:377–380. doi:10.1080/03009749850154429
Sone H, Sakauchi M, Takahashi A, Suzuki H, Inoue N, Iida K et al (2001) Elevated levels of vascular endothelial growth factor in the sera of patients with rheumatoid arthritis correlation with disease activity. Life Sci 69:1861–1869. doi:10.1016/S0024-3205(01)01264-4
Lu J, Kasama T, Kobayashi K, Yoda Y, Shiozawa F, Hanyuda M et al (2000) Vascular endothelial growth factor expression and regulation of murine collagen-induced arthritis. J Immunol 164:5922–5927
Sone H, Kawakami Y, Sakauchi M, Nakamura Y, Takahashi A, Shimano H et al (2001) Neutralization of vascular endothelial growth factor prevents collagen-induced arthritis and ameliorates established disease in mice. Biochem Biophys Res Commun 281:562–568. doi:10.1006/bbrc.2001.4395
Taylor PC, Williams RO, Maini RN (2000) Anti-TNF alpha therapy in rheumatoid arthritis—current and future directions. Curr Dir Autoimmun 2:83–102. doi:10.1159/000060499
Smolen JS, Beaulieu A, Rubbert-Roth A, Ramos-Remus C, Rovensky J, Alecock E et al (2008) Effect of interleukin-6 receptor inhibition with tocilizumab in patients with rheumatoid arthritis (OPTION study): a double-blind, placebo-controlled, randomised trial. Lancet 371:987–997. doi:10.1016/S0140-6736(08)60453-5
Paleolog EM, Young S, Stark AC, McCloskey RV, Feldmann M, Maini RN (1998) Modulation of angiogenic vascular endothelial growth factor by tumor necrosis factor alpha and interleukin-1 in rheumatoid arthritis. Arthritis Rheum 41:1258–1265. doi:10.1002/1529-0131(199807)41:7<1258::AID-ART17>3.0.CO;2-1
Nakahara H, Song J, Sugimoto M, Hagihara K, Kishimoto T, Yoshizaki K et al (2003) Anti-interleukin-6 receptor antibody therapy reduces vascular endothelial growth factor production in rheumatoid arthritis. Arthritis Rheum 48:1521–1529. doi:10.1002/art.11143
Saito A, Sugawara A, Uruno A, Kudo M, Kagechika H, Sato Y et al (2007) All-trans retinoic acid induces in vitro angiogenesis via retinoic acid receptor: possible involvement of paracrine effects of endogenous vascular endothelial growth factor signaling. Endocrinology 148:1412–1423. doi:10.1210/en.2006-0900
Chen Y, Wei T, Yan L, Lawrence F, Qian HR, Burkholder TP, et al. (2008) Developing and applying a gene functional association network for anti-angiogenic kinase inhibitor activity assessment in an angiogenesis co-culture model. BMC Genomics 9:264. doi:10.1186/1471-2164-9-264
Mihara M, Moriya Y, Kishimoto T, Ohsugi Y (1995) Interleukin-6 (IL-6) induces the proliferation of synovial fibroblastic cells in the presence of soluble IL-6 receptor. Br J Rheumatol 34:321–325. doi:10.1093/rheumatology/34.4.321
Mihara M, Moriya Y, Ohsugi Y (1996) IL-6-soluble IL-6 receptor complex inhibits the proliferation of dermal fibroblasts. Int J Immunopharmacol 18:89–94. doi:10.1016/0192-0561(95)00106-9
Kanzaki Y, Onoue F, Ishikawa F, Ide T (2002) Telomerase rescues the expression levels of keratinocyte growth factor and insulin-like growth factor-II in senescent human fibroblasts. Exp Cell Res 297:321–329. doi:10.1006/excr.2002.5607
Celeste AJ, Iannazzi JA, Taylor RC, Hewick RM, Rosen V, Wang EA et al (1990) Identification of transforming growth factor beta family members present in bone-inductive protein purified from bovine bone. Proc Natl Acad Sci USA 87:9843–9847. doi:10.1073/pnas.87.24.9843
Lagoo-Deenadayalan S, Laggo AS, Barber WH, Hardy KJ (1993) A standardized approach to PCR-based semiquantitation of multiple cytokine gene transcripts from small cell samples. Lymphokine Cytokine Res 12:59–67
Kameyama Y, Yamashita K, Kobayashi K, Hosokawa M, Chiba K (2005) Functional characterization of SLCO1B1 (OATP-C) variants, SLCO1B1*5, SLCO1B1*15 and SLCO1B1*15 + C1007G, by using transient expression systems of HeLa and HEK293 cells. Pharmacogenet Genomics 15:513–522. doi:10.1097/01.fpc.0000170913.73780.5f
Ko Y, Totzke G, Gouni-Berthold I, Sachinidis A, Vetter H (1999) Cytokine-inducible growth factor gene expression in human umbilical endothelial cells. Mol Cell Probes 13:203–211. doi:10.1006/mcpr.1999.0236
Charles P, Elliott MJ, Davis D, Potter A, Kalden JR, Antoni C et al (1999) Regulation of cytokines, cytokine inhibitors, and acute-phase proteins following anti-TNF-alpha therapy in rheumatoid arthritis. J Immunol 163:1521–1528
Patterson C, Perrella MA, Endege WO, Yoshizumi M, Lee ME, Haber E (1996) Downregulation of vascular endothelial growth factor receptors by tumor necrosis factor-alpha in cultured human vascular endothelial cells. J Clin Invest 98:490–496. doi:10.1172/JCI118816
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hashizume, M., Hayakawa, N., Suzuki, M. et al. IL-6/sIL-6R trans-signalling, but not TNF-α induced angiogenesis in a HUVEC and synovial cell co-culture system. Rheumatol Int 29, 1449–1454 (2009). https://doi.org/10.1007/s00296-009-0885-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-009-0885-8