Abstract
We report hereby the results of the french multicentric randomized PEGASE 04 protocol established to evaluate the impact on survival of high-dose chemotherapy over conventional chemotherapy for MBC patients.
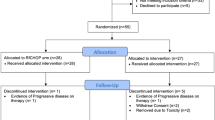
Patients and methods:Inclusion criteria were : age < 60 year, PS < 2, adenocarcinoma initially metastatic or in first relapse, chemosensitive disease. Randomization was done after 4-6 courses of conventionnal chemotherapy between high-dose (Mitoxantrone, 45 mg/m2, Cyclophosphamide 120 mg/kg, Melphalan 140 mg/m2), and the pursuit of the same conventionnal chemotherapy. Between 09/92 and 12/96, 61 chemosensitive patients were enrolled 29 were referred to standard chemotherapy, 32 to intensive therapy. At randomization, 13 pts (21.3%) were in complete response and 48 in partial response.
ResultsThe median progression-free survivals were 20 and 35.3 months in the standard and intensive groups (p=0.06). The relapse rates were respectively 79.3% vs 50.8% at 3 years and 90.8% vs 90.7% at 5 years. The median overall survivals were 20 and 43.4 months, with an overall survival rate of 18.5% vs 29.8% at 5 years (p=0.12).
Conclusion The CMA regimen could prolong the progression-free survival of MBC patients, however without any significant impact on overall survival.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Remerciements: Ligue Nationale Contre le Cancer, les laboratoires Amgen, Pharmacia-Upjohn et Lederle
Rights and permissions
About this article
Cite this article
Lotz, JP., Curé, H., Janvier, M. et al. Intensification thérapeutique et autogreffe de cellules souches hématopoïétiques (CSH) dans le traitement des cancers du sein métastatiques : résultats du programme national Pegase 04. Hematol Cell Ther 41, 71–74 (1999). https://doi.org/10.1007/s00282-999-0071-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00282-999-0071-1