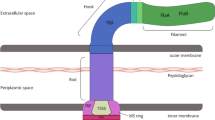
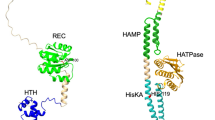
Abstract
We recently demonstrated that osmoregulated periplasmic glucans (OPGs) of Salmonella enterica serovar Typhimurium are required for optimal mouse virulence. However, lack of OPGs also generated pleiotropic phenotypes such as reduced motility and slower growth rate under hypoosmotic growth conditions. Whether the observed suboptimal virulence of opg mutants was due to reduced motility was investigated by isolating fully motile revertants of opgGH mutants. Motility revertants remained defective in OPGs synthesis and restitution of motility did not restore mouse virulence. In Escherichia coli, inactivation of rcsB, rcsD, and rcsF lead to restoration of motility in opg mutants, while in Salmonella strains, inactivation of the Rcs pathway is known to attenuate virulence. DNA sequence analysis revealed that except for two silent mutations no other changes in the DNA sequences of Rcs pathway genes were detected in the motility-revertant strain. Moreover, transcripts of all the Rcs phosphorelay pathway genes were detected in opgGH mutants and revertant strain. The data suggest that Salmonella may have distinctive regulatory elements in addition to Rcs phosphorelay genes to rescue motility of opg mutants and affecting also mouse virulence.



Similar content being viewed by others
References
Arricau N, Hermant D, Waxin H et al (1998) The RcsB-RcsC regulatory system of Salmonella typhi differentially modulates the expression of invasion proteins, flagellin and Vi antigen in response to osmolarity. Mol Microbiol 29:835–850
Bhagwat AA, Phadke RP, Wheeler D et al (2003) Computational methods and evaluation of RNA stabilization reagents for genome-wide expression studies. J Microbiol Methods 55:399–409
Bhagwat AA, Jun W, Liu L et al (2009) Osmoregulated periplasmic glucans of Salmonella enterica serovar Typhimurium are required for optimal virulence in mice. Microbiology 155:229–237
Chen R, Bhagwat AA, Keister DL (2003) A motility revertant of the ndvB mutation in Bradyrhizobium japonicum. Curr Microbiol 47:431–433
Datsenko KA, Wanner BL (2000) One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc Natl Acad Sci USA 97:6640–6645
Detweiler CS, Monack DM, Brodsky IE et al (2003) virK, somA and rcsC are important for systemic Salmonella enterica serovar Typhimurium infection and cationic peptide resistance. Mol Microbiol 48:385–400
Ebel W, Vaughn GJ, Peters HK III et al (1997) Inactivation of mdoH leads to increased expression of colanic acid capsular polysaccharide in Escherichia coli. J Bacteriol 179:6858–6861
Erickson KD, Detweiler CS (2006) The Rcs phosphorelay system is specific to enteric pathogens/commensals and activates ydeI, a gene important for persistent Salmonella infection of mice. Mol Microbiol 62:883–894
Fiedler W, Rotering H (1988) Properties of Escherichia coli mutants lacking membrane-derived oligosaccharides. J Biol Chem 263:14684–14689
Francez-Charlot A, Laugel B, Van Gemert A et al (2003) RcsCDB His-Asp phosphorelay system negatively regulates the flhDC operon in Escherichia coli. Mol Microbiol 49:823–832
Garcia-Calderon CB, Garcia-Quintanilla M, Casadesus J et al (2005) Virulence attenuation in Salmonella enterica rcsC mutants with constitutive activation of the Rcs system. Microbiology 151:579–588
Girgis HS, Liu Y, Ryu WS et al (2007) A comprehensive genetic characterization of bacterial motility. PLoS Genet 3:1644–1660
Huang Y, Ferrieres L, Clarke DJ (2006) The role of the Rcs phosphorelay in Enterobacteriaceae. Res Microbiol 157:206–212
Kennedy EP (1982) Osmotic regulation and biosynthesis of membrane-derived oligosaccharides in Escherichia coli. Proc Natl Acad Sci USA 79:1092–1095
Kennedy EP (1996) Membrane-derived oligosaccharides (periplasmic beta-D-glucans) of Escherichia coli. In: Neidhardt FC, Curtis R III, Ingraham JL, Lin ECC, Low KB, Magasanik B, Reznikoff WS, Riley M, Schaechter M, Umbarger HE (eds) Escherichia coli and Salmonella cellular and molecular biology. American Society for Microbiology, Washington, DC, pp 1064–1074
Kennedy EP, Rumley MK (1988) Osmotic regulation of biosynthesis of membrane-derived oligosaccharides in Escherichia coli. J Bacteriol 170:2457–2461
Liu L, Tan S, Jun W et al (2009) Osmoregulated periplasmic glucans are needed for competitive growth and biofilm formation by Salmonella enterica serovar Typhimurium in leafy-green vegetable wash waters and colonization in mice. FEMS Microbiol Lett 292:13–20
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-delataCT) method. Methods 25:402–408
Majdalani N, Gottesman S (2005) The Rcs phosphorelay: a complex signal transduction system. Annu Rev Microbiol 59:379–405
McQuiston J, Fields PI, Tauxe RV et al (2008) Do Salmonella carry spare tires? Trends Microbiol 16:142–148
Miller KJ, Kennedy EP, Reinhold VN (1986) Osmotic adaptation by Gram-negative bacteria: possible role for periplasmic oligosaccharides. Science 231:48–51
Mouslim C, Delgado M, Groisman EA (2004) Activation of the RcsC/YojN/RcsB phosphorelay system attenuates Salmonella virulence. Mol Microbiol 54:386–395
Pfaffl MW (2001) A new mathematical model for relative quantitation in real-time RT-PCR. Nucleic Acids Res 29:2002–2007
Wang Q, Zhao Y, McClelland M et al (2007) The RcsCDB signalling system and swarming motility in Salmonella enterica serovar Typhimurium: dual regulation of flagellar and SPI-2 virulence genes. J Bacteriol 189:8447–8457
Acknowledgment
We thank Claudia Lam for able technical expertise.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kannan, P., Dharne, M., Smith, A. et al. Motility Revertants of opgGH Mutants of Salmonella enterica Serovar Typhimurium Remain Defective in Mice Virulence. Curr Microbiol 59, 641–645 (2009). https://doi.org/10.1007/s00284-009-9486-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-009-9486-8