Abstract
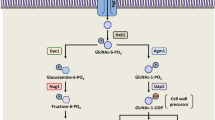
Candida albicans is an important opportunistic fungal pathogen of immunocompromised individuals. The ability to switch between yeast, pseudohyphal, and hyphal growth forms (polymorphism) is one of the most investigated virulence attributes of C. albicans. The usual method for inducing hypha formation in the lab is by diluting cells from a saturated culture into fresh medium at 37 °C. The molecular mechanism at action under these conditions has been previously investigated. C. albicans can also form hyphae in growing cells without dilution. The ability of C. albicans to form hyphae in different cell states facilitates the fungus to adapt varied host environments during infection. A recent study by Su et al. uncovered the molecular mechanism for how C. albicans develops hyphae under the condition without inoculation. N-Acetylglucosamine (GlcNAc) stimulates filamentation in log phase cells through transcriptional down-regulation of NRG1, the major repressor of hyphal development. Instead of cAMP–PKA pathway, GlcNAc sensor Ngs1 is responsible for this process. Ngs1 binds to GlcNAc to activate its N-acetyltransferase activity, leading to the induction of BRG1 expression. The increased level of BRG1 could repress NRG1 transcripts, resulting in hyphal growth. Hyphal development in log phase cells induced by serum or neutral pH also requires activation of BRG1 to down-regulate NRG1 transcription. Therefore, hyphal induction under the condition without inoculation is trigged by Brg1-mediated removal of Nrg1 inhibition. This review describes our current understanding of the molecular mechanism underlying hyphal development, the best studied virulence factor in C. albicans. These will expand the number of potential drug targets with novel modes of action for anti-virulence therapeutics.

Similar content being viewed by others
References
Braun BR, Kadosh D, Johnson AD (2001) NRG1, a repressor of filamentous growth in C. albicans, is down-regulated during filament induction. EMBO J 20:4753–4761. https://doi.org/10.1093/emboj/20.17.4753
Calderone RA, Fonzi WA (2001) Virulence factors of Candida albicans. Trends Microbiol 9:327–335
Dalle F, Wachtler B, L’Ollivier C, Holland G, Bannert N, Wilson D, Labruere C, Bonnin A, Hube B (2010) Cellular interactions of Candida albicans with human oral epithelial cells and enterocytes. Cell Microbiol 12:248–271. https://doi.org/10.1111/j.1462-5822.2009.01394.x
Davis-Hanna A, Piispanen AE, Stateva LI, Hogan DA (2008) Farnesol and dodecanol effects on the Candida albicans Ras1-cAMP signalling pathway and the regulation of morphogenesis. Mol Microbiol 67:47–62. https://doi.org/10.1111/j.1365-2958.2007.06013.x
Du H, Huang G (2016) Environmental pH adaption and morphological transitions in Candida albicans. Curr Genet 62:283–286. https://doi.org/10.1007/s00294-015-0540-8
Du H, Guan G, Li X, Gulati M, Tao L, Cao C, Johnson AD, Nobile CJ, Huang G (2015) N-Acetylglucosamine-induced cell death in Candida albicans and its implications for adaptive mechanisms of nutrient sensing in yeasts. MBio 6:e01376-01315. https://doi.org/10.1128/mBio.01376-15
Homann OR, Dea J, Noble SM, Johnson AD (2009) A phenotypic profile of the Candida albicans regulatory network. PLoS Genet 5:e1000783. https://doi.org/10.1371/journal.pgen.1000783
Hornby JM, Jensen EC, Lisec AD, Tasto JJ, Jahnke B, Shoemaker R, Dussault P, Nickerson KW (2001) Quorum sensing in the dimorphic fungus Candida albicans is mediated by farnesol. Appl Environ Microbiol 67:2982–2992. https://doi.org/10.1128/AEM.67.7.2982-2992.2001
Huang G, Yi S, Sahni N, Daniels KJ, Srikantha T, Soll DR (2010) N-Acetylglucosamine induces white to opaque switching, a mating prerequisite in Candida albicans. PLoS Pathog 6:e1000806. https://doi.org/10.1371/journal.ppat.1000806
Kadosh D, Johnson AD (2005) Induction of the Candida albicans filamentous growth program by relief of transcriptional repression: a genome-wide analysis. Mol Biol Cell 16:2903–2912. https://doi.org/10.1091/mbc.E05-01-0073
Kirkpatrick CH (1994) Chronic mucocutaneous candidiasis. J Am Acad Dermatol 31:S14-17
Klein BS, Tebbets B (2007) Dimorphism and virulence in fungi. Curr Opin Microbiol 10:314–319. https://doi.org/10.1016/j.mib.2007.04.002
Klengel T, Liang WJ, Chaloupka J, Ruoff C, Schroppel K, Naglik JR, Eckert SE, Mogensen EG, Haynes K, Tuite MF, Levin LR, Buck J, Muhlschlegel FA (2005) Fungal adenylyl cyclase integrates CO2 sensing with cAMP signaling and virulence. Curr Biol 15:2021–2026. https://doi.org/10.1016/j.cub.2005.10.040
Konopka JB (2012) N-Acetylglucosamine (GlcNAc) functions in cell signaling. Scientifica (Cairo) 2012:489208
Lorenz MC, Bender JA, Fink GR (2004) Transcriptional response of Candida albicans upon internalization by macrophages. Eukaryot Cell 3:1076–1087. https://doi.org/10.1128/EC.3.5.1076-1087.2004
Lu Y, Su C, Wang A, Liu H (2011) Hyphal development in Candida albicans requires two temporally linked changes in promoter chromatin for initiation and maintenance. PLoS Biol 9:e1001105. https://doi.org/10.1371/journal.pbio.1001105
Lu Y, Su C, Solis NV, Filler SG, Liu H (2013) Synergistic regulation of hyphal elongation by hypoxia, CO(2), and nutrient conditions controls the virulence of Candida albicans. Cell Host Microbe 14:499–509. https://doi.org/10.1016/j.chom.2013.10.008
Lu Y, Su C, Unoje O, Liu H (2014) Quorum sensing controls hyphal initiation in Candida albicans through Ubr1-mediated protein degradation. Proc Natl Acad Sci USA 111:1975–1980. https://doi.org/10.1073/pnas.1318690111
Moyes DL, Wilson D, Richardson JP, Mogavero S, Tang SX, Wernecke J, Hofs S, Gratacap RL, Robbins J, Runglall M, Murciano C, Blagojevic M, Thavaraj S, Forster TM, Hebecker B, Kasper L, Vizcay G, Iancu SI, Kichik N, Hader A, Kurzai O, Luo T, Kruger T, Kniemeyer O, Cota E, Bader O, Wheeler RT, Gutsmann T, Hube B, Naglik JR (2016) Candidalysin is a fungal peptide toxin critical for mucosal infection. Nature 532:64–68. https://doi.org/10.1038/nature17625
Murad AM, Leng P, Straffon M, Wishart J, Macaskill S, MacCallum D, Schnell N, Talibi D, Marechal D, Tekaia F, d’Enfert C, Gaillardin C, Odds FC, Brown AJ (2001) NRG1 represses yeast-hypha morphogenesis and hypha-specific gene expression in Candida albicans. EMBO J 20:4742–4752. https://doi.org/10.1093/emboj/20.17.4742
Naseem S, Gunasekera A, Araya E, Konopka JB (2011) N-acetylglucosamine (GlcNAc) induction of hyphal morphogenesis and transcriptional responses in Candida albicans are not dependent on its metabolism. J Biol Chem 286:28671–28680. https://doi.org/10.1074/jbc.M111.249854
Odds FC (1987) Candida infections: an overview. Crit Rev Microbiol 15:1–5. https://doi.org/10.3109/10408418709104444
Park YN, Morschhauser J (2005) Tetracycline-inducible gene expression and gene deletion in Candida albicans. Eukaryot Cell 4:1328–1342. https://doi.org/10.1128/EC.4.8.1328-1342.2005
Parrino SM, Si H, Naseem S, Groudan K, Gardin J, Konopka JB (2017) cAMP-independent signal pathways stimulate hyphal morphogenesis in Candida albicans. Mol Microbiol 103:764–779. https://doi.org/10.1111/mmi.13588
Romani L, Bistoni F, Puccetti P (2003) Adaptation of Candida albicans to the host environment: the role of morphogenesis in virulence and survival in mammalian hosts. Curr Opin Microbiol 6:338–343
Saville SP, Lazzell AL, Monteagudo C, Lopez-Ribot JL (2003) Engineered control of cell morphology in vivo reveals distinct roles for yeast and filamentous forms of Candida albicans during infection. Eukaryot Cell 2:1053–1060
Saville SP, Lazzell AL, Bryant AP, Fretzen A, Monreal A, Solberg EO, Monteagudo C, Lopez-Ribot JL, Milne GT (2006) Inhibition of filamentation can be used to treat disseminated candidiasis. Antimicrob Agents Chemother 50:3312–3316. https://doi.org/10.1128/AAC.00628-06
Schaller M, Korting HC, Schafer W, Bastert J, Chen W, Hube B (1999) Secreted aspartic proteinase (Sap) activity contributes to tissue damage in a model of human oral candidosis. Mol Microbiol 34:169–180
Schmid J, Magee PT, Holland BR, Zhang N, Cannon RD, Magee BB (2016) Last hope for the doomed? Thoughts on the importance of a parasexual cycle for the yeast Candida albicans. Curr Genet 62:81–85. https://doi.org/10.1007/s00294-015-0516-8
Shapiro RS, Cowen L (2010) Coupling temperature sensing and development: Hsp90 regulates morphogenetic signalling in Candida albicans. Virulence 1:45–48. https://doi.org/10.4161/viru.1.1.10320
Shapiro RS, Uppuluri P, Zaas AK, Collins C, Senn H, Perfect JR, Heitman J, Cowen LE (2009) Hsp90 orchestrates temperature-dependent Candida albicans morphogenesis via Ras1-PKA signaling. Curr Biol 19:621–629. https://doi.org/10.1016/j.cub.2009.03.017
Staab JF, Bradway SD, Fidel PL, Sundstrom P (1999) Adhesive and mammalian transglutaminase substrate properties of Candida albicans Hwp1. Science 283:1535–1538
Su C, Lu Y, Liu H (2016) N-Acetylglucosamine sensing by a GCN5-related N-acetyltransferase induces transcription via chromatin histone acetylation in fungi. Nat Commun 7:12916. https://doi.org/10.1038/ncomms12916
Su C, Yu J, Sun Q, Liu Q, Lu Y (2018) Hyphal induction under the condition without inoculation in Candida albicans is triggered by Brg1-mediated removal of NRG1 inhibition. Mol Microbiol. https://doi.org/10.1111/mmi.13944
Sullivan PA, Shepherd MG (1982) Gratuitous induction by N-acetylmannosamine of germ tube formation and enzymes for N-acetylglucosamine utilization in Candida albicans. J Bacteriol 151:1118–1122
Woolford CA, Lagree K, Aleynikov T, Mitchell AP (2017) Negative control of Candida albicans filamentation-associated gene expression by essential protein kinase gene KIN28. Curr Genet 63:1073–1079. https://doi.org/10.1007/s00294-017-0705-8
Xu XL, Lee RT, Fang HM, Wang YM, Li R, Zou H, Zhu Y, Wang Y (2008) Bacterial peptidoglycan triggers Candida albicans hyphal growth by directly activating the adenylyl cyclase Cyr1p. Cell Host Microbe 4:28–39. https://doi.org/10.1016/j.chom.2008.05.014
Zhao X, Oh SH, Cheng G, Green CB, Nuessen JA, Yeater K, Leng RP, Brown AJ, Hoyer LL (2004) ALS3 and ALS8 represent a single locus that encodes a Candida albicans adhesin; functional comparisons between Als3p and Als1p. Microbiology 150:2415–2428. https://doi.org/10.1099/mic.0.26943-0
Acknowledgements
This work was supported by the National Natural Science Foundation of China Grant 31770162 to Y. L. and Grant 31700133 to C. S.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Kupiec.
Rights and permissions
About this article
Cite this article
Su, C., Yu, J. & Lu, Y. Hyphal development in Candida albicans from different cell states. Curr Genet 64, 1239–1243 (2018). https://doi.org/10.1007/s00294-018-0845-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-018-0845-5