Abstract
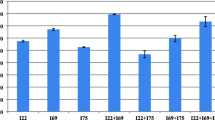
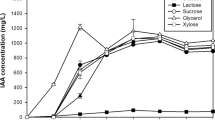
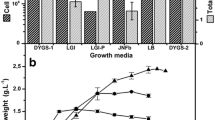
Production of Indole-3-acetic acid (IAA) in 35 different symbiotic and non-symbiotic nitrogen-fixing bacteria strains isolated from soil and plant roots was studied and assayed by chromatography and colorimetric methods. These bacteria included Agrobacterium, Paenibacillus, Rhizobium, Klebsiella oxytoca, and Azotobacter. The best general medium and synergism effects of isolates for IAA production were investigated. Effects of different variables containing physical parameters and key media components and optimization of condition for IAA production were performed using the Design of Experiments. Qualitek-4 (W32b) software for automatic design and analysis of the experiments, both based on Taguchi method was used. The results showed that Rhizobium strains, symbiotic, and Paenibacillus non-symbiotic bacteria yielded the highest concentrations of IAA (in the range of 5.23–0.27 and 4.90–0.19 ppm IAA/mg biomass, respectively) and IAA production was increased by synergism effect of them. Yeast Extract Mannitol medium supplemented with l-tryptophan was the best general medium for IAA production. The analysis of experimental data using Taguchi method indicated that nitrogen source is very prominent variable in affecting the yield and mannitol as carbon source, potassium nitrate (1%), and l-tryptophan (3 g/l) as nitrogen sources after 72-h incubation at 30°C were the optimum conditions for production of IAA. 5.89 ppm IAA/mg biomass was produced under these optimal conditions.






Similar content being viewed by others
References
Ahmad F, Ahmad I, Khan MS (2005) Indole acetic acid production by the indigenous isolates of Azotobacter and fluorescent Pseudomonas in the presence and absence of tryptophan. Turk J Biol 29:29–34
Bric JM, Bostock RM, Silverstone SE (1991) Rapid in situ assay for indoleacetic acid production by bacteria immobilized on a nitrocellulose membrane. Appl Environ Microbiol 57:535–538
Cattelan AJ, Hartel PG, Fuhrmann JJ (1999) Screening for plant growth-promoting rhizobacteria to promote early soybean growth. Plant Cell Physiol 63:1670–1680
Datta C, Basu PS (2000) Indole acetic acid production by a Rhizobium species from root nodules of a leguminous shrub. Microbiol Res 155:123–127
Diemaite J (2004) Peculiarities of the formation of indole-3-acetic acid–protein complexes in yeast Saccharomyces cerevisiae plasmalemma. Biologija 10:36–38
Garrity GM, Brenner DG, Krieg NR (2005) Bergey’s manual of Systematic Bacteriology. Vol. 2. Part. A. Springer, New York
Glickmann E, Dessaux Y (1995) A critical examination of the specificity of the Salkowski reagent for indolic compounds produced by phytopathogenic bacteria. Appl Environ Microbiol 61:793–796
Kang BR, Yang KY, Cho BH et al (2006) Production of indole-3-acetic acid in the plant-beneficial strain Pseudomonas chlororaphis O6 is negatively regulated by the Global Sensor Kinase GacS. Curr Microbiol 52:473–476
Kawaguchi M, Syono K (1996) The excessive production of indole-3-acetic acid and its significance in studies of the biosynthesis of this regulator of plant growth and development. Plant Cell Physiol 37(8):1043–1048
Kittell BL, Helinski DR, Ditta GS (1989) Aromatic aminotransferase activity and indoleacetic acid production in Rhizobium meliloti. J Bacteriol 171:5458–5466
Leveau JHJ, Lindow SE (2005) Utilization of the plant hormone indole-3-acetic acid for growth by Pseudomonas putida strain 1290. Appl Environ Microbiol 71:2365–2371
Lindow SE, Desurmont C, Elkins R et al (1998) Occurrence of indole-3-acetic acid-producing bacteria on pear trees and their association with fruit russet. Phytopathology 88:1149–1157
Matsukawa E, Nakagawa Y, Iimura Y et al (2007) Stimulatory effect of indole-3-acetic acid on aerial mycelium formation and antibiotic production in Streptomyces spp. Actinomycetologica 21:32–39
Phi QT, Park SY, Park SH et al (2008) Isolation and characterization of transposon-insertional mutants from Paenibacillus polymyxa E681 altering the biosynthesis of indole-3-acetic acid. Curr Microbiol 56(5):524–530
Robinson M, Riov J, Sharon A (1998) Indole-3-acetic acid biosynthesis in Colletotrichum gloeosporioides f. sp. Aeschynomene. Appl Environ Microbiol 64:5030–5032
Spaepen S, Vanderleyden J, Remans R (2007) Indole-3-acetic acid in microbial and microorganism-plant signaling. FEMS Microbiol Rev 31(4):425–448
Sreenivas RR, Prakasham RS, Krishna (2004) Xylitol production by Candida sp.: parameter optimization using Taguchi approach. Process Biochem 39:951–956
Sridevi M, Mallaiah KV (2007) Production of indole-3-acetic acid by Rhizobium isolates from sesbania species. Afr J Microbiol Res 1(7):125–128
Suzuki Sh, He Y, Oyaizu H (2003) Indole-3-acetic acid production in Pseudomonas fluorescens HP72 and its association with suppression of creeping bentgrass brown Patch. Curr Microbiol 47:138–143
Teale WD, Paponov IA, Palme K (2006) Auxin in action: signaling, transport and the control of plant growth and development. Mol Cell Biol 7:847–859
Acknowledgments
This work was supported financially by the University of Isfahan. The authors wish to thank Afsaneh Shokri and Dr.Arman Rostamzad for their valuable assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shokri, D., Emtiazi, G. Indole-3-Acetic Acid (IAA) Production in Symbiotic and Non-Symbiotic Nitrogen-Fixing Bacteria and its Optimization by Taguchi Design. Curr Microbiol 61, 217–225 (2010). https://doi.org/10.1007/s00284-010-9600-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-010-9600-y