Summary
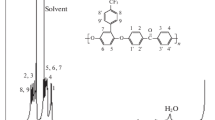
A new poly(phenylene ether) derivative, poly(2,6-dihydroxy-1,4-oxyphenylene), was synthesized by demethylation of poly(2,6-dimethoxy-1,4-oxyphenylene) with an excess of boron tribromide in methylene chloride. The resulting polymer was soluble in N,N-dimethylformaide, dimethyl sulfoxide, and actone, however, insoluble in chloroform, methanol, and benzene. NMR and IR analyses showed that the extent of the demethylation was 93 % and the polymer was composed of 2,6-dihydroxy-1,4-oxyphenylene unit. The polymer was stable below 300 °C under nitrogen.
Similar content being viewed by others
References
Aycock D, Abolins V,. White DM (1986) Encyclopedia of Polymer Science and Engineering, 2nd ed., John Wiley & Sons: New York, Vol. 13, pp. 1–30
Hay AS, Blanchard HS, Endres GF, Eustance JW (1959) J. Am. Chem. Soc. 81: 6335
Hay AS (1962) J. Polym. Sci. 58: 581
Hay AS (1977) Adv. Polym. Sci. 4: 496
Kobayashi S, Shoda S, Uyama H (1995) Adv. Polym. Sci. 121: 1
Ikeda R, Uyama H, Kobayashi S (1996) Macromolecules 29: 3053
McOmie JFW, Watts ML, West DE (1968) Tetrahedron 24: 2289
Karas M, Bachmann D, Bahr U, Hillenkamp F (1987) Int. J. Mass Spectrom. Ion Proc. 78: 53
Räder HJ, Spickermann J, Müllen K (1995) Macromol. Chem. Phys. 196: 3967
Ikeda R, Sugihara J, Uyama H, Kobayashi S, Macromolecules in press
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ikeda, R., Uyama, H. & Kobayashi, S. Poly(2,6-dihydroxy-1,4-oxyphenylene. Polymer Bulletin 38, 273–277 (1997). https://doi.org/10.1007/s002890050048
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002890050048