Abstract
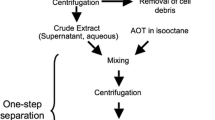
A new and efficient method for the purification of levansucrase from cell-free extracts of a flocculant mutant of Zymomonas mobilisATCC 10988 was developed. Levansucrase activity was almost completely recovered and purified by a factor of 15 after precipitation with 0.1 m MnCl2 as a first capturing step. The enzyme was homogeneously purified by ultrafiltration and anion-exchange chromatography and exhibited a levan-forming activity of 39.2 U mg−1. The native enzyme formed large aggregates with an apparent molecular mass of more than 106 Da as determined by size-exclusion chromatography, whereas denaturing SDS-PAGE indicated an apparent molecular mass of 50 kDa for the subunits.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 10 October 2000 / Accepted: 17 November 2000
Rights and permissions
About this article
Cite this article
Vigants, A., Hicke, HG. & Marx, S. A Simple and Efficient Method for the Purification of Membrane-Bound Levansucrase from Zymomonas mobilis . Curr Microbiol 42, 415–418 (2001). https://doi.org/10.1007/s002840010239
Published:
Issue Date:
DOI: https://doi.org/10.1007/s002840010239