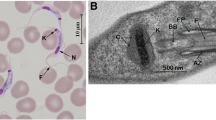
Abstract
Kinetoplastids are flagellated protozoans, whose members include the pathogens Trypanosoma brucei, T. cruzi and Leishmania species, that are considered among the earliest diverging eukaryotes with a mitochondrion. This organelle has become famous because of its many unusual properties, which are unique to the order Kinetoplastida, including an extensive kinetoplast DNA network and U-insertion/deletion type RNA editing of its mitochondrial transcripts. In the last decade, considerable progress has been made in elucidating the complex machinery of RNA editing. Moreover, our understanding of the structure and replication of kinetoplast DNA has also dramatically improved. Much less however, is known, about the developmental regulation of RNA editing, its integration with other RNA maturation processes, stability of mitochondrial mRNAs, or evolution of the editing process itself. Yet the profusion of genomic data recently made available by sequencing consortia, in combination with methods of reverse genetics, hold promise in understanding the complexity of this exciting organelle, knowledge of which may enable us to fight these often medically important protozoans.


Similar content being viewed by others
References
Abraham JM, Feagin JE, Stuart K (1988) Characterization of cytochrome c oxidase III transcripts that are edited only in the 3′ region. Cell 55:267–272
Abu-Elneel K, Robinson DR, Drew ME, Englund PT, Shlomai J (2001) Intramitochondrial localization of universal minicircle sequence-binding protein, a trypanosomatid protein that binds kinetoplast minicircle replication origins. J Cell Biol 153:725–733
Adler BK, Harris ME, Bertrand KI, Hajduk SL (1991) Modification of Trypanosoma brucei mitochondrial rRNA by posttranscriptional 3′ polyuridine tail formation. Mol Cell Biol 11:5878–5884
Alfonzo JD, Thiemann OH, Simpson L (1998) Purification and characterization of MAR1—mitochondrial associated ribonuclease from Leishmania tarentolae. J Biol Chem 273:30003–30011
Anderson L et al (1981) Identification of mitochondrial proteins and some of their precursors in two-dimensional electrophoretic maps of human cells. Proc Natl Acad Sci USA 78:2407–2411
Aphasizhev R, Sbicego S, Peris M, Jang SH, Aphasizheva I, Simpson AM, Rivlin A, Simpson L (2002) Trypanosome mitochondrial 3′ terminal uridylyl transferase (TUTase): the key enzyme in U-insertion/deletion RNA editing. Cell 108:637–648
Aphasizhev R, Aphasizheva I, Nelson RE, Simpson L (2003a) A 100-kD complex of two RNA-binding proteins from mitochondria of Leishmania tarentolae catalyzes RNA annealing and interacts with several RNA editing components. RNA 9:62–76
Aphasizhev R, Aphasizheva I, Simpson L (2003b) A tale of two TUTases. Proc Natl Acad Sci USA 100:10617–10622
Aphasizhev R, Aphasizheva I, Nelson RE, Gao GH, Simpson AM, Kang XD, Falick AM, Sbicego S, Simpson L (2003c) Isolation of a U-insertion/deletion editing complex from Leishmania tarentolae mitochondria. EMBO J 22:913–924
Aphasizhev R, Aphasizheva I, Simpson L (2004) Multiple terminal uridylyltransferases of trypanosomes. FEBS Lett 572:15–18
Aphasizheva I, Aphasizhev R, Simpson L (2004) RNA-editing terminal uridylyl transferase 1—Identification of functional domains by mutational analysis. J Biol Chem 279:24123–24130
Arts G-J, Sloof P, Benne R. (1995) A possible role for the guide RNA U-tail as a specificity determinant in formation of guide RNA-messenger RNA chimeras in mitochondrial extracts of Crithidia fasciculata. Mol Biochem Parasitol. 73:211–222
Avliyakulov NK, Lukeš J, Ray DS (2004) Mitochondrial histone-like DNA-binding proteins are essential for normal cell growth and mitochondrial function in Crithidia fasciculata. Eukaryot Cell 3:518–526
Bakalara N, Simpson AM, Simpson L (1989). The leishmania kinetoplast-mitochondrion contains terminal uridylyltransferase and RNA ligase activies. J Biol Chem 264:18679–18686
Basselin M, Badet-Denisot M-A, Robert-Gero M (1998) Modification of kinetoplast DNA minicircle composition in pentamidine-resistant Leishmania. Acta Trop 70:43–61
Beattie DS, Howton MM (1996) The presence of rotenone-sensitive NADH dehydrogenase in the long slender bloodstream and the procyclic forms of Trypanosoma brucei brucei. Eur J Biochem 241:888–894
Benne R, Van den Burg J, Brakenhoff JPJ, Sloof P, Van Boom JH, Tromp MC (1986) Major transcript of the frameshifted coxII gene from trypanosome mitochondria contains 4 nucleotides that are not encoded in the DNA. Cell 46:819–826
Benne R (1990) RNA editing in trypanosomes—is there a message? Trends Genet 6:177–181
Berriman M, et al. (2005) The genome of the African trypanosome Trypanosoma brucei. Science 309:416–422
Bessolitsina EA, Fedyakov AV, Merzlyak EM, Kolesnikov AA (2005) The ATPase subunit 6 gene of Leptomonas seymouri (Trypanosomatidae) is transcribed and edited as a polycistronic mRNA. Mol Biol (Moscow) 39:53–57
Besteiro S, Barrett MP, Riviere L, Bringaud F (2005) Energy generation in insect stages of Trypanosoma brucei: metabolism in flux. Trends Parasitol 21:185–191
Bhat GJ, Koslowsky DJ, Feagin JE, Smiley BL, Stuart K (1990) An extensively edited mitochondrial transcript in kinetoplastids encodes a protein homologous to ATPse subunit 6. Cell 61:885–894
Bhat GJ, Souza AE, Feagin JE, Stuart K (1992) Transcript-specific developmental regulation of polyadenylation in Trypanosoma brucei mitochondria. Mol Biochem Parasitol 52:231–240
Birkenmeyer L, Sugisaki H, Ray DS (1987) Structural characterization of site-specific discontinuities associated with replication origins of minicircle DNA from Crithidia fasciculata. J Biol Chem 262:2384–2392
Blom D, de Haan A, van den Berg M, Sloof P, Jirků M, Lukeš J, Benne R (1998) RNA editing in the free-living bodonid Bodo saltans. Nucleic Acids Res 26:1205–1213
Blom D, de Haan A, van den Burg J, van den Berg M, Sloof P, Jirků M, Lukeš J, Benne R (2000) Mitochondrial minicircles in the free-living bodonid Bodo saltans contain two gRNA gene cassettes and are not found in large networks. RNA 6:1–15
Blom D, van den Burg J, Breek CKD, Speijer D, Muijsers AO, Benne R (2001) Cloning and characterization of two guide RNA-binding proteins from mitochondria of Crithidia fasciculata: gBP27, a novel protein, and gBP29, the orthologue of Trypanosoma brucei gBP21. Nucleic Acids Res 29:2950–2962
Blum B, Bakalara N, Simpson L (1990) A model for RNA editing in kinetoplastid mitochondria: guide RNA molecules transcribed from maxicircle DNA provide the edited information. Cell 60:198–198
Blum B, Sturm NR, Simpson AM, Simpson L (1991) Chimeric gRNA messenger-RNA molecules with oligo(U) tails covalently linked at sites of RNA editing suggest that U-addition occurs by transesterification. Cell 65:543–550
Blum B, Simpson L (1992) Formation of guide RNA messenger-RNA chimeric molecules in vitro, the initial step of RNA editing, is dependent on an anchor sequence. Proc Natl Acad Sci USA 89:11944–11948
Bochud-Allemann N, Schneider A (2002) Mitochondrial substrate level phosphorylation is essential for growth of procyclic Trypanosoma brucei. J Biol Chem 277:32849–32854
Borst P (1991) Why kinetoplast DNA networks? Trends Genet 7:139–141
Brecht M, Niemann M, Schulter E, Müller UF, Stuart K, Göringer HU (2005) TbMP42, a protein component of the RNA editing complex in African trypanosomes, has endo-exoribonuclease activity. Mol Cell 17:621–630
Bringaud F, Peris M, Zen KH, Simpson L (1995) Characterization of two nuclear-encoded protein components of mitochondrial ribonucleoprotein complexes from Leishmania tarentolae. Mol Biochem Parasitol 71:65–79
Burd CG, Dreyfuss G (1994) Conserved structures and diversity of functions of RNA-binding proteins. Science 265:615–21
Burgess MLK, Stuart K (2000) Sequence bias in edited kinetoplastid RNAs. RNA 6:1492–1497
Burgess MLK, Heidmann S, Stuart K (1999) Kinetoplastid RNA editing does not require the terminal 3′ hydroxyl of guide RNA, but modifications to the guide RNA terminus can inhibit in vitro U-insertion. RNA 5:883–892
Carpousis AJ, Vanzo NF, Raynal LC (1999) mRNA degradation. A tale of poly(A) and multiprotein machines. Trends Genet 15:24–28
Carrillo R, Thiemann OH, Alfonzo JD, Simpson L (2001) Uridine insertion/deletion RNA editing in Leishmania tarentolae mitochondria shows cell cycle dependence. Mol Biochem Parasitol 113:175–181
Cavalier-Smith T (1997) Cell and genome coevolution: facultative anaerobiosis, glycosomes and kinetoplastan RNA editing. Trends Genet 13:6–9
Cech TR (1991) RNA editing: world’s smallest introns? Cell 64:667–669
Chaudhuri M, Ajayi W, Hill GC (1998) Biochemical and molecular properties of the Trypanosoma brucei alternative oxidase. Mol Biochem Parasitol 95:53–68
Chen JH, Rauch CA, White JH, Englund PT, Cozzarelli NR (1995a) The topology of the kinetoplast DNA network. Cell 80:61–69
Chen JH, Rauch CA, White JH, Cozzarelli NR, Englund PT (1995b) Changes in network topology during the replication of kinetoplast DNA. EMBO J 14:6339–6347
Clement SL, Mingler MK, Koslowsky DJ (2004) An intragenic guide RNA location suggests a complex mechanism for mitochondrial gene expression in Trypanosoma brucei. Eukaryot Cell 3:862–869
Corell RA, Feagin JE, Riley GR, Strickland T, Guderian JA, Myler PJ, Stuart K (1993) Trypanosoma brucei minicircles encode multiple guide RNAs which can direct editing of extensively overlapping sequences. Nucleic Acids Res 21:4313–4320
Corell RA, Read LK, Riley GR, Nellissery JK, Allen TE, Kable ML, Wachal MD, Seiwert SD, Myler PJ, Stuart KD (1996) Complexes from Trypanosoma brucei that exhibit deletion editing and other editing-associated properties. Mol Cell Biol 16:1410–1418
Covello PS, Gray MW (1993) On the evolution of RNA editing. Trends Genet 9:265–268
Cruz-Reyes J, Rusché LN, Sollner-Webb B (1998) Trypanosoma brucei U-insertion and U-deletion activities co-purify with an enzymatic editing complex but are differentially optimized. Nucleic Acids Res 26:3634–3639
Cruz-Reyes J, Piller KJ, Rusché LN, Mukherjee M, Sollner-Webb B (1998a) Unexpected electrophoretic migration of RNA with different 3′ termini causes a RNA sizing ambiguity that can be resolved using nuclease P1-generated sequencing ladders. Biochemistry 37:6059–6064
Cruz-Reyes J, Rusché LN, Piller KJ, Sollner-Webb B (1998b) Trypanosma brucei RNA editing: adenosine nucleotides inversely affect U-deletion and U-insertion reactions at mRNA cleavage. Mol Cell 1:401–409
Cruz-Reyes J, Zhelonkina A, Rusché L, Sollner-Webb B (2001) Trypanosome RNA editing: simple guide RNA features enhance U-deletion 100-fold. Mol Cell Biol 21:884–892
Cruz-Reyes J, Zhelonkina AG, Huang CE, Sollner-Webb B (2002) Distinct functions of two RNA ligases in active Trypanosoma brucei RNA editing complexes. Mol Cell Biol 22:4652–4660
De la Cruz V, Lake JA, Simpson AM, Simpson L (1985) A minimal ribosomal-RNA-sequence and secondary structure of the 9S kinetoplast ribosomal-RNA from Leishmania tarentolae. Proc Natl Acad Sci USA 82:1401–1405
Decker CJ, Sollner-Webb B (1990) RNA editing involves indiscriminate U changes throughout precisely defined editing domains. Cell 61:1001–1011
Doležel D, Jirků M, Maslov DA, Lukeš J (2000) Phylogeny of the bodonid flagellates (Kinetoplastida) based on small subunit rRNA gene sequences. Int J Syst Evol Microbiol 50:1943–1951
Domingo GJ, Palazzo SS, Wang B, Pannicucci B, Salavati R, Stuart KD (2003) Dyskinetoplastic Trypanosoma brucei contains functional editing complexes. Eukaryot Cell 2:569–577
Downey N, Hines JC, Sinha KM, Ray DS (2005) Mitochondrial DNA ligases of Trypanosoma brucei. Eukaryot Cell 4:765–774
Drew ME, Englund PT (2001) Intramitochondrial location and dynamics of Crithidia fasciculata kinetoplast minicircle replication intermediates. J Cell Biol 153:735–743
Drozdz M, Palazzo SS, Salavati R, O’Rear J, Clayton C, Stuart K (2002) TbMP81 is required for RNA editing in Trypanosoma brucei. EMBO J 21:1791–1799
Dyková I, Fiala I, Lom J, Lukeš J (2003) Perkinsiella amoebae-like endosymbionts of Neoparamoeba spp., relatives of the kinetoplastid Ichthyobodo. Europ J Protistol 39:37–52
Dziembowski A, Piwowarski J, Hoser R, Minczuk M, Dmochowska A, Siep M, van der Spek H, Grivell L, Stepien PP (2003) The yeast mitochondrial degradosome, its composition, interplay between RNA helicase and RNAse activities and the role in mitochondrial RNA metabolism. J Biol Chem 278:1603–1611
El-Sayed NM, et al. (2005) The genome sequence of Trypanosoma cruzi, etiologic agent of Chagas disease. Science 309:409–415
Engel ML, Ray DS (1999) The kinetoplast structure-specific endonuclease I is related to the 5′ exo/endonuclease domain of bacterial DNA polymerase I and colocalizes with the kinetoplast topoisomerase II and DNA polymerase beta during replication. Proc Natl Acad Sci USA 96:8455–8460
Ernst NL, Panicucci B, Igo RP, Panigrahi AK, Salavati R, Stuart K (2003) TbMP57 is a 3′ terminal uridylyl transferase (TUTase) of the Trypanosoma brucei editosome. Mol Cell 11:1525–1536
Estévez AM, Kierszenbaum F, Wirtz E, Bringaud F, Grunstein J, Simpson L. (1999) Knockout of the glutamate dehydrogenase gene in bloodstream Trypanosoma brucei in culture has no effect on editing of mitochondrial mRNAs. Mol Biochem Parasitol. 100(1):5–17
Estévez AM, Simpson L (1999) Uridine insertion/deletion RNA editing in trypanosome mitochondria—a review. Gene 240:247–260
Feagin JE, Stuart K (1988) Developmental aspects of uridine addition within mitochondrial transcripts of Trypanosoma brucei. Mol Cell Biol 8:1259–1265
Feagin JE, Abraham JM, Stuart K (1988a) Extensive editing of the cytochrome c oxidase III transcript in Trypanosoma brucei. Cell 53:413–422
Feagin JE, Shaw JM, Simpson L, Stuart K (1988b) Creation of AUG initiation codons by addition of uridines within cytochrome b transcripts of kinetoplastids. Proc Natl Acad Sci USA 85:539–543
Fernandez AP, Nelson K, Beverly SM (1993) Evolution of nuclear ribosomal RNAs in kinetoplastid protozoa: perspective on the age and origins of parasitism. Proc Natl Acad Sci USA 90:11608–11612
Gao G, Kapushoc ST, Simpson AM, Thiemann OH, Simpson L (2001) Guide RNAs of the recently isolated LEM125 strain of Leishmania tarentolae: an unexpected complexity. RNA 7:1335–1347
Gao G, Simpson L (2003) Is the Trypanosoma brucei REL1 RNA ligase specific for U-deletion RNA editing, and is the REL2 RNA ligase specific for U-insertion editing? J Biol Chem 278:27570–27574
Gao G, Simpson AM, Kang X, Rogers K, Nebohacova M, Li F, Simpson L (2005) Functional complementation of Trypanosoma brucei RNA in vitro editing with recombinant RNA ligase. Proc Natl Acad Sci USA 102:4712–4717
Gažiová I, Lukeš J (2003). Mitochondrial and nuclear localization of topoisomerase II in the flagellate Bodo saltans (Kinetoplastida), a species with non-catenated kinetoplast DNA. J Biol Chem 278:10900–10907
Golden DE, Hajduk SL (2005) The 3′-untranslated region of cytochrome oxidase II mRNA functions in RNA editing of African trypanosomes `exclusively as a cis guide RNA. RNA 11:29–37
Grams J, McManus MT, Hajduk SL (2000) Processing of polycistronic guide RNAs is associated with RNA editing complexes in Trypanosoma brucei. EMBO J 19:5525–5532
Grams J, Morris JC, Drew ME, Wang ZF, Englund PT, Hajduk SL (2002) A trypanosome mitochondrial RNA polymerase is required for transcription and replication. J Biol Chem 277:16952–16959
Gray MW, Lang BF, Burger G (2004) Mitochondria of protists. Annu Rev Genet 38:477–524
Guilbride DL, Englund PT (1998) The replication mechanism of kinetoplast DNA networks in several trypanosomatid species. J Cell Sci 111:675–679
Hajduk SL, Klein VA, Englund PT (1984) Replication of kinetoplast DNA maxicircles. Cell 36:483–492
Halbig K, De Nova-Ocampo M, Cruz-Reyes J (2004) Complete cycles of bloodstream trypanosome RNA editing in vitro. RNA 10:914–920
Hancock K, Hajduk SL (1990) The mitochondrial tRNAs of Trypanosoma brucei are nuclear encoded. J Biol Chem 265:19208–19215
Harris ME, Hajduk SL (1992) Kinetoplastid RNA editing: in vitro formation of cytochrome b gRNA-mRNA chimeras from synthetic substrate RNAs. Cell 68:1091–1099
Hayman ML, Read LK (1999) Trypanosoma brucei RBP16 is a mitochondrial Y-box family protein with guide RNA binding activity. J Biol Chem 274:12067–12074
Hayman ML, Miller MM, Chandler DM, Goulah CC, Read LK (2001) The trypanosome homolog of human p32 interacts with RBP16 and stimulates its gRNA binding activity. Nucleic Acids Res 29:5216–5225
Hermann T, Schmid B, Heumann H, Göringer HU (1997) A three-dimensional working model for a guide RNA from Trypanosoma brucei. Nucleic Acids Res 25:2311–2318
Hernandez FR, Turrens JF (1998) Rotenone at high concentrations inhibits NADH-fumarate reductase and the mitochondrial respiratory chain of Trypanosoma brucei and T. cruzi. Mol Biochem Parasitol 93:135–137
Horváth A, Neboháčová M, Lukeš J, Maslov DA (2002) Unusual polypeptide synthesis in the kinetoplast-mitochondria from Leishmania tarentolae—Identification of individual de novo translation products. J Biol Chem 277:7222–7230
Horváth A, Berry EA, Maslov DA (2000a) Translation of the edited mRNA for cytochrome b in trypanosome mitochondria. Science 287:1639–1640
Horváth A, Kingan TG, Maslov DA (2000b) Detection of the mitochondrially encoded cytochrome c oxidase subunit I in the trypanosomatid protozoan Leishmania tarentolae. Evidence for translation of unedited mRNA in the kinetoplast. J Biol Chem 275:17160–17165
Horváth A, Horáková E, Dunajčíková P, Verner Z, Pravdová E, Šlapetová I, Cuninková L, Lukeš J (2005) Down-regulation of the nuclear-encoded subunits of the complexes III and IV disrupts their respective complexes but not complex I in procyclic Trypanosoma brucei. Mol Microbiol 58:116–130
Huang CE, Cruz-Reyes J, Zhelonkina AG, O’Hearn S, Wirtz E, Sollner-Webb B (2001) Roles for ligases in the RNA editing complex of Trypanosoma brucei: band IV is needed for U-deletion and RNA repair. EMBO J 20:4694–4703
Huang CE, O’Hearn SF, Sollner-Webb B (2002) Assembly and function of the RNA editing complex in Trypanosoma brucei requires band III protein. Mol Cell Biol 22:3194–3203
Igo RP Jr, Palazzo SS, Burgess ML, Panigrahi AK, Stuart K (2000) Uridylate addition and RNA ligation contribute to the specificity of kinetoplastid insertion RNA editing. Mol Cell Biol 20:8447–8457
Igo RP Jr, Lawson SD, Stuart K (2002) RNA sequence and base pairing effects on insertion editing in Trypanosoma brucei. Mol Cell Biol 22:567–1576
Ivens AC et al (2005) The genome of the kinetoplastid parasite, Leishmania major. Science 309:436–442
Kable ML, Seiwert SD, Heidmann S, Stuart K (1996) RNA editing: a mechanism for gRNA-specified uridylate insertion into precursor mRNA. Science 273:1189–1195
Kang XD, Falick AM, Nelson RE, Gao GH, Rogers K, Aphasizhev R, Simpson L (2004) Disruption of the zinc finger motifs in the Leishmania tarentolae LC-4 (=TbMP63) L-complex editing protein affects the stability of the L-complex. J Biol Chem 279:3893–3899
Kang XD, Rogers K, Gao GH, Falick AM, Zhou S, Simpson L (2005) Reconstitution of uridine-deletion precleaved RNA editing with two recombinant enzymes. Proc Natl Acad Sci USA 102:1017–1022
Kao CY, Read LK (2005) Opposing effect of polyadenylation on the stability of edited and unedited mitochondrial RNAs in Trypanosoma brucei. Mol Cell Biol 25:1634–1644
Kapushoc ST, Simpson L (1999) In vitro uridine insertion RNA editing mediated by cis-acting guide RNAs. RNA 5:656–669
Kim KS, Teixeira SM, Kirchhoff LV, Donelson JE (1994) Transcription and editing of cytochrome oxidase II RNAs in Trypanosoma cruzi. J Biol Chem 269:1206–1211
Klingbeil MM, Englund PT (2004) Closing the gaps in kinetoplast DNA network replication. Proc Natl Acad Sci USA 101:4333–4334
Klingbeil MM, Drew ME, Liu YN, Morris JC, Motyka SA, Saxowsky TT, Wang ZF, Englund PT (2001) Unlocking the secrets of trypanosome kinetoplast DNA network replication. Protist 152:255–262
Klingbeil MM, Motyka SA, Englund PT (2002) Multiple mitochondrial DNA polymerases in Trypanosoma brucei. Mol Cell 10:175–186
Köller J, Nörskau G, Paul AS, Stuart K, Göringer HU (1994) Different Trypanosoma brucei guide RNA molecules associate with an identical complement of mitochondrial proteins in vitro. Nucleic Acids Res 22:1988–1995
Köller J, Müller UF, Schmid B, Missel A, Kruft V, Stuart K, Göringer HU (1997) Trypanosoma brucei gBP21. An arginine-rich mitochondrial protein that binds to guide RNA with high affinity. J Biol Chem 272:3749–3757
Koslowsky DJ, Bhat GJ, Read LK, Stuart K (1991) Cycles of progressive realignment of gRNA with mRNA in RNA editing. Cell 67:537–546
Koslowsky DJ, Yahampath G (1997) Mitochondrial mRNA 3′ cleavage/polyadenylation and RNA editing in Trypanosoma brucei are independent events. Mol Biochem Parasitol 90:81–94
Koslowsky DJ, Bhat GJ, Perrolaz AL, Feagin JE, Stuart K (1990) The MURF3 gene of Trypanosoma brucei contains multiple domains of extensive editing and is homologous to a subunit of NADH dehydrogenase. Cell 62:901–911
Lambert L, Müller UF, Souza AE, Göringer HU (1999) The involvement of gRNA-binding protein gBP21 in RNA editing—an in vitro and in vivo analysis. Nucleic Acids Res 27:1429–1436
Landweber LF, Gilbert W (1993) RNA editing as a source of genetic variation. Nature 363:179–182
Lang BF, Burger G, O’Kelly CJ, Cedergren R, Golding GB, Lemieux C, Sankoff D, Turmel M, Gray MW (1997) An ancestral mitochondrial DNA resembling a eubacterial genome in miniature. Nature 387:493–497
Lee S-Y, Lee S-T, Chang K-P (1992) Transkinetoplastidy—a novel phenomenon involving bulk alterations of mitochondrion-kinetoplast DNA of a trypanosomatid protozoan. J Protozool 39:190–196
Leegwater P, Speijer D, Benne R (1995) Identification by UV cross-linking of oligo(U)-binding proteins in mitochondria of the insect trypanosomatid Crithidia fasciculata. Eur J Biochem 227:780–786
Leung SS, Koslowsky DJ (2001a) Interactions of mRNA and gRNAs involved in trypanosome mitochondrial RNA editing: structure probing of an mRNA bound to its cognate gRNA. RNA 7:1803–1816
Leung SS, Koslowsky DJ (2001b) RNA editing in Trypanosoma brucei: characterization of gRNA U-tail interactions with partially edited mRNA substrates. Nucleic Acids Res 29:703–709
Li C, Englund PT (1997) A mitochondrial DNA primase from the trypanosomatid Crithidia fasciculata. J Biol Chem 272:20878–20792
Linial M, Shlomai J (1987) Sequence-directed bent DNA helix is the specific binding site for Crithidia fasciculata nicking enzyme. Proc Natl Acad Sci USA 84:8205–8209
Liu B, Liu Y, Motyka SA, Agbo EEC, Englund PT (2005) Fellowship of the rings: the replication of kinetoplast DNA. Trends Parasitol 21:363–369
Lukeš J, Arts GJ, Van den Burg J, deHaan A, Opperdoes F, Sloof P, Benne R (1994) Novel pattern of editing regions in mitochondrial transcripts of the cryptobiid Trypanoplasma borreli. EMBO J 13:5086–5098
Lukeš J, Jirků M, Avliyakulov N, Benada O (1998) Pankinetoplast DNA structure in a primitive bodonid flagellate, Cryptobia helicis. EMBO J 17:838–846
Lukeš J, Hines JC, Evans CJ, Avliyakulov NK, Prabhu VP, Chen J, Ray DS (2001). Disruption of the Crithidia fasciculata KAP1 gene results in extensive structural rearrangement of the kinetoplast disc. Mol Biochem Parasitol 117:179–186
Lukeš J, Guilbride DL, Votýpka J, Zíková A, Benne R, Englund PT (2002). The kinetoplast DNA network: evolution of an improbable structure. Eukaryot Cell 1:495–502
Madison-Antenucci S, Hajduk SL (2001) RNA editing-associated protein 1 is an RNA binding protein with specificity for preedited mRNA. Mol Cell 7:879–886
Madison-Antenucci S, Sabatini RS, Pollard VW, Hajduk SL (1998) Kinetoplastid RNA-editing-associated protein 1 (REAP-1): a novel editing complex protein with repetitive domains. EMBO J 17:6368–6376
Madison-Antenucci S, Grams J, Hajduk SL (2002) Editing machines: the complexities of trypanosome RNA editing. Cell 108:435–438
Marande W, Lukeš J, Burger G (2005) Unique mitochondrial genome structure in diplonemids, the sister group of kinetoplastids. Eukaryot Cell 4:1137–1146
Margossian SP, Li H, Zassenhous HP, Butow RA (1996) The DexH box protein SUV3p is a component of a yeast mitochondrial 3′–5′ exoribonuclease that suppresses group I intron toxicity. Cell 84:199–209
Marini JC, Levene SD, Crothers DM, Englund PT (1982) Bent helical structure in kinetoplast DNA. Proc Natl Acad Sci USA 79:7664–7668
Maslov DA, Kolesnikov AA, Zaitseva GN (1984) Conservative and divergent base sequence regions in the maxicircle kinetoplast DNA of several trypanosomatid flagellates. Mol Biochem Parasitol 12:351–364
Maslov DA, Simpson L (1992) The polarity of editing within a multiple gRNA-mediated domain is due to formation of anchors for upstream gRNAs by downstream editing. Cell 70:459–467
Maslov DA, Simpson L (1994) RNA editing and mitochondrial genomic organization in the cryptobiid kinetoplastid protozoan Trypanoplasma borreli. Mol Cell Biol 14:8174–8182
Maslov DA, Zíková A, Kyselová I, Lukeš J (2002) A putative novel nuclear-encoded subunit of the cytochrome c oxidase complex in trypanosomatids. Mol Biochem Parasitol 125:113–25
McManus MT, Adler BK, Pollard VW , Hajduk SL (2000) Trypanosoma brucei guide RNA poly(U) tail formation is stabilized by cognate mRNA. Mol Cell Biol 20:883–891
Militello KT, Read LK (2000) UTP-dependent and independent pathway of mRNA turnover in Trypanosoma brucei mitochondria. Mol Cell Biol 20:2308–2316
Militello KT, Read LK (1999) Coordination of kRNA editing and polyadenylation in Trypanosoma brucei mitochondria: complete editing is not required for long poly(A) tract addition. Nucleic Acids Res 27:1377–1385
Miller MM, Read LK (2003) Trypanosoma brucei: functions of RBP16 cold shock and RGG domains in macromolecular interactions. Exp Parasitol 105:140–148
Missel A, Souza AE, Nörskau G, Göringer HU (1997) Disruption of a gene encoding a novel mitochondrial DEAD-box protein in Trypanosoma brucei affects edited mRNAs. Mol Cell Biol 17:4895–4903
Moreira D, López-Garcia P, Vickerman K (2004) An updated view of kinetoplastid phylogeny using environmental sequences and a closer outgroup: proposal for a new classification of the class Kinetoplastea. Int J Syst Evol Microbiol 54:1861–1875
Morris JC, Drew ME, Klingbeil MM, Motyka SA, Saxowsky TT, Wang ZF, Englund PT (2001) Replication of kinetoplast DNA: an update for the new millenium. Int J Parasitol 31:453–458
Müller UF, Lambert L, Göringer HU (2001) Annealing of RNA editing substrates facilitated by guide RNA-binding protein gBP21. EMBO J 20:1394–1404
Müller UF, Göringer HU (2002) Mechanism of the gBP21-mediated RNA/RNA annealing reaction: matchmaking and charge reduction. Nucleic Acids Res 30:447–455
Myler PJ, Glick D, Feagin JE, Morales TH, Stuart KD (1993) Structural organization of the maxicircle variable region of Trypanosoma brucei—identification of potential replication origins and topoisomerase II binding sites. Nucleic Acids Res 21:687–694
Nitz N, Gomes C, de Cassia Rosa A, D’Souza-Ault MR, Moreno F, Lauria-Pires L, Nascimento RJ, Teixeira ARL (2004) Heritable integration of kDNA minicircle sequences from Trypanosoma cruzi into the avian genome: insights into human Chagas disease. Cell 118:175–186
O’Hearn SF, Huang CE, Hemann M, Zhelonkina A, Sollner-Webb B (2003) Trypanosoma brucei RNA editing complex: band II is structurally critical and maintains band V ligase, which is nonessential. Mol Cell Biol 23:7909–7919
Ogbadoyi EO, Robinson DR, Gull K (2003) A high-order trans-membrane structural linkage is responsible for mitochondrial genome positioning and segregation by flagellar basal bodies in trypanosomes. Mol Biol Cell 14:1769–1779
Onn I, Milman-Shtepel N, Shlomai J (2004) Redox potential regulates binding of universal minicircle sequence binding protein at the kinetoplast DNA replication origin. Eukaryot Cell 3:277–287
Ouaissi A, Vergnes B, Borges M, Guilvard E (2000) Identification and molecular characterization of two novel Trypanosoma cruzi genes encoding polypeptides sharing sequence motifs found in proteins involved in RNA editing reactions. Gene 253:271–280
Palazzo SS, Panigrahi AK, Igo RP, Salavati R, Stuart K (2003) Kinetoplastid RNA editing ligases: complex association, characterization, and substrate requirements. Mol Biochem Parasitol 127:161–167
Palmer JD (1997) Going, going, gone. Science 275:790–791
Panigrahi AK, Schnaufer A, Carmean N, Igo RP, Gygi SP, Ernst NL, Palazzo SS, Weston DS, Abersold R, Salavati R, Stuart KD (2001a) Four related proteins of the Trypanosoma brucei RNA editing complex. Mol Cell Biol 21:6833–6840
Panigrahi AK, Gygi SP, Ernst NL, Igo RP, Palazzo SS, Schnaufer A, Weston DS, Carmean N, Salavati R, Aebersold R, Stuart KD (2001b) Association of two novel proteins, TbMP52 and TbMP48, with the Trypanosoma brucei RNA editing complex. Mol Cell Biol 21:380–389
Panigrahi AK, Schnaufer A, Ernst NL, Wang B, Carmean N, Salavati R, Stuart K (2003a) Identification of novel components of Trypanosoma brucei editosomes. RNA 9:484–492
Panigrahi AK, Allen TE, Stuart K, Haynes PA, Gygi SP (2003b) Mass spectrometric analysis of the editosome and other multiprotein complexes in Trypanosoma brucei. J Am Soc Mass Spec 14:728–735
Pelletier M, Read LK (2003) RBP16 is a multifunctional gene regulatory protein involved in editing and stabilization of specific mitochondrial mRNAs in Trypanosoma brucei. RNA 9:457–468
Penschow JL, Sleve DA, Ryan CM, Read LK (2004) TbDSS-1, an essential Trypanosoma brucei exoribonuclease homolog that has pleiotropic effects on mitochondrial RNA metabolism. Eukaryot Cell 3:1206–1216
Peris M, Frech GC, Simpson AM, Bringaud F, Byrne E, Bakker A, Simpson L (1994) Characterization of two classes of ribonucleoprotein complexes possibly involved in RNA editing from Leishmania tarentolae mitochondria. EMBO J 13:1664–1672
Piller KJ, Rusché LN, Cruz-Reyes J, Sollner-Webb B (1997) Resolution of the RNA editing gRNA-directed endonuclease from two other endonucleases of Trypanosoma brucei mitochondria. RNA 3:279–290
Pollard VW, Rohrer SP, Michelotti EF, Hancock K, Hajduk SL (1990) Organization of minicircle genes for guide RNAs in Trypanosoma brucei. Cell 63: 783–790
Pollard VW, Harris ME, Hajduk SL (1992) Native mRNA editing complexes from Trypanosoma brucei mitochondria. EMBO J 11:4429–4438
Rauch CA, Pérez-Morga D, Cozzarelli NR, Englund PT (1993) The absence of supercoiling in kinetoplast DNA minicircles. EMBO J 12:403–411
Ray DS (1989) Conserved sequence blocks in kinetoplast minicircles from diverse species of trypanosomes. Mol Cell Biol 9:1365–1367
Ray DS, Hines JC (1995) Disruption of the Crithidia fasciculata RNH1 gene results in the loss of two active forms of ribonuclease H. Nucleic Acids Res 23:2526–2530
Read LK, Myler PJ, Stuart K (1992) Extensive editing of both processed and preprocessed maxicircle CR6 transcripts in Trypanosoma brucei. J Biol Chem 267:1123–1128
Read LK, Stankey KA, Fish WR, Muthiani AM, Stuart K (1994) Developmental regulation of RNA editing and polyadenylation in four life cycle stages of Trypanosoma congolense. Mol Biochem Parasitol 68:197–206
Riley GR, Corepp RA, Stuart K (1994) Multiple guide RNAs for identical editing of Trypanosoma brucei apocytochrome b messenger RNA have an unusual minicircle location and are developmentally regulated. J Biol Chem 269:6101–6108
Riley GR, Myler PJ, Stuart K (1995) Quantitation of RNA editing substrates, products and potential intermediates—implications for developmental regulation. Nucleic Acids Res 23:708–712
Robinson KR, Beverley SM (2003) Improvements in transfection efficiency and tests of RNA interference (RNAi) approaches in the protozoan parasite Leishmania. Mol Biochem Parasitol 128:217–228
Rusché LN, Piller KJ, Sollner-Webb B (1995) Guide RNA-messenger-RNA chimeras, which are potential RNA editing intermediates, are formed by endonuclease and RNA ligase in trypanosome mitochondrial extract. Mol Cell Biol 15:2933–2941
Rusché LN, Cruz-Reyes J, Piller KJ, Sollner-Webb B (1997) Purification of a functional enzymatic editing complex from Trypanosoma brucei mitochondria. EMBO J 16:4069–4081
Rusché LN, Huang CE, Piller KJ, Hemann M, Wirtz E, Sollner-Webb B (2001) The two RNA ligases of the Trypanosoma brucei RNA editing complex: cloning the essential band IV gene and identifying the band V gene. Mol Cell Biol 21:979–989
Ryan CM, Read LK (2005) UTP-dependent turnover of Trypanosoma brucei mitochondrial mRNA requires UTP polymerization and involves the RET1 TUTase. RNA 11:1–11
Ryan CM, Militello KT, Read LK (2003) Polyadenylation regulates the stability of Trypanosoma brucei mitochondrial RNAs. J Biol Chem 278:32753–32762
Sabatini R, Hajduk SL (1995) RNA ligase and its involvement in guide RNA/ mRNA chimera formation. Evidence for a cleavage-ligation mechanism of Trypanosoma brucei mRNA editing. J Biol Chem 270:7233–7240
Salavati R, Panigrahi AK, Stuart KD (2001) Mitochondrial ribonuclease P activity of Trypanosoma brucei. Mol Biochem Parasitol 115:109–117
Salavati R, Panigrahi AK, Morach BA, Palazzo SS, Igo RP, Stuart K (2002) Endoribonuclease activities of Trypanosoma brucei mitochondria. Mol Biochem Parasitol 120:23–31
Saxowsky TT, Choudhary G, Klingbeil MM, Englund PT (2003) Trypanosoma brucei has two distinct mitochondrial DNA polymerase beta enzymes. J Biol Chem 278:49095–49101
Sbicego S, Alfonzo JD, Estévez AM, Rubio MA, Kang X, Turck CW, Peris M, Simpson L (2003) RBP38, a novel RNA-binding protein from trypanosomatid mitochondria, modulates RNA stability. Eukaryot Cell 2:560–568
Scheffler IE (1999) Mitochondria. Wiley-Liss, New York
Schmid B, Riley GR, Stuart K, Goringer HU (1995) The secondary structure of guide RNA molecules from Trypanosoma brucei. Nucleic Acids Res 23:3093–3102
Schnaufer A, Panigrahi AK, Panicucci B, Igo RP Jr, Wirtz E, Salavati R, Stuart K (2001) An RNA ligase essential for RNA editing and survival of the bloodstream form of Trypanosoma brucei. Science 291:2159–2162
Schnaufer A, Sbicego S, Blum B (2000) Antimycin A resistance in a mutant Leishmania tarentolae strain is correlated to a point mutation in the mitochondrial apocytochrome b gene. Curr Genet 37:234–241
Schnaufer A, Domingo GJ, Stuart K (2002) Natural and induced dyskinetoplastic trypanosomatids: how to live without mitochondrial DNA. Int J Parasitol 32:1071–1084
Schnaufer A, Ernst NL, Palazzo SS, O’Rear J, Salavati R, Stuart K (2003) Separate insertion and deletion subcomplexes of the Trypanosoma brucei RNA editing complex. Mol Cell 12:307–319
Schneider A, Marechal-Drouard L (2000) Mitochondrial tRNA import: are there distinct mechanisms? Trends Cell Biol 10:509–513
Seiwert SD, Stuart K (1994) RNA editing: transfer of genetic information from gRNA to precursor mRNA in vitro. Science 266:114–117
Seiwert SD, Heidmann S, Stuart K (1996) Direct visualization of uridylate deletion in vitro suggests a mechanism for kinetoplastid RNA editing. Cell 84:831–841
Shapiro TA (1993) Kinetoplast DNA maxicircles-networks within networks. Proc Natl Acad Sci USA 90:7809–7813
Shapiro TA, Englund PT (1995) The structure and replication of kinetoplast DNA. Annu Rev Microbiol 49:117–143
Shlomai J (2004) The structure and replication of kinetoplast DNA. Curr Mol Med 4:623–647
Simpson AGB, Lukeš J, Roger AJ (2002) The evolutionary history of kinetoplastids and their kinetoplast. Mol Biol Evol 19:2071–2083
Simpson AGB, Roger AJ (2004) Protein phylogenies robustly resolve the deep-level relationships within Euglenozoa. Mol Phylogenet Evol 30:201–212
Simpson L (1973) Structure and function of the kinetoplast DNA. J Protozool 20:2–8
Simpson L, Maslov DA (1994) RNA editing and the evolution of parasites. Science 264:1870–1871
Simpson L, Thiemann OH, Savill NJ, Alfonso JD, Maslov DA (2000) Evolution of RNA editing in trypanosome mitochondria. Proc Natl Acad Sci USA 97:6986–6993
Simpson L, Sbicego S, Aphasizhev R (2003) Uridine insertion/deletion RNA editing in trypanosome mitochondria: a complex business. RNA 9:265–276
Simpson L, Aphasizhev R, Gao G, Kang X (2004) Mitochondrial proteins and complexes in Leishmania and Trypanosoma involved in U-insertion/deletion RNA editing. RNA 10:159–170
Simpson L, Neckelmann N, de la Cruz VF, Simpson AM, Feagin JE, Jasmer DP, Stuart JE (1987) Comparison of the maxicircle (mitochondrial) genomes of Leishmania tarentolae and Trypanosoma brucei at the level of nucleotide sequence. Biol Chem. 262:6182–96
Sinha KM, Hines JC, Downey N, Ray DS (2004) Mitochondrial DNA ligase in Crithidia fasciculata. Proc Natl Acad Sci USA 101:4361–4366
Sloof P, De Haan A, Eier W, Van Iersel M, Boel E, Van Steeg H, Benne R (1992) The nucleotide sequence of the variable region in Trypanosoma brucei completes the sequence analysis of the maxicircle component of mitochondrial kinetoplast DNA. Mol Biochem Parasitol 56:289–299
Sloof P, Van den Burg J, Voogd A, Benne R, Agostinelli M, Borst P, Gutell R, Noller H (1985) Further characterization of the extremely small mitochondrial ribosomal RNAs from trypanosomes: a detailed comparison of the 9S and 12S RNAs from Crithidia fasciculata and Trypanosoma brucei with rRNAs from other organisms. Nucleic Acids Res 13:4171–4190
Souza AE, Myler PJ, Stuart K (1992) Maxicircle CR1 transcripts of Trypanosoma brucei are edited and developmentally regulated and encode a putative iron-sulfur protein homologous to an NADH dehydrogenase subunit. Mol Cell Biol 12:2100–2107
Souza AE, Shu HH, Read LK, Myler PJ, Stuart KD (1993) Extensive editing of CR2 maxicircle transcripts of Trypanosoma brucei predicts a protein with homology to a subunit of NADH dehydrogenase. Mol Cell Biol 13:6832–6840
Speijer D, Breek CK, Muijsers AO, Hartog AF, Berden JA, Albracht SP, Samyn B, Van Beeumen J, Benne R (1997) Characterization of the respiratory chain from cultured Crithidia fasciculata. Mol Biochem Parasitol 85:171–186
Stuart KD, Panigrahi AK (2002) RNA editing: complexity and complications. Mol Microbiol 45:591–596
Stuart KD, Feagin JE, Abraham JM (1989) The creation of nucleotide sequences in messenger RNA—a minireview. Gene 15:155–160
Stuart KD, Allen TE, Heidmann S, Seiwert SD (1997) RNA editing in kinetoplastid protozoa. Microbiol Mol Biol Rev 61:105–120
Stuart KD, Panigrahi AK, Schnaufer A, Drozdz M, Clayton C, Salavati R (2002) Composition of the editing complex of Trypanosoma brucei. Phil Trans R Soc Lond B Biol Sci 357:71–79
Stuart K, Salavati R, Igo RP, Lewis Ernst N, Palazzo SS, Wang B (2004) In vitro assays for kinetoplastid U-insertion-deletion editing and associated activities. Methods Mol Biol 265:251–272
Stuart KD, Schnaufer A, Ernst NL, Panigrahi AK (2005) Complex management: RNA editing in trypanosomes. Trens Biochem Sci 30:97–105
Sturm NR, Simpson L (1990a) Partially edited mRNAs for cytochrome b and subunit III of cytochrome oxidase from Leishmania tarentolae mitochondria: RNA editing intermediates. Cell 61:871–878
Sturm NR, Simpson L (1990b) Kinetoplast DNA minicircles encode guide RNAs for editing of cytochrome oxidase subunit III mRNA. Cell 61:879–884
Sturm NR, Maslov DA, Blum B, Simpson L (1992) Generation of unexpected editing patterns in Leishmania tarentolae mitochondrial mRNAs: misediting produced by misguiding. Cell 70:469–476
Tan TH, Pach R, Crausaz A, Ivens A, Schneider A (2002) tRNAs in Trypanosoma brucei: genomic organization, expression, and mitochondrial import. Mol Cell Biol 22:3707–3717
Temperley RJ, Seneca SH, Tonska K, Bartnik E, Bindoff LA, Lightowlers RN, Chrzanowska-Lightowlers ZM (2003) Investigation of a pathogenic mtDNA microdeletion reveals a translation-dependent deadenylation decay pathway in human mitochondria. Hum Mol Genet 12:2341–2348
Tittawella I (1998) Protein synthesis in mitochondria isolated from the trypanosomatid protozoan Crithidia fasciculata. FEBS Lett 423:351–356
Torri AF, Kunkel TA, Englund PT (1994) A beta-like DNA polymerase from the mitochondrion of the trypanosomatid Crithidia fasciculata. J Biol Chem 269:8165–8171
Ullu E, Tschudi C, Chakraborty T (2004) RNA interference in protozoan parasites. Cell Microbiol 6:509–519
Van der Spek H, Arts GJ, Zwaal RR, van den Burg J, Sloof P, Benne R (1991) Conserved genes encode guide RNAs in mitochondria of Crithidia fasciculata. EMBO J 10:1217–1224
Van Weelden SWH, Fast B, Vogt A, Van Der Meer P, Saas J, Van Hellemond JJ, Tielens AGM, Boshart M. (2003) Procyclic Trypanosoma brucei do not use Krebs cycle activity for energy generation. J Biol Chem 278:12854–12863
Vanhamme L, Pérez-Morga D, Marechal C, Speijer D, Lambert L, Geuskens M, Alexandre S, Ismaili N, Göringer U, Benne R, Pays E (1998) Trypanosoma brucei TbRGG1, a mitochondrial oligo(U)-binding protein that co-localizes with an in vitro RNA editing activity. J Biol Chem 273:21825–21833
Vickerman K (1990) The evolutionary expansion of the trypanosomatid flagellates. Int J Parasitol 20:1317–1331
Vickerman K (2000) Diplonemids. In: The illustrated guide to the protozoa, pp. 1157–1159
Vondrušková E, van den Burg J, Zíková A, Ernst NL, Stuart K, Benne R, Lukeš J (2005) RNA interference analyses suggest a transcript-specific regulatory role for MRP1 and MRP2 in RNA editing and other RNA processing in Trypanosoma brucei. J Biol Chem 280:2429–2438
Wang Z, Englund PT (2001) RNA interference of a trypanosome topoisomerase II causes progressive loss of mitochondrial DNA. EMBO J 20:4674–4683
Wang BB, Ernst NL, Palazzo SS, Panigrahi AK, Salavati R, Stuart K (2003) TbMP44 is essential for RNA editing and structural integrity of the editosome in Trypanosoma brucei. Eukaryot Cell 2:578–587
Worthey EA, Schnaufer A, Mian IS, Stuart K, Salavati R (2003) Comparative analysis of editosome proteins in trypanosomatids. Nucleic Acids Res 31:6392–6408
Xu CW, Hines JC, Engel ML, Russell DG, Ray DS (1996) Nucleus-encoded histone H1-like proteins are associated with kinetoplast DNA in the trypanosomatid Crithidia fasciculata. Mol Cell Biol 16:564–576
Acknowledgements
We thank Rob Benne (University of Amsterdam) and Dmitri A. Maslov (University of California) for critical reading of the manuscript and Jana Fišáková for artistic talent. This work was supported by grants from the Grant Agency of the Czech Academy of Sciences 5022302 and Z60220518, the National Institutes of Health 5R03TW6445-2 and the Ministry of Education of the Czech Republic 6007665801.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by R. Bock
Rights and permissions
About this article
Cite this article
Lukeš, J., Hashimi, H. & Zíková, A. Unexplained complexity of the mitochondrial genome and transcriptome in kinetoplastid flagellates. Curr Genet 48, 277–299 (2005). https://doi.org/10.1007/s00294-005-0027-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-005-0027-0