Abstract
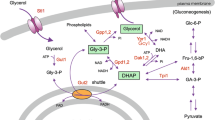
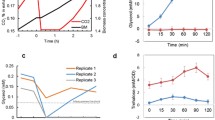
In this study the yeast Saccharomyces cerevisiae, which is a genetically tractable model for analysis of osmoregulation, has been used for analysis of heterologous aquaporins. Aquaporin water channels play important roles in the control of water homeostasis in individual cells and multicellular organisms. We have investigated the effects of functional expression of the mammalian aquaporins AQP1 and AQP5 and the aquaglyceroporins AQP3 and AQP9. Expression of aquaporins caused moderate growth inhibition under hyperosmotic stress, while expression of aquaglyceroporins mediated strong growth inhibition due to glycerol loss. Water transport was monitored in protoplasts, where the kinetics of bursting was influenced by presence of aquaporins but not aquaglyceroporins. We observed glycerol transport through aquaglyceroporins, but not aquaporins, in a yeast strain deficient in glycerol production, whose growth depends on glycerol inflow. In addition, a gene reporter assay allowed to indirectly monitor the effect of AQP9-mediated enhanced glycerol loss on osmoadaptation. Transport activity of certain aqua(glycero)porins was diminished by low pH or CuSO4, suggesting that yeast can potentially be used for screening of putative aquaporin inhibitors. We conclude that yeast is a versatile system for functional studies of aquaporins, and it can be developed to screen for compounds of potential pharmacological use.







Similar content being viewed by others
References
Albertyn J, Hohmann S, Thevelein JM, Prior BA (1994) GPD1, which encodes glycerol-3-phosphate dehydrogenase, is essential for growth under osmotic stress in Saccharomyces cerevisiae, and its expression is regulated by the high-osmolarity glycerol response pathway. Mol Cell Biol 14:4135–4144
Ansell R, Granath K, Hohmann S, Thevelein JM, Adler L (1997) The two isoenzymes for yeast NAD+-dependent glycerol 3-phosphate dehydrogenase encoded by GPD1 and GPD2 have distinct roles in osmoadaptation and redox regulation. EMBO J 16:2179–2187
Bill RM (2001) Yeast—a panacea for the structure-function analysis of membrane proteins? Curr Genet 40:157–171
Bonhivers M, Carbrey JM, Gould SJ, Agre P (1998) Aquaporins in Saccharomyces. Genetic and functional distinctions between laboratory and wild-type strains. J Biol Chem 273:27565–27572
Brooks HL, Regan JW, Yool AJ (2000) Inhibition of aquaporin-1 water permeability by tetraethylammonium: involvement of the loop E pore region. Mol Pharmacol 57:1021–1026
Carbrey JM, Gorelick-Feldman DA, Kozono D, Praetorius J, Nielsen S, Agre P (2003) Aquaglyceroporin AQP9: solute permeation and metabolic control of expression in liver. Proc Natl Acad Sci USA 100:2945–2950
Chen DC, Yang BC, Kuo TT (1992) One-step transformation of yeast in stationary phase. Curr Genet 21:83–84
Coury LA, Mathai JC, Prasad GV, Brodsky JL, Agre P, Zeidel ML (1998) Reconstitution of water channel function of aquaporins 1 and 2 by expression in yeast secretory vesicles. Am J Physiol 274:F34–F42
Daniels MJ, Wood MR, Yeager M (2006) In vivo functional assay of a recombinant aquaporin in Pichia pastoris. Appl Environ Microbiol 72:1507–1514
Dihazi H, Kessler R, Eschrich K (2004) High osmolarity glycerol (HOG) pathway-induced phosphorylation and activation of 6-phosphofructo-2-kinase are essential for glycerol accumulation and yeast cell proliferation under hyperosmotic stress. J Biol Chem 279:23961–23968
Eriksson P, Andre L, Ansell R, Blomberg A, Adler L (1995) Cloning and characterization of GPD2, a second gene encoding sn-glycerol 3-phosphate dehydrogenase (NAD+) in Saccharomyces cerevisiae, and its comparison with GPD1. Mol Microbiol 17:95–107
Hashimoto K, Saito M, Matsuoka H, Iida K, Iida H (2004) Functional analysis of a rice putative voltage-dependent Ca2+ channel, OsTPC1, expressed in yeast cells lacking its homologous gene CCH1. Plant Cell Physiol 45:496–500
Hohmann I, Bill RM, Kayingo I, Prior BA (2000) Microbial MIP channels. Trends Microbiol 8:33–38
Hohmann S (2002) Osmotic stress signaling and osmoadaptation in yeasts. Microbiol Mol Biol Rev 66:300–372
Hohmann S, Nielsen S, Agre P (eds) (2001) Aquaporins. Academic
Ikeda M, Beitz E, Kozono D, Guggino WB, Agre P, Yasui M (2002) Characterization of aquaporin-6 as a nitrate channel in mammalian cells. Requirement of pore-lining residue threonine 63. J Biol Chem 277:39873–39879
Jung JS, Preston GM, Smith BL, Guggino WB, Agre P (1994) Molecular structure of the water channel through aquaporin CHIP. The hourglass model. J Biol Chem 269:14648–14654
Karlgren S, Pettersson N et al (2005) Conditional osmotic stress in yeast: a system to study transport through aquaglyceroporins and osmostress signaling. J Biol Chem 280:7186–7193
King LS, Yasui M, Agre P (2000) Aquaporins in health and disease. Mol Med Today 6:60–65
Kiser GL, Gentzsch M, Kloser AK, Balzi E, Wolf DH, Goffeau A, Riordan JR (2001) Expression and degradation of the cystic fibrosis transmembrane conductance regulator in Saccharomyces cerevisiae. Arch Biochem Biophys 390:195–205
Klipp E, Nordlander B, Kruger R, Gennemark P, Hohmann S (2005) Integrative model of the response of yeast to osmotic shock. Nat Biotechnol 23:975–982
Kuriyama H, Shimomura I et al (2002) Coordinated regulation of fat-specific and liver-specific glycerol channels, aquaporin adipose and aquaporin 9. Diabetes 51:2915–2921
Ladds G, Goddard A, Davey J (2005) Functional analysis of heterologous GPCR signalling pathways in yeast. Trends Biotechnol 23:367–373
Laize V, Rousselet G et al (1995) Functional expression of the human CHIP28 water channel in a yeast secretory mutant. FEBS Lett 373:269–274
Laize V, Tacnet F, Ripoche P, Hohmann S (2000) Polymorphism of Saccharomyces cerevisiae aquaporins. Yeast 16:897–903
Larsson K, Ansell R, Eriksson P, Adler L (1993) A gene encoding sn-glycerol 3-phosphate dehydrogenase (NAD+) complements an osmosensitive mutant of Saccharomyces cerevisiae. Mol Microbiol 10:1101–1111
Li H, Black PN, DiRusso CC (2005) A live-cell high-throughput screening assay for identification of fatty acid uptake inhibitors. Anal Biochem 336:11–19
Liu Z, Carbrey JM, Agre P, Rosen BP (2004) Arsenic trioxide uptake by human and rat aquaglyceroporins. Biochem Biophys Res Commun 316:1178–1185
Liu Z, Shen J, Carbrey JM, Mukhopadhyay R, Agre P, Rosen BP (2002) Arsenite transport by mammalian aquaglyceroporins AQP7 and AQP9. Proc Natl Acad Sci USA 99:6053–6058
Luyten K, Albertyn J, Skibbe WF, Prior BA, Ramos J, Thevelein JM, Hohmann S (1995) Fps1, a yeast member of the MIP family of channel proteins, is a facilitator for glycerol uptake and efflux and is inactive under osmotic stress. EMBO J 14:1360–1371
Ma T, Yang B, Verkman AS (1997) Cloning of a novel water and urea-permeable aquaporin from mouse expressed strongly in colon, placenta, liver, and heart. Biochem Biophys Res Commun 240:324–328
Manley GT, Fujimura M et al (2000) Aquaporin-4 deletion in mice reduces brain edema after acute water intoxication and ischemic stroke. Nat Med 6:159–163
Matejckova-Forejtova A, Kinclova O, Sychrova H (1999) Degradation of Candida albicans Can1 permease expressed in Saccharomyces cerevisiae. FEMS Microbiol Lett 176:257–262
Morishita Y, Sakube Y, Sasaki S, Ishibashi K (2004) Molecular mechanisms and drug development in aquaporin water channel diseases: aquaporin superfamily (superaquaporins): expansion of aquaporins restricted to multicellular organisms. J Pharmacol Sci 96:276–279
Nejsum LN, Kwon TH et al (2002) Functional requirement of aquaporin-5 in plasma membranes of sweat glands. Proc Natl Acad Sci USA 99:511–516
Niemietz CM, Tyerman SD (2002) New potent inhibitors of aquaporins: silver and gold compounds inhibit aquaporins of plant and human origin. FEBS Lett 531:443–447
Pahlman AK, Granath K, Ansell R, Hohmann S, Adler L (2001) The yeast glycerol 3-phosphatases Gpp1p and Gpp2p are required for glycerol biosynthesis and differentially involved in the cellular responses to osmotic, anaerobic, and oxidative stress. J Biol Chem 276:3555–3563
Pettersson N, Filipsson C, Becit E, Brive L, Hohmann S (2005) Aquaporins in yeasts and filamentous fungi. Biol Cell 97:487–500
Preston GM, Carroll TP, Guggino WB, Agre P (1992) Appearance of water channels in Xenopus oocytes expressing red cell CHIP28 protein. Science 256:385–387
Raina S, Preston GM, Guggino WB, Agre P (1995) Molecular cloning and characterization of an aquaporin cDNA from salivary, lacrimal, and respiratory tissues. J Biol Chem 270:1908–1912
Reiser V, Raitt DC, Saito H (2003) Yeast osmosensor Sln1 and plant cytokinin receptor Cre1 respond to changes in turgor pressure. J Cell Biol 161:1035–1040
Saadoun S, Papadopoulos MC, Hara-Chikuma M, Verkman AS (2005) Impairment of angiogenesis and cell migration by targeted aquaporin-1 gene disruption. Nature 434:786–792
Sarkar G, Sommer SS (1990) The “megaprimer” method of site-directed mutagenesis. Biotechniques 8:404–407
Suga S, Maeshima M (2004) Water channel activity of radish plasma membrane aquaporins heterologously expressed in yeast and their modification by site-directed mutagenesis. Plant Cell Physiol 45:823–830
Takata K, Matsuzaki T, Tajika Y (2004) Aquaporins: water channel proteins of the cell membrane. Prog Histochem Cytochem 39:1–83
Tamas MJ, Karlgren S et al (2003) A short regulatory domain restricts glycerol transport through yeast Fps1p. J Biol Chem 278:6337–6345
Tamas MJ, Luyten K et al (1999) Fps1p controls the accumulation and release of the compatible solute glycerol in yeast osmoregulation. Mol Microbiol 31:1087–1104
Thomas BJ, Rothstein R (1989) Elevated recombination rates in transcriptionally active DNA. Cell 56:619–630
Verkman AS (2005) More than just water channels: unexpected cellular roles of aquaporins. J Cell Sci 118:3225–3232
Wieczorke R, Dlugai S, Krampe S, Boles E (2003) Characterisation of mammalian GLUT glucose transporters in a heterologous yeast expression system. Cell Physiol Biochem 13:123–134
Zelenina M, Bondar AA, Zelenin S, Aperia A (2003) Nickel and extracellular acidification inhibit the water permeability of human aquaporin-3 in lung epithelial cells. J Biol Chem 278:30037–30043
Zelenina M, Tritto S, Bondar AA, Zelenin S, Aperia A (2004) Copper inhibits the water and glycerol permeability of aquaporin-3. J Biol Chem 279:51939–51943
Acknowledgments
We thank members of the group for helpful discussions and for critical reading of the manuscript. This work was supported by grants from the European Commission (contracts BIO4-CT98-0024 and FMRX-CT96-0128 to SH and contract LSHG-CT-2004-504601 to RMB) and the Swedish Research Council (research position and research grant to SH).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by L. Tomaska
Rights and permissions
About this article
Cite this article
Pettersson, N., Hagström, J., Bill, R.M. et al. Expression of heterologous aquaporins for functional analysis in Saccharomyces cerevisiae . Curr Genet 50, 247–255 (2006). https://doi.org/10.1007/s00294-006-0092-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-006-0092-z