Abstract
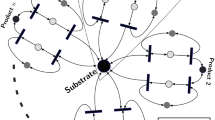
Positive feedback loops are common regulatory elements in metabolic and protein signalling pathways. The length of such feedback loops determines stability and sensitivity to network perturbations. Here we provide a mathematical analysis of arbitrary length positive feedback loops with protein production and degradation. These loops serve as an abstraction of typical regulation patterns in protein signalling pathways. We first perform a steady state analysis and, independently of the chain length, identify exactly two steady states that represent either biological activity or inactivity. We thereby provide two formulas for the steady state protein concentrations as a function of feedback length, strength of feedback, as well as protein production and degradation rates. Using a control theory approach, analysing the frequency response of the linearisation of the system and exploiting the Small Gain Theorem, we provide conditions for local stability for both steady states. Our results demonstrate that, under some parameter relationships, once a biological meaningful on steady state arises, it is stable, while the off steady state, where all proteins are inactive, becomes unstable. We apply our results to a three-tier feedback of caspase activation in apoptosis and demonstrate how an intermediary protein in such a loop may be used as a signal amplifier within the cascade. Our results provide a rigorous mathematical analysis of positive feedback chains of arbitrary length, thereby relating pathway structure and stability.



Similar content being viewed by others
References
Albeck JG, Burke JM, Spencer SL, Lauffenburger DA, Sorger PK (2008) Modeling a snap-action, variable-delay switch controlling extrinsic cell death. PLoS Biol 6(12):e299. doi:10.1371/journal.pbio.0060299
Alon U (2007) An introduction to systems biology: design principles of biological circuits, vol 10. CRC press, Boca Raton
Arcak M, Sontag ED (2006) Diagonal stability of a class of cyclic systems and its connection with the secant criterion. Automatica 42:1531–1537
Bentele M, Lavrik I, Ulrich M, Stosser S, Heermann DW, Kalthoff H, Krammer PH, Eils R (2004) Mathematical modeling reveals threshold mechanism in CD95-induced apoptosis. J Cell Biol 166(6):839–851. doi:10.1083/jcb.200404158
Bernstein DS (2009) Matrix mathematics: theory, facts, and formulas, 2nd edn. Princeton University Press, Princeton
Brandman O, Meyer T (2008) Feedback loops shape cellular signals in space and time. Science 322(5900):390–395. doi:10.1126/science.1160617
Cowling V, Downward J (2002) Caspase-6 is the direct activator of caspase-8 in the cytochrome c-induced apoptosis pathway: absolute requirement for removal of caspase-6 prodomain. Cell Death Diff 9(10):1046–1056. doi:10.1038/sj.cdd.4401065
Craciun G, Helton JW, Williams RJ (2008) Homotopy methods for counting reaction network equilibria. Math Biosci 216(2):140–149. doi:10.1016/j.mbs.2008.09.001
Du C, Fang M, Li Y, Li L, Wang X (2000) Smac, a mitochondrial protein that promotes cytochrome c-dependent caspase activation by eliminating IAP inhibition. Cell 102(1):33–42
Eissing T, Conzelmann H, Gilles ED, Allgower F, Bullinger E, Scheurich P (2004) Bistability analyses of a caspase activation model for receptor-induced apoptosis. J Biol Chem 279(35):36,892–36,897. doi:10.1074/jbc.M404893200
Feinberg M (1995) The existence and uniqueness of steady states for a class of chemical reaction networks. Arch Rational Mech Anal 132:311–370. doi:10.1007/BF00375614
Ferrell JE Jr (2002) Self-perpetuating states in signal transduction: positive feedback, double-negative feedback and bistability. Curr Opin Cell Biol 14(2):140–148
Graham RK, Ehrnhoefer DE, Hayden MR (2011) Caspase-6 and neurodegeneration. Trends Neurosci 34(12):646–656. doi:10.1016/j.tins.2011.09.001
Green DR, Kroemer G (2004) The pathophysiology of mitochondrial cell death. Science 305(5684):626–629. doi:10.1126/science.1099320
Hori Y, Kim TH, Hara S (2011) Existence criteria of periodic oscillations in cyclic gene regulatory networks. Automatica 47(6):1203–1209. doi:10.1016/j.automatica.2011.02.042 (Special Issue on Systems Biology)
Huber HJ, Duessmann H, Wenus J, Kilbride SM (1813) Prehn JH (2011) Mathematical modelling of the mitochondrial apoptosis pathway. Biochim Biophys Acta Mol Cell Res 4:608–615. doi:10.1016/j.bbamcr.2010.10.004
Huber HJ, Laussmann MA, Prehn JH, Rehm M (2010) Diffusion is capable of translating anisotropic apoptosis initiation into a homogeneous execution of cell death. BMC Syst Biol 4(9): doi:10.1186/1752-0509-4-9
Inoue S, Browne G, Melino G, Cohen GM (2009) Ordering of caspases in cells undergoing apoptosis by the intrinsic pathway. Cell Death Diff 16(7):1053–1061. doi:10.1038/cdd.2009.29
Jovanovic M, Arcak M, Sontag E (2008) A passivity-based approach to stability of spatially distributed systems with a cyclic interconnection structure. IEEE Trans Autom Control 53(Special Issue):75–86. doi:10.1109/TAC.2007.911318
Kappelhoff J, Liu S, Dugdale M, Dymianiw D, Linton L, Huber R (2009) Practical considerations when using temperature to obtain rate constants and activation thermodynamics of enzymes with two catalytic steps: Native and N460T-\(\beta \)-Galactosidase E. coli as examples. Protein J 28(2):96–103. doi: 10.1007/s10930-009-9168-1
Khalil HK (2001) Nonlinear systems, 3rd edn. Prentice Hall, Upper Saddle River
Kuranaga E (2011) Caspase signaling in animal development. Dev Growth Diff 53(2):137–148. doi:10.1111/j.1440-169X.2010.01237.x
Legewie S, Blüthgen N, Herzel H (2006) Mathematical modeling identifies inhibitors of apoptosis as mediators of positive feedback and bistability. PLoS Comput Biol 2(9):e120+. doi:10.1371/journal.pcbi.0020120
López-Caamal F, García MR, Middleton RH, Huber HJ (2012) Positive feedback in the Akt/mTOR pathway and its implications for growth signal progression in skeletal muscle cells: an analytical study. J Theor Biol 301:15–27. doi:10.1016/j.jtbi.2012.01.026
Martnez-Antonio A, Janga SC, Thieffry D (2008) Functional organisation of escherichia coli transcriptional regulatory network. J Mol Biol 381(1):238–247. doi:10.1016/j.jmb.2008.05.054
Nikolaev A, McLaughlin T, O’Leary DD, Tessier-Lavigne M (2009) APP binds DR6 to trigger axon pruning and neuron death via distinct caspases. Nature 457(7232):981–989. doi:10.1038/nature07767
O’Connor CL, Anguissola S, Huber HJ, Dussmann H, Prehn JH, Rehm M (2008) Intracellular signaling dynamics during apoptosis execution in the presence or absence of x-linked-inhibitor-of-apoptosis-protein. Biochim Biophys Acta Mol Cell Res 1783(10):1903–1913. doi:10.1016/j.bbamcr.2008.05.025
Ogata K (2001) Modern control engineering, 4th edn. Prentice Hall PTR, Upper Saddle River
Rehm M, Huber H, Dussmann H, Prehn J (2006) Systems analysis of effector caspase activation and its control by x-linked inhibitor of apoptosis protein. EMBO
Skogestad S, Postlethwaite I (2005) Multivariable feedback control: analysis and design. Wiley, New York
Slee EA, Keogh SA, Martin SJ (2000) Cleavage of BID during cytotoxic drug and UV radiation-induced apoptosis occurs downstream of the point of bcl-2 action and is catalysed by caspase-3: a potential feedback loop for amplification of apoptosis-associated mitochondrial cytochrome c release. Cell Death Diff. 7(6):556–565. doi:10.1038/sj.cdd.4400689
Strogatz SH (1994) Nonlinear dynamics and chaos: with applications to physics, biology, chemistry, and engineering (studies in nonlinearity). In: Studies in nonlinearity, 1st edn. Perseus Books Group, New York
Turing AM (1952) The chemical basis of morphogenesis. Phil Trans R Soc Lond Ser B Biol Sci 237(641): 37–72
Williams DW, Kondo S, Krzyzanowska A, Hiromi Y, Truman JW (2006) Local caspase activity directs engulfment of dendrites during pruning. Nat Neurosci 9(10):1234–1236. doi:10.1038/nn1774
Würstle ML, Laussmann MA, Rehm M (2010) The caspase-8 dimerization/dissociation balance is a highly potent regulator of caspase-8, -3, -6 signaling. J Biol Chem 285(43):33,209–33,218. doi:10.1074/jbc.M110.113860
Zou H, Yang R, Hao J, Wang J, Sun C, Fesik SW, Wu JC, Tomaselli KJ, Armstrong RC (2003) Regulation of the apaf-1/caspase-9 apoptosome by caspase-3 and XIAP. J Biol Chem 278(10):8091–8098. doi:10.1074/jbc.M204783200
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors would like to thank Ms. Niamh Connolly and the anonymous reviewers for thoroughly reviewing the manuscript and providing helpful ideas. F.L.-C. acknowledges that this work was supported by National Biophotonics and Imaging Platform, Ireland, and funded by the Irish Government’s Programme for Research in Third Level Institutions, Cycle 4, Ireland EU Structural Funds Programmes 2007-2013. H. J. H. acknowledges the support by Science Foundation Ireland via grant 08/IN.1/B1949.
Appendix
Appendix
In this section, we present three proofs required in Section 6, to prove the local stability of the off and on steady state.
1.1 A1: Proof of Lemma 1
Proof
From \(h^i(s)\) in (54), we note that the roots of its characteristic polynomial are all negative if
In the following, we show that \({\mu ^{{i}}}\) is positive for both steady states and for all \(i\).
-
1.
Off Steady State Considering the definition of the off steady state in (35a) and the definition of \({\sigma ^{{i}}}\) and \({\mu ^{{i}}}\), (72) becomes
$$\begin{aligned} {\mu ^{{i}}}_{\mathrm{off}} ={k_{3f}^{{i}}}>0. \end{aligned}$$ -
2.
On Steady State The on steady state is given by (35b), which is parametrised by \({\vartheta ^{{i+1}}}\) defined in (36). We note that (39b) allows us to express \({\mu ^{{i}}}_{\mathrm{on}}\) in (50) as
$$\begin{aligned} {\mu ^{{i}}}_{\mathrm{on}} = {k_{1}^{{i}}} {{\bar{c}}^{{i+1}}_{2}} + {k_{3f}^{{i}}}&= \frac{{k_{2}^{{i+1}}}{k_{3f}^{{i}}} {\mathrm{d}\left( {\vartheta ^{{i+1}}} \right)} -{k_{1}^{{i}}}{\mathrm{n}\left( {\vartheta ^{{i+1}}} \right)}}{{k_{2}^{{i+1}}}{\mathrm{d}\left( {\vartheta ^{{i+1}}} \right)}}. \end{aligned}$$(73)Furthermore, we claim
$$\begin{aligned} {k_{2}^{{i+1}}}{k_{3f}^{{i}}}{\mathrm{d}\left( {\vartheta ^{{i+1}}} \right)} = {\mathrm{d}\left( {\alpha ^{{i}}} \right)} {\mathrm{d}\left( {\vartheta ^{{i}}} \right)} + {k_{1}^{{i}}} {\mathrm{n}\left( {\vartheta ^{{i+1}}} \right)}. \end{aligned}$$(74)We provide a proof of this statement in Claim 1, below. Hence \({\mu ^{{i}}}\) in (73) becomes
$$\begin{aligned} {\mu ^{{i}}}_{\mathrm{on}} = \frac{{\mathrm{d}\left( {\alpha ^{{i}}} \right)} {\mathrm{d}\left( {\vartheta ^{{i}}} \right)}}{{k_{2}^{{i+1}}}{\mathrm{d}\left( {\vartheta ^{{i+1}}} \right)}} >0 \quad \forall \quad i \in [1,{p}]. \end{aligned}$$(75)
\(\square \)
Now, we present the proof of (74).
Claim 1
For all \(i \in [1,p]\)
where \({\alpha ^{{i}}}\) has been defined in (41a).
Proof
The strategy of the proof is to rewrite \({\mathrm{d}\left( {\vartheta ^{{i+1}}} \right)}\) in terms of \({\mathrm{d}\left( {\vartheta ^{{i}}} \right)}\). From (36), we have
\(\square \)
1.2 A2: Proof of Corollary 1
Proof
The closed-form expressions of \(\det \left( {\mathbf{A}^{{} {}}}_{\mathrm{off}} \right)\) and \(\det \left( {\mathbf{A}^{{} {}}}_{\mathrm{on}} \right)\) in (63a) and (63c), respectively, are derived in Lemma 4 in Sect. 8.
-
1.
Off Steady State The off steady state is given in (35a). We note that when we evaluate the conditions (55b) at the off steady state (35a), we obtain
$$\begin{aligned} {k_{2}^{{i}}} > {k_{3f}^{{i}}}, \quad {k_{3f}^{{i}}} \ge {k_{3f}^{{i}}}, \quad and \quad \frac{1}{\left( {k_{3f}^{{i}}} \right)^2} < \frac{1}{\left( {k_{2}^{{i}}} \right)^2} + \frac{1}{\left({k_{3f}^{{i}}}\right)^2}. \end{aligned}$$The later two expressions are always satisfied, given the parameters are positive. Now, the former condition \({k_{2}^{{i}}} > {k_{3f}^{{i}}}\) combined with the condition \({k_{2}^{{i}}} < {k_{3f}^{{i}}}\) in (55a), shows that we can guarantee the local stability of the off steady state for all parameter values that satisfy (56). Substituting (35a) into (56) yields
$$\begin{aligned} \gamma _{\mathrm{off}} := \prod _{i=1}^{p}\frac{{k_{1}^{{i}}} {k_{3b}^{{i}}}}{{k_{2}^{{i}}} {k_{3f}^{{i}}}} < 1, \end{aligned}$$(76)or equivalently
$$\begin{aligned} \prod _{i=1}^{p}{k_{2}^{{i}}} {k_{3f}^{{i}}} - \prod _{i=1}^{p}{k_{1}^{{i}}} {k_{3b}^{{i}}} > 0, \end{aligned}$$(63a)as stated in (63a).
-
2.
On Steady State The on steady state is given by (35b), which is parametrised by \({\vartheta ^{{i+1}}}\) defined in (36). Moreover, we recall that
$$\begin{aligned} {\mu ^{{i}}}_{\mathrm{on}} = \frac{{\mathrm{d}\left( {\alpha ^{{i}}} \right)} {\mathrm{d}\left( {\vartheta ^{{i}}} \right)}}{{k_{2}^{{i+1}}}{\mathrm{d}\left( {\vartheta ^{{i+1}}} \right)}}. \end{aligned}$$(75)In addition, we note that, with the expressions (38a) and (39b), \({\sigma ^{{i}}}\) in (50) may be rewritten as
$$\begin{aligned} {\sigma ^{{i}}}_{\mathrm{on}} = {k_{1}^{{i}}}{{\bar{c}}^{{i}}_{1}} = \frac{{k_{2}^{{i+1}}} {\mathrm{d}\left( {\vartheta ^{{i+1}}} \right)} }{{\mathrm{d}\left( {\vartheta ^{{i}}} \right)} }. \end{aligned}$$With the two former expressions, the stability condition (56) becomes
$$\begin{aligned} \gamma _{\mathrm{on}}:=\prod _{i=1}^{p}\gamma ^i_{\mathrm{on}} = \prod _{i=1}^{p}\frac{{k_{2}^{{i+1}}}}{{k_{2}^{{i}}}} \left( \frac{{\mathrm{d}\left( {\vartheta ^{{i+1}}} \right)}}{{\mathrm{d}\left( {\vartheta ^{{i}}} \right)}} \right)^2 \frac{{k_{2}^{{i+1}}}{k_{3f}^{{i}}}}{{k_{1}^{{i}}}{k_{3b}^{{i}}}}<1. \end{aligned}$$(77)Consequently, the small gain stability condition in (56) is
$$\begin{aligned} \prod _{i=1}^{p}{k_{1}^{{i}}} {k_{3b}^{{i}}} - \prod _{i=1}^{p}{k_{2}^{{i}}}{k_{3f}^{{i}}}&>0, \end{aligned}$$(63c)where we have taken into account the modularity of the superindex \(i\) in \({k_{2}^{{i+1}}}\).
\(\square \)
1.3 A3: Closed form of the determinant of \({\mathbf{A}^{{} {}}}_{\mathrm{off}}\) and \({\mathbf{A}^{{} {}}}_{\mathrm{on}}\)
In order to prove the closed-form expression of the determinant of (47) in Theorem 4, we will exploit the following lemma that presents a formula for the determinant of a block matrix.
Lemma 2
Let a square matrix \(\mathbf M\) be defined by blocks:
Provided the existence of \(\left(\mathbf{M^{22}}\right)^{-1}\), the determinant of \(\mathbf{M}\) is given by
where \(\mathrm{Adj}^T\left(\mathbf{X}\right)\) denotes the transpose of the Adjugate or Adjoint matrix of \(\,\mathbf{X}\).
We will also avail of the following theorem, which shows how to compute the determinant of a sum of matrices.
Lemma 3
(Matrix Determinant Lemma) Let \(\mathbf{A}\) be a non-singular square matrix and \(\mathbf{x, y}\) be column vectors of the dimension of \(\mathbf{A}\), then
The proof of the previous lemmas can be found in Bernstein (2009), for example. Now, we use these theorems to prove the forthcoming lemma.
Lemma 4
The determinant of (47) is given by
-
1.
Off Steady State
$$\begin{aligned} \det \left( \mathbf{A_{\mathrm{off}}} \right) = \prod _{i=1}^{p}{k_{2}^{{i}}} {k_{3f}^{{i}}} - \prod _{i=1}^{p}{k_{1}^{{i}}} {k_{3b}^{{i}}} \end{aligned}$$ -
2.
On Steady State
$$\begin{aligned} \det \left(\mathbf{A_{\mathrm{on}}} \right) = \prod _{i=1}^{p}{k_{1}^{{i}}} {k_{3b}^{{i}}} - \prod _{i=1}^{p}{k_{2}^{{i}}} {k_{3f}^{{i}}} \end{aligned}$$
Proof
The strategy of the proof follows a recursive application of the Lemma 2, finalised by the application of Lemma 3 to obtain a closed-form expression for the determinant. We note that applying \({p}-1\) times Lemma 2, the determinant of (47) can be expressed as
Moreover, we note that
where \({\sigma ^{{i}}}\) and \({\mu ^{{i}}}\) have been defined in (50). Hence
Substituting the expression above into (78), yields
By Lemma 3 and \({\mathbf{A}^{{i} {i}}}\) in (49a), the expression above becomes
We further consider the definition of the steady states, to conclude the proof.
-
1.
Off Steady State By substituting the definition of this equilibrium point in (35a), into (79), we obtain the expression
$$\begin{aligned} \det \left(\mathbf{A_{\mathrm{off}}} \right)&= \prod _{i=1}^{p}{k_{2}^{{i}}} {k_{3f}^{{i}}} - \prod _{i=1}^{p}{k_{1}^{{i}}} {k_{3b}^{{i}}}, \end{aligned}$$as desired.
-
2.
On Steady State This equilibrium point is parametrised by \({\vartheta ^{{i+1}}}\), defined in (36), via the relationships (39a) and (39b). Firstly, we analyse the product of the main blocks determinants in (79). By means of (39b) we can rewrite it as
$$\begin{aligned} \det \left( {\mathbf{A}^{{i} {i}}}_{\mathrm{on}} \right)&= {k_{2}^{{i}}}\frac{{k_{3f}^{{i}}} {k_{2}^{{i+1}}}{\mathrm{d}\left( {\vartheta ^{{i+1}}} \right)} - {k_{1}^{{i}}} {\mathrm{n}\left( {\vartheta ^{{i+1}}} \right)} }{{k_{2}^{{i+1}}}{\mathrm{d}\left( {\vartheta ^{{i+1}}} \right)}}\\ \det \left( {\mathbf{A}^{{i} {i}}}_{\mathrm{on}} \right)&= {\mathrm{d}\left( {\alpha ^{{i}}} \right)} \frac{{k_{2}^{{i}}} {\mathrm{d}\left( {\vartheta ^{{i}}} \right)}}{{k_{2}^{{i+1}}}{\mathrm{d}\left( {\vartheta ^{{i+1}}} \right)}}, \end{aligned}$$where we availed of the expression in (74). From the expression above, we note
$$\begin{aligned} \prod _{i=1}^{p}\det \left( {\mathbf{A}^{{i} {i}}}_{\mathrm{on}} \right)&= \prod _{i=1}^{p}{\mathrm{d}\left( {\alpha ^{{i}}} \right)} \frac{{k_{2}^{{i}}} {\mathrm{d}\left( {\vartheta ^{{i}}} \right)}}{{k_{2}^{{i+1}}}{\mathrm{d}\left( {\vartheta ^{{i+1}}} \right)}}\nonumber \\ \prod _{i=1}^{p}\det \left( {\mathbf{A}^{{i} {i}}}_{\mathrm{on}} \right)&= \prod _{i=1}^{p}{k_{1}^{{i}}}{k_{3b}^{{i}}} . \end{aligned}$$(80)Secondly, we note that, with the expressions (38a) and (39b), \({{\bar{c}}^{{i}}_{1}}\) may be rewritten as
$$\begin{aligned} {{\bar{c}}^{{i}}_{1}} = \frac{{k_{2}^{{i+1}}} {\mathrm{d}\left( {\vartheta ^{{i+1}}} \right)} }{{k_{1}^{{i}}} {\mathrm{d}\left( {\vartheta ^{{i}}} \right)} }. \end{aligned}$$Hence the later product in (79) becomes
$$\begin{aligned} \prod _{i=1}^{p}{k_{3f}^{{i}}}{k_{1}^{{i}}}{{\bar{c}}^{{1}}_{i}} = \prod _{i=1}^{p}{k_{3f}^{{i}}}{k_{2}^{{i+1}}}. \end{aligned}$$Subtraction of (80) and the relationship above, yields
$$\begin{aligned} \det \left( \mathbf{A}_{\mathrm{on}} \right) = \prod _{i=1}^{p}{k_{1}^{{i}}}{k_{3b}^{{i}}} - \prod _{i=1}^{p}{k_{3f}^{{i}}}{k_{2}^{{i}}}, \end{aligned}$$when exploiting the modularity of the product with respect to \({p}\).
\(\square \)
Now, we provide the proof of Theorem 5, which determines the stability of the on steady state under the conditions on the parameters described in (64).
1.4 A4: Proof of Theorem 5
Proof
Accounting for the conditions (64), the solution for \(\varOmega ^*\) in (59) is positive and real, for the positive sign of the square root. Let us rewrite this solution as
where
Then, from (58), the gain of the system evaluated in the peak frequency (81) is
In the derivation of the expression above, we exploited the definitions in (82) and (50). Except for the last factor, the expression above resembles the gain for the on steady state in (77), analysed in Theorem 4. Moreover, we have just considered that for some \(i\), the conditions in (64) are satisfied. Then the stability condition form the Small Gain Theorem can be expressed as
where the definition of \(\theta ^i\) is given in (66). Although exact, the condition above might be an intricate function of the parameters. In the following, we pursue a tractable bound of \(\gamma _{\mathrm{on}}^i\). To this end we note that (64c) implies
Using these two last inequalities in (83), we obtain the bound
Here we used the definitions of \({\mu ^{{i}}}_\mathrm{{on}}\) and \({\sigma ^{{i}}}\) in (50). Again, the expression above resembles the gain for the on steady state in (77), analysed in Theorem 4. Since the conditions in (64) just hold for some \(i\), the stability of the closed loop is guaranteed if (67) holds. \(\square \)
Rights and permissions
About this article
Cite this article
López-Caamal, F., Middleton, R.H. & Huber, H.J. Equilibria and stability of a class of positive feedback loops. J. Math. Biol. 68, 609–645 (2014). https://doi.org/10.1007/s00285-013-0644-z
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-013-0644-z