Abstract
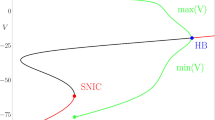
A technique is presented, based on the differential geometry of planar curves, to evaluate the excitability threshold of neuronal models. The aim is to determine regions of the phase plane where solutions to the model equations have zero local curvature, thereby defining a zero-curvature (inflection) set that discerns between sub-threshold and spiking electrical activity. This transition can arise through a Hopf bifurcation, via the so-called canard explosion that happens in an exponentially small parameter variation, and this is typical for a large class of planar neuronal models (FitzHugh–Nagumo, reduced Hodgkin–Huxley), namely, type II neurons (resonators). This transition can also correspond to the crossing of the stable manifold of a saddle equilibrium, in the case of type I neurons (integrators). We compute inflection sets and study how well they approximate the excitability threshold of these neuron models, that is, both in the canard and in the non-canard regime, using tools from invariant manifold theory and singularity theory. With the latter, we investigate the topological changes that inflection sets undergo upon parameter variation. Finally, we show that the concept of inflection set gives a good approximation of the threshold in both the so-called resonator and integrator neuronal cases.










Similar content being viewed by others
References
Benoît E, Callot JL, Diener F, Diener M (1981) Chasse au canard. Collect Math (31–32):37–119
Brøns M (1988) Bifurcations and instabilities in the Greitzer model for compressor system surge. Eng Ind 2(1):51–63
Brøns M (1989) Canards and excitation in Liénard equations. MAT-REPORT 1989–22, Mathematical Institute, Technical University of Denmark
Brøns M, Bar-Eli K (1991) Canard explosion and excitation in a model of the Belousov–Zhabotinskii reaction. J Phys Chem 95:8706–8713
Brøns M, Bar-Eli K (1994) Asymptotic analysis of canards in the EOE equations and the role of the inflection line. Proc Math Phys Sci 445:305–322
Bruce J, Giblin P (1992) Curves and singularities: a geometrical introduction to singularity theory. Cambridge University Press, Cambridge
Desroches M, Jeffrey MR (2011) Canards and curvature: the “smallness” of \(\varepsilon \) in slow-fast dynamics. Proc R Soc Lond A 467:2404–2421
Destexhe A, Bal T (2009) Dynamic-clamp: from principles to applications. Springer, Berlin
Eckhaus W (1983) Relaxation oscillations including a standard chase on French ducks. In: Verhulst F (ed) Asymptotic analysis II. Lecture notes in mathematics, vol 985. Springer, New York, pp 449–494
Fenichel N (1979) Geometric singular perturbation theory for ordinary differential equations. J Differ Equ 31(1):53–98
Fitzhugh R (1961) Impulses and physiological states in theoretical models of nerve membrane. Biophys J 1(6):445–466
Ginoux JM, Rossetto B (2006) Differential geometry and mechanics: applications to chaotic dynamical systems. Int J Bifurc Chaos Appl Sci Eng 16(4):887
Golubitsky M, Schaeffer DG (1988) Singularities and groups in bifurcation theory, vol 1. Applied mathematical sciences, series vol 51. Springer, New York
Guckenheimer J, Holmes P (1997) Nonlinear oscillations, dynamical systems, and bifurcations of vector fields. Springer, Berlin
Hodgkin A, Huxley A (1952) A quantitative description of membrane current and its application to conduction and excitation in nerve. J Physiol 117(4):500
Izhikevich E (2007) Dynamical systems in neuroscience: the geometry of excitability and bursting. The MIT press, Cambridge
Keener J, Sneyd J (1998) Mathematical physiology. Springer, Berlin
Krupa M, Popovic N, Kopell N, Rotstein H (2008) Mixed-mode oscillations in a three time-scale model for the dopaminergic neuron. Chaos 18(1):5106
Krupa M, Szmolyan P (2001) Relaxation oscillation and canard explosion. J Differ Equ 174(2):312–368
Krupa M, Szmolyan P (2001) Extending geometric singular perturbation theory to non hyperbolic points—fold and canard points in two dimensions. SIAM J Math Anal 33(2):286–314
Moehlis J (2006) Canards for a reduction of the Hodgkin–Huxley equations. J Math Biology 52(2):141–153
Munoz L, Stockton JF, Otani N (2010) Applications of control theory to the dynamics and propagation of cardiac action pontentials. Ann Biomed Eng 38(9):2865–2876
Nagumo J, Arimoto S, Yoshizawa S (1962) An active pulse transmission line simulating nerve axon. Proc IRE 50(10):2061–2070
Okuda M (1976) A new method of nonlinear analysis for shaping and threshold actions. J Phys Soc Jpn 41:1815–1816
Peng B, Gaspar V, Showalter K (1991) False bifurcations in chemical systems: canards. Phil Trans R Soc Lond A 337(1646):275–289
Platkiewicz J, Brette R (2010) A threshold equation for action potential initiation. PLoS Comput Biol 6(7):e1000850
Prescott S, De Koninck Y, Sejnowski T (2008) Biophysical basis for three distinct dynamical mechanisms of action potential initiation. PLoS Comput Biol 4(10)
Rinzel J (1985) Excitation dynamics: insights from simplified membrane models. Fed Proc 44:2944
Rossokhin A, Saakyan Y (1992) Study of the dependence of the generation threshold of the nerve impulse on the mode of formation of the input signal on the basis of the hodgkin-huxley model. Biophysics 37(6):969–973
Rotstein H, Wechselberger M, Kopell N (2008) Canard induced mixed-mode oscillations in a medial entorhinal cortex layer ii stellate cell model. SIAM J Appl Dyn Syst 7(4):1582–1611
Rubin J, Wechselberger M (2008) The selection of mixed-mode oscillations in a hodgkin-huxley model with multiple timescales. Chaos 18(1):15105
Sekerli M, Del Negro C, Lee R, Butera R (2004) Estimating action potential thresholds from neuronal time-series: new metrics and evaluation of methodologies. IEEE Trans Biomed Eng 51(9):1665–1672
Sieber J, Gonzalez-Buelga A, Neild S, Wagg D, Krauskopf B (2008) Experimental continuation of periodic orbits through a fold. Phys Rev Lett 100(24):244101
Sontag E (1998) Mathematical control theory: deterministic finite dimensional systems. Springer, Berlin
Tsitolovsky L, Babkina N (2002) Neurons evaluate both the amplitude and the meaning of signals. Brain Res 946(1):104–118
Tsitolovsky L, Shvedov A (1997) Instrumental conditioning of the activity of putative command neurons in the mollusk Helix. Brain Res 745(1–2):271–282
Acknowledgments
The research of M.D. and S.R. was supported by EPSRC under grant EP/E032249/1.
Author information
Authors and Affiliations
Corresponding author
Appendix: The full Hodgkin–Huxley equations
Appendix: The full Hodgkin–Huxley equations
We recall here the four-dimensional Hodgkin–Huxley model from Hodgkin and Huxley (1952) together with the original parameter values. The equations read
where
The original parameter values for the Hodgkin–Huxley equation are given in the table below
Ionic conductances (\(\text{ mmho}/\text{ cm}^2\)) | Reversal potentials (\(\text{ m}V\)) | Membrane capacitance (\({\mu }F/\text{ cm}^2\)) | ||||
|---|---|---|---|---|---|---|
\(\bar{g}_{Na}\) | \(\bar{g}_K\) | \(g_L\) | \(V_{Na}\) | \(V_K\) | \(V_L\) | \(C\) |
\(120\) | \(36\) | \(0.3\) | \(50\) | \(-77\) | \(-54.4\) | \(1\) |
Rights and permissions
About this article
Cite this article
Desroches, M., Krupa, M. & Rodrigues, S. Inflection, canards and excitability threshold in neuronal models. J. Math. Biol. 67, 989–1017 (2013). https://doi.org/10.1007/s00285-012-0576-z
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-012-0576-z
Keywords
- Slow-fast systems
- Canard solutions
- Excitability
- Singularity theory
- Inflection set
- Threshold
- Resonators
- Integrators