Abstract
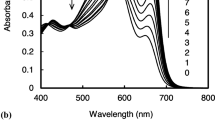
We have designed a colorimetric conjugated polymer (PDMAp) for selective ATP sensing based on poly(2,5-dimethoxyaniline) bearing phthalimide which can form hydrogen bonds with adenine. PDMAp have been synthesized via chemical oxidative polymerization with CSA and DDQ and dedoping with an aqueous ammonia solution. PDMAp was displayed to signal the colorimetric selective detection for ATP over various nucleotides such as ADP, AMP, CTP, TTP, and GTP in DMSO–water (9:1, v/v) mixture. The absorption spectral change can be attributed to a doping process of ATP to PDMAp through hydrogen bonds between phthalimide and adenine as well as electrostatic interactions between the phosphate group of ATP and the backbone of PDMAp.





Similar content being viewed by others
References
Jose DA, Mishra S, Ghosh A, Shrivastav A, Mishra SK, Das A (2007) Colorimetric sensor for ATP in aqueous solution. Org Lett 9:1979–1982
Yao Z, Li C, Shi G (2008) Optically active supramolecular complexes of water-soluble achiral polythiophenes and folic acid spectroscopic studies and sensing applications. Langmuir 24:12829–12835
Secor KE, Glass TE (2004) Selective amine recognition: development of a chemosensor for dopamine and norepinephrine. Org Lett 6:3727–3730
Liu Y, Dong H, Zhang W, Ye Z, Wang G, Yuan J (2010) Preparation of a novel colorimetric luminescence sensor strip for the detection of indole-3-acetic acid. Biosens Bioelectron 25:2375–2378
Baumes LA, Sogo MB, Montes-Navajas P, Corma A, Garcia H (2010) A colorimetric sensor array for the detection of the date-rape drug γ-hydroxybutyric acid (GHB): a supramolecular approach. Chem Eur J 16:4489–4495
Kornberg A (1988) DNA replication. J Biol Chem 263:1–4
Shen X, Mizuguchi G, Hamiche A, Wu C (2000) A chromatin remodelling complex involved in transcription and DNA processing. Nature 406:541–544
Stekhoven FS (1972) Energy transfer factor A.D (ATP synthetase) as a complex Pi–ATP exchange enzyme and its stimulation by phospholipids. Biochem Biophys Res Commum 47:7–14
Higgins CF, Hiles ID, Salmond GPC (1986) A family of related ATP-binding subunits coupled to many distinct biological processes in bacteria. Nature 323:448–450
Bush KT, Keller ST, Nigam SK (2000) Genesis and reversal of the ischemic phenotype in epithelial cells. J Clin Invest 106:621–626
Dimonte DA (1991) Mitochondrial DNA and Parkinson’s disease. Neurology 41:38–42
Harkness RA, Saugstad OD (1997) The importance of the measurement of ATP depletion and subsequent cell damage with an estimate of size and nature of the market for a practicable method: a review designed for technology transfer. Scand J Clin Lab Invest 57:655–672
Przedborski S, Vila S (2001) MPTP: a review of its mechanisms of neurotoxicity. Clin Neurosci Res 1:407–418
Burnstock G (2006) Pathophysiology and therapeutic potential of purinergic signaling. Pharm Rev 58:58–86
Singh N, Jang DO (2011) A selective ATP chromogenic sensor for use in an indicator displacement assay. Tetrahedron Lett 52:5094–5097
Marbumrung S, Wongravee K, Ruangpornvisuti V, Tumcharern G, Tuntulani T, Tomapatanaget B (2012) Discrimination of nucleotides by single fluorescence sensor under solvent-dependent recognition patterns. Sens Actuators B 171–172:969–975
Ghosh A, Shrivastav A, Jose DA, Mishra SK, Chandrakanth CK, Mishra S, Das A (2008) Colorimetric sensor for triphosphates and their application as a viable staining agent for prokaryotes and eukaryotes. Anal Chem 80:5312–5319
McQuade DT, Pullen AE, Swager TM (2000) Conjugated polymer-based chemical sensors. Chem Rev 100:2537–2574
Basabe-Desmonts L, Reinhoudt DN, Crego-Calama M (2007) Design of fluorescent materials for chemical sensing. Chem Soc Rev 36:993–1017
Song Y, Wei W, Qu X (2011) Colorimetric biosensing using smart materials. Adv Mater 23:4215–4236
Pringsheim E, Terpetschnig E, Piletsky SA, Wolfbeis OS (1999) A polyaniline with near-infrared optical response to saccharides. Adv Mater 11:865–868
Huh P, Kim SC, Kim Y, Wang Y, Singh J, Kumar J, Samuelson LA, Kim BS, Jo NJ, Lee JO (2007) Optical and electrochemical detection of saccharides with poly(aniline-co-3-aminobenzeneboronic acid) prepared from enzymatic polymerization. Biomacromolecules 8:3602–3607
Antony MJ, Jayakannan M (2011) Polyaniline nanoscaffolds for colorimetric sensing of biomolecules via electron transfer process. Langmuir 27:6268–6278
Bossi A, Piletsky SA, Piletska EV, Righetti PG, Turner APF (2000) An assay for ascorbic acid based on polyaniline-coated microplates. Anal Chem 72:4296–4300
Ray A, Richter AF, MacDiarmid AG, Epstein AJ (1989) Polyaniline: protonation/deprotonation of amine and imine sites. Synth Met 29:E151–E156
MacDiarmid AG, Epstein AJ (1989) Polyanilines: a novel class of conducting polymers. Faraday Discuss Chem Soc 88:317–332
Huang J, Virji S, Weiller BH, Kaner RB (2003) Polyaniline nanofiber: facile synthesis and chemical sensors. J Am Chem Soc 125:314–315
Ciric-Marjanovic G (2013) Recent advances in polyaniline research: polymerization mechanism, structural aspects, properties and applications. Synth Met 177:1–47
Antony MJ, Jayakannan M (2011) Polyaniline nanoscaffolds for colorimetric sensing of biomolecules via electron transfer process. Langmuir 27:6268–6278
Ding B, Si Y, Wang X, Yu J, Feng L, Sun G (2011) Label-free ultrasensitive colorimetric detection of copper(II) ions utilizing polyaniline/polyamide-6 nano-fiber/net sensor strips. J Mater Chem 21:13345–13353
D’Aprano G, Leclerc M (1995) Synthesis and characterization of polyaniline derivatives: poly(2-alkoxyanilines) and poly (2,5-dialkoxyanilines). Chem Mater 7:33–42
Huang LM, Wen TC, Gopalan A (2002) In situ UV-visible spectroelectrochemical studies on electrochromic behavior of poly(2,5-dimethoxy aniline). Synth Met 130:155–163
Sajadi M, Obernhuber T, Kovalenko SA, Mosquera M, Dick B, Ernsting NP (2009) Dynamic polar solvation is reported by fluorescing 4-aminophthalimide faithfully despite H-bonding. J Phys Chem A 113:44–55
Weinberger M, Berndt F, Mahrwald R, Ernsting NP, Wagenknecht HA (2013) Synthesis of 4-aminophthalimide and 2,4-diaminopyrimidine C-nucleosides as isosteric fluorescent DNA base substitutes. J Org Chem 78:2589–2599
Watson JD, Crick FHC (1953) A structure for deoxyribonucleic acid. Nature 171:737–738
Hino T, Kumakura T, Kuramoto N (2006) Optically active fluoro-substituted polyaniline prepared in organic media: the synthesis, chiroptical properties, and comparison with optically active non-substituted polyaniline. Polymer 47:5295–5302
Norris ID, Kane-Maguire LAP, Wallace GG (2000) Electrochemical synthesis and chiroptical properties of optically active poly(o-methoxyaniline). Macromolecules 33:3237–3243
Ray A, Asturias GE, Kershner DL, Richter AF, MacDiarmid AG, Epstein AJ (1989) Polyaniline: doping, structure, and derivatives. Synth Met 29:E141–E150
Yuan GL, Kuramoto N (2002) Water-processable chiral polyaniline derivatives doped and intertwined with dextran sulfate: synthesis and chiroptical properties. Macromolecules 35:9773–9779
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kato, A., Fukushima, Y. Colorimetric chemosensor for ATP based on phthalimide-appended poly(2,5-dimethoxyaniline). Polym. Bull. 70, 3519–3527 (2013). https://doi.org/10.1007/s00289-013-1037-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-013-1037-6