Abstract
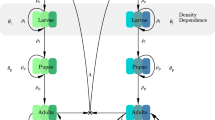
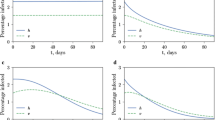
There is considerable interest in the management of insecticide resistance in mosquitoes. One possible approach to slowing down the evolution of resistance is to use late-life-acting (LLA) insecticides that selectively kill only the old mosquitoes that transmit malaria, thereby reducing selection pressure favoring resistance. In this paper we consider an age-structured compartmental model for malaria with two mosquito strains that differ in resistance to insecticide, using an SEI approach to model malaria in the mosquitoes and thereby incorporating the parasite developmental times for the two strains. The human population is modeled using an SEI approach. We consider both conventional insecticides that target all adult mosquitoes, and LLA insecticides that target only old mosquitoes. According to linearised theory the potency of the insecticide affects mainly the speed of evolution of resistance. Mutations that confer resistance can also affect other parameters such as mean adult life span and parasite developmental time. For both conventional and LLA insecticides the stability of the malaria-free equilibrium, with only the resistant mosquito strain present, depends mainly on these other parameters. This suggests that the main long term role of an insecticide could be to induce genetic changes that have a desirable effect on a vital parameter such as adult life span. However, when this equilibrium is unstable, numerical simulations suggest that a potent LLA insecticide can slow down the spread of malaria in humans but that the timing of its action is very important.




Similar content being viewed by others
References
Charlwood JD, Smith T, Billingsley PF, Takken W, Lyimo EOK, Meuwissen JHET (1997) Survival and infection probabilities of anthropophagic anophelines from an area of high prevalence of Plasmodium falciparum in humans. Bull Entomol Res 87:445–453
Chitnis N, Cushing JM, Hyman JM (2006) Bifurcation analysis of a mathematical model for malaria transmission. SIAM J Appl Math 67:24–45
Chitnis N, Hyman JM, Cushing JM (2008) Determining important parameters in the spread of malaria through the sensitivity analysis of a mathematical model. Bull Math Biol 70:1272–1296
Dietz K (1988) Mathematical models for transmission and control of malaria. Malar Princ Pract Malariol 2:1091–1133
Gazave E, Chevillon C, Lenormand T, Marquine M, Raymond M (2001) Dissecting the cost of insecticide resistance genes during the overwintering period of the mosquito Culex pipiens. Heredity 87:441–448
Glunt KD, Thomas MB, Read AF (2011) The effects of age, exposure history and malaria infection on the susceptibility of anopheles mosquitoes to low concentrations of pyrethroid. PLoS One 6(9):e24968. doi:10.1371/journal.pone.0024968
Gourley SA, Liu R, Wu J (2006/07) Some vector borne diseases with structured host populations: extinction and spatial spread. SIAM J Appl Math 67:408–433
Gourley SA, Liu R, Wu J (2011) Slowing the evolution of insecticide resistance in mosquitoes: a mathematical model. Proc R Soc Lond Ser A 467:2127–2148
Gurney WSC, Blythe SP, Nisbet RM (1980) Nicholson’s blowflies revisited. Nature 287:17–21
Hemingway J, Field L, Vontas J (2002) An overview of insecticide resistance. Science 298:96–97
Hemingway J, Hawkes NJ, McCarroll L, Ranson H (2004) The molecular basis of insecticide resistance in mosquitoes. Insect Biochem Mol Biol 34:653–665
Kelly-Hope L, Ranson H, Hemingway J (2008) Lessons from the past: managing insecticide resistance in malaria control and eradication programmes. Lancet Infect Dis 8:387–389
Killeen GF, McKenzie FE, Foy BD, Schieffelin C, Billingsley PF, Beier JC (2000) A simplified model for predicting malaria entomologic inoculation rates based on entomologic and parasitologic parameters relevant to control. Am J Trop Med Hyg 62:535–544
Koella JC, Rieu L, Paul REL (2002) Stage-specific manipulation of a mosquito’s host-seeking behavior by the malaria parasite Plasmodium gallinaceum. Behav Ecol 13:816–820
Koella JC, Lynch PA, Thomas MB, Read AF (2009) Towards evolution-proof malaria control with insecticides. Evol Appl 2:469–480
Lehmann T, Dalton R (2006) Kim EH, Dahl E, Diabate A, Dabire R, Dujardin JP. Genetic contribution to variation in larval development time, adult size, and longevity of starved adults of Anopheles gambiae. Infect Genet Evol 6:410–416
Read AF, Lynch PA, Thomas MB (2009) How to make evolution-proof insecticides for malaria control. PLoS Biol 7(4):e1000058. doi:10.1371/journal.pbio.1000058
Read AF, Thomas MB (2009) Mosquitoes cut short. Science 323:51–52
Rivero A, Vezilier J, Weill M, Read AF, Gandon S (2010) Insecticide control of vector-borne diseases: when is insecticide resistance a problem? PLoS Pathog 6(8):e1001000. doi:10.1371/journal.ppat.1001000
Rowland M, Hemingway J (1987) Changes in malathion resistance with age in Anopheles stephensi from Pakistan. Pesticide Biochem Physiol 28:239–247
Ruan S, Xiao D, Beier JC (2008) On the delayed Ross–Macdonald model for malaria transmission. Bull Math Biol 70(4):1098–1114
Smith HL (1995) Monotone dynamical systems. An introduction to the theory of competitive and cooperative systems. Mathematical Surveys and Monographs, vol 41. American Mathematical Society, Providence
Thomas MB, Read AF (2007) Can fungal biopesticides control malaria? Nature Rev Microbiol 5:377–383
Wonham MJ, de-Camino-Beck T, Lewis MA (2004) An epidemiological model for West Nile virus: invasion analysis and control applications. Proc R Soc Lond Ser B 271:501–507
Acknowledgments
We thank the referees for their helpful comments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, C., Gourley, S.A. & Liu, R. Delayed action insecticides and their role in mosquito and malaria control. J. Math. Biol. 68, 417–451 (2014). https://doi.org/10.1007/s00285-012-0638-2
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-012-0638-2