Abstract
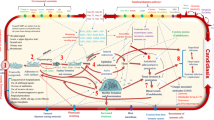
Invasive candidiasis is associated with high mortality in immunocompromised and hospitalized patients. Candida albicans is the main pathological agent followed by Candida glabrata, Candida krusei, Candida parapsilosis, and Candida tropicalis. These pathogens colonize different host tissues in humans as they are able to neutralize the reactive species generated from nitrogen and oxygen during the respiratory burst. Among the enzymatic mechanisms that Candida species have developed to protect against free radicals are enzymes with antioxidant and immunodominant functions such as flavohemoglobins, catalases, superoxide dismutases, glutathione reductases, thioredoxins, peroxidases, heat-shock proteins, and enolases. These mechanisms are under transcriptional regulation by factors such as Cta4p, Cwt1p, Yap1p, Skn7p, Msn2p, and Msn4p. However, even though it has been proposed that all Candida species have similar enzymatic systems, it has been observed that they respond differentially to various types of stress. These differential responses may explain the colonization of different organs by each species. Here, we review the enzymatic mechanisms developed by C. albicans and C. glabrata species in response to oxidative and nitrosative stresses. Lack of experimental information for other pathogenic species limits a comparative approach among different organisms.
Similar content being viewed by others
References
Alarco AM, Raymond M (1999) The bZip transcription factor Cap1p is involved in multidrug resistance and oxidative stress response in Candida albicans. J Bacteriol 181:700–708
Bhattacharjee A, Majumdar U, Maity D, Sarkar TS, Goswami AM, Sahoo R, Ghosh S (2010) Characterizing the effect of nitrosative stress in Saccharomyces cerevisiae. Arch Biochem Biophys 496:109–116
Brown JP, Haynes K, Quinn J (2009) Nitrosative and oxidative stress responses in fungal pathogenicity. Curr Opin Microbiol 12:384–391
Calderone RA (2002) Candida and candidiasis. ASM Press, Washington, DC
Chauhan N, Latge JP, Calderone R (2006) Signalling and oxidant adaptation in Candida albicans and Aspergillus fumigatus. Nat Rev Microbiol 4:435–444
Chaves GM, Bates S, MacCallum DM, Odds FC (2007) Candida albicans GRX2, encoding a putative glutaredoxin, is required for virulence in a murine model. Genet Mol Res 6:1051–1063
Chaves GM, da Silva WP (2012) Superoxide dismutases and glutaredoxins have a distinct role in the response of Candida albicans to oxidative stress generated by the chemical compounds menadione and diamide. Mem Inst Oswaldo Cruz 107:998–1005
Chiranand W, McLeod I, Zhou H, Lynn JJ, Vega LA, Myers H, Yates JR 3rd, Lorenz MC, Gustin MC (2008) CTA4 transcription factor mediates induction of nitrosative stress response in Candida albicans. Eukaryot Cell 7:268–278
Cuéllar-Cruz M, Briones-Martin-del-Campo M, Cañas-Villamar I, Montalvo-Arredondo J, Riego-Ruiz L, Castaño I, De Las Peñas A (2008) High resistance to oxidative stress in the fungal pathogen Candida glabrata is mediated by a single catalase, Cta1p, and is controlled by the transcription factors Yap1p, Skn7p, Msn2p, and Msn4p. Eukaryot Cell 7:814–825
Cuéllar-Cruz M, Castaño I, Arroyo-Helguera O, De Las Peñas A (2009) Oxidative stress response to menadione and cumene hydroperoxide in the opportunistic fungal pathogen Candida glabrata. Mem Inst Oswaldo Cruz 104:649–654
Cuéllar-Cruz M, Gutiérrez-Sánchez G, López-Romero E, Ruiz-Baca E, Villagómez-Castro JC, Rodríguez-Sifuentes L (2013) Identification of heat shock proteins in Candida albicans and enolases in Candida glabrata and Candida krusei involved in the response to oxidative stress. Cent Eur J Biol 8:337–345
Enjalbert B, Nantel A, Whiteway M (2003) Stress-induced gene expression in Candida albicans: absence of a general stress response. Mol Biol Cell 14:1460–1467
Estruch F, Carlson M (1993) Two homologous zinc finger genes identified by multicopy suppression in a SNF1 protein kinase mutant of Saccharomyces cerevisiae. Mol Cell Biol 13:3872–3881
Fernández-Arenas E, Cabezón V, Bermejo C, Arroyo J, Nombela C, Diez-Orejas R, Gil C (2007) Integrated proteomics and genomics strategies bring new insight into Candida albicans response upon macrophage interaction. Mol Cell Proteomics 6:460–478
Ferrari CK, Souto PC, França EL, Honorio-França AC (2011) Oxidative and nitrosative stress on phagocytes’ function: from effective defense to immunity evasion mechanisms. Arch Immunol Ther Exp 59:441–448
Fradin C, De Groot P, MacCallum D, Schaller M, Klis F, Odds FC, Hube B (2005) Granulocytes govern the transcriptional response, morphology and proliferation of Candida albicans in human blood. Mol Microbiol 56:397–415
Frohner IE, Bourgeois C, Yatsyk K, Majer O, Kuchler K (2009) Candida albicans cell surface superoxide dismutases degrade host-derived reactive oxygen species to escape innate immune surveillance. Mol Microbiol 71:240–252
Gardner PR, Gardner AM, Martin LA, Salzman AL (1998) Nitric oxide dioxygenase: an enzymic function for flavohemoglobin. Proc Natl Acad Sci USA 95:10378–10383
Gonzalez-Parraga P, Hernandez JÁ, Arguelles JC (2003) Role of antioxidant enzymatic defences against oxidative stress H(2)O(2) and the acquisition of oxidative tolerance in Candida albicans. Yeast 20:1161–1169
Herrero E (2005) Evolutionary relationships between Saccharomyces cerevisiae and other fungal species as determined from genome comparisons. Rev Iberoam Micol 22:217–222
Horan S, Bourges I, Meunier B (2006) Transcriptional response to nitrosative stress in Saccharomyces cerevisiae. Yeast 23:519–535
Hromatka BS, Noble SM, Johnson AD (2005) Transcriptional response of Candida albicans to nitric oxide and the role of the YHB1 gene in nitrosative stress and virulence. Mol Biol Cell 16:4814–4826
Hwang CS, Baek YU, Yim HS, Kang SO (2003) Protective roles of mitochondrial manganese-containing superoxide dismutase against various stresses in Candida albicans. Yeast 20:929–941
Hwang CS, Rhie GE, Oh JH, Huh WK, Yim HS, Kang SO (2002) Copper- and zinc-containing superoxide dismutase (Cu/ZnSOD) is required for the protection of Candida albicans against oxidative stresses and the expression of its full virulence. Microbiology 148:3705–3713
Jong AY, Chen SH, Stins MF, Kim KS, Tuan TL, Huang SH (2003) Binding of Candida albicans enolase to plasmin(ogen) results in enhanced invasion of human brain microvascular endothelial cells. J Med Microbiol 52:615–622
Kaloriti D, Tillmann A, Cook E, Jacobsen M, You T, Lenardon M, Ames L, Barahona M, Chandrasekaran K, Coghill G, Goodman D, Gow NA, Grebogi C, Ho HL, Ingram P, McDonagh A, de Moura AP, Pang W, Puttnam M, Radmaneshfar E, Romano MC, Silk D, Stark J, Stumpf M, Thiel M, Thorne T, Usher J, Yin Z, Haynes K, Brown AJ (2012) Combinatorial stresses kill pathogenic Candida species. Med Mycol 50:699–709
Krems B, Charizanis C, Entian KD (1995) Mutants of Saccharomyces cerevisiae sensitive to oxidative and osmotic stress. Curr Genet 27:427–434
Kusch H, Engelmann S, Albrecht D, Morschhäuser J, Hecker M (2007) Proteomic analysis of the oxidative stress response in Candida albicans. Proteomics 7:686–697
Laín A, Elguezabal N, Amutio E, Fernández de Larrinoa I, Moragues MD, Pontón J (2008) Use of recombinant antigens for the diagnosis of invasive candidiasis. Clin Dev Immunol. doi:10.1155/2008/721950
Lushchak OV, Inoue Y, Lushchak VI (2010) Regulatory protein Yap1 is involved in response of yeast Saccharomyces cerevisiae to nitrosative stress. Biochemistry (Mosc) 75:629–664
Lushchak OV, Lushchak VI (2008) Catalase modifies yeast Saccharomyces cerevisiae response towards S-nitrosoglutathione-induced stress. Redox Rep 13:283–291
Martchenko M, Alarco AM, Harcus D, Whiteway M (2004) Superoxide dismutases in Candida albicans: transcriptional regulation and functional characterization of the hyphal-induced SOD5 gene. Mol Biol Cell 15:456–467
Martínez-Pastor MT, Marchler G, Schüller C, Marchler-Bauer A, Ruis H, Estruch F (1996) The Saccharomyces cerevisiae zinc finger proteins Msn2p and Msn4p are required for transcriptional induction through the stress response element (STRE). EMBO J 15:2227–2235
Miceli MH, Diaz JA, Lee SA (2011) Emerging opportunistic yeast infections. Lancet Infect Dis 11:142–151
Miramón P, Kasper L, Hube B (2013) Thriving within the host: Candida spp. interactions with phagocytic cells. Med Microbiol Immunol. doi:10.1007/s00430-013-0288-z
Moradas-Ferreira P, Costa V (2000) Adaptive response of the yeast Saccharomyces cerevisiae to reactive oxygen species: defences, damage and death. Redox Rep 5:277–285
Moreno I, Martinez-Esparza M, Laforet LC, Sentandreu R, Ernst JF, Valentin E (2010) Dosage-dependent roles of the Cwt1 transcription factor for cell wall architecture, morphogenesis, drug sensitivity and virulence in Candida albicans. Yeast 27:77–87
Murray WW, Rachubinski RA (1989) Nucleotide sequence of peroxisomal catalase from the yeast Candida tropicalis pK233: identification of an upstream BamHlI site polymorphism. Nucleic Acids Res 17:3600
Nakagawa Y, Kanbe T, Mizuguchi I (2003) Disruption of the human pathogenic yeast Candida albicans catalase gene decreases survival in mouse-model infection and elevates susceptibility to higher temperature and to detergents. Microbiol Immunol 47:395–403
Nantel A, Dignard D, Bachewich C, Harcus D, Marcil A, Bouin AP, Sensen CW, Hogues H, van het Hoog M, Gordon P, Rigby T, Benoit F, Tessier DC, Thomas DY, Whiteway M (2002) Transcription profiling of Candida albicans cells undergoing the yeast-to-hyphal transition. Mol Biol Cell 13:3452–3465
Nathan C, Shiloh MU (2000) Reactive oxygen and nitrogen intermediates in the relationship between mammalian hosts and microbial pathogens. Proc Natl Acad Sci USA 97:8841–8848
Nicholls S, Straffon M, Enjalbert B, Nantel A, Macaskill S, Whiteway M, Brown AJ (2004) Msn2- and Msn4-like transcription factors play no obvious roles in the stress responses of the fungal pathogen Candida albicans. Eukaryot Cell 3:1111–1123
Okada H, Ueda M, Sugaya T, Atomi H, Mozaffar S, Hishida T, Teranishi Y, Okazaki K, Takechi T, Kamiryo T, Tanaka A (1987) Catalase gene of the yeast Candida tropicalis. Sequence analysis and comparison with peroxisomal and cytosolic catalases from other sources. Eur J Biochem 170:105–110
Palmerini CA, Palombari R, Perito S, Arienti G (2003) NO synthesis in human saliva. Free Radic Res 37:29–31
Pfaller MA, Diekema DJ (2007) Epidemiology of invasive candidiasis: a persistent public health problem. Clin Microbiol Rev 20:133–163
Poole RK, Hughes MN (2000) New functions for the ancient globin family: bacterial responses to nitric oxide and nitrosative stress. Mol Microbiol 36:775–783
Rodríguez-Manzaneque MT, Ros J, Cabiscol E, Sorribas A, Herrero E (1999) Grx5 Glutaredoxin plays a central role in protection against protein oxidative damage in Saccharomyces cerevisiae. Mol Cell Biol 19:8180–8190
Roetzer A, Gregori C, Jennings AM, Quintin J, Ferrandon D, Butler G, Kuchler K, Ammerer G, Schüller C (2008) Candida glabrata environmental stress response involves Saccharomyces cerevisiae Msn2/4 orthologous transcription factors. Mol Microbiol 69:603–620
Roetzer A, Klopf E, Gratz N, Marcet-Houben M, Hiller E, Rupp S, Gabaldón T, Kovarik P, Schüller C (2011) Regulation of Candida glabrata oxidative stress resistance is adapted to host environment. FEBS Lett 585:319–327
Rogers PD, Vermitsky JP, Edlind TD, Hilliard GM (2006) Proteomic analysis of experimentally induced azole resistance in Candida glabrata. J Antimicrob Chemother 58:434–438
Schüller C, Brewster JL, Alexander MR, Gustin MC, Ruis H (1994) The HOG pathway controls osmotic regulation of transcription via the stress response element (STRE) of the Saccharomyces cerevisiae CTT1 gene. EMBO J 13:4382–4389
Sellam A, Tebbji F, Whiteway M, Nantel A (2012) A novel role for the transcription factor Cwt1p as a negative regulator of nitrosative stress in Candida albicans. PLoS ONE 7:e43956
Sen CK (1998) Redox signaling and the emerging therapeutic potential of thiol antioxidants. Biochem Pharmacol 55:1747–1758
Seneviratne CJ, Wang Y, Jin L, Abiko Y, Samaranayake LP (2008) Candida albicans biofilm formation is associated with increased anti-oxidative capacities. Proteomics 8:2936–2947
Shin DH, Jung S, Park SJ, Kim YJ, Ahn JM, Kim W, Choi W (2005) Characterization of thiol-specific antioxidant 1 (TSA1) of Candida albicans. Yeast 22:907–918
Singh P, Chauhan N, Ghosh A, Dixon F, Calderone R (2004) SKN7 of Candida albicans: mutant construction and phenotype analysis. Infect Immun 72:2390–2394
Sobko T, Reinders CI, Jansson E, Norin E, Midtvedt T, Lundberg JO (2005) Gastrointestinal bacteria generate nitric oxide from nitrate and nitrite. Nitric Oxide 13:272–278
Srikantha T, Zhao R, Daniels K, Radke J, Soll DR (2005) Phenotypic switching in Candida glabrata accompanied by changes in expression of genes with deduced functions in copper detoxification and stress. Eukaryot Cell 4:1434–1445
Srinivasa K, Kim NR, Kim J, Kim M, Bae JY, Jeong W, Kim W, Choi W (2012) Characterization of a putative thioredoxin peroxidase prx1 of Candida albicans. Mol Cells 33:301–307
Tillmann A, Gow NAR, Brown AJP (2011) Nitric oxide and nitrosative stress tolerance in yeast. Biochem Soc Trans 39:219–223
Ullmann BD, Myers H, Chiranand W, Lazzell AL, Zhao Q, Vega LA, Lopez-Ribot JL, Gardner PR, Gustin MC (2004) Inducible defense mechanism against nitric oxide in Candida albicans. Eukaryot Cell 3:715–723
Wysong DR, Christin L, Sugar AM, Robbins PW, Diamond RD (1998) Cloning and sequencing of a Candida albicans catalase gene and effects of disruption of this gene. Infect Immun 66:1953–1961
Zhang X, De Micheli M, Coleman ST, Sanglard D, Moye-Rowley WS (2000) Analysis of the oxidative stress regulation of the Candida albicans transcription factor, Cap1p. Mol Microbiol 36:618–629
Znaidi S, Barker KS, Weber S, Alarco AM, Liu TT, Boucher G, Rogers PD, Raymond M (2009) Identification of the Candida albicans Cap1p regulon. Eukaryot Cell 8:806–820
Acknowledgments
The authors are thankful for the financial support provided by Grants: Proyecto-Institucional-UGTO-id202/2013 from the University of Guanajuato, México and PROMEP-UGTO-PTC-328 (M Cuéllar-Cruz, respectively).
Conflict of interest
The authors declare that there were no conflicts of interest with any organization or entity with a financial interest or financial conflict with the material discussed in this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cuéllar-Cruz, M., López-Romero, E., Ruiz-Baca, E. et al. Differential Response of Candida albicans and Candida glabrata to Oxidative and Nitrosative Stresses. Curr Microbiol 69, 733–739 (2014). https://doi.org/10.1007/s00284-014-0651-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-014-0651-3