Abstract
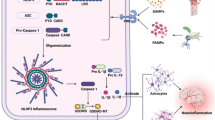
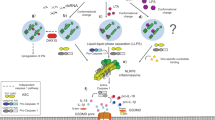
The recent identification of cytosolic pattern recognition receptors (PRRs) with leucine-rich repeats, which recognize pathogen-associated molecular patterns (PAMPs), has been garnering considerable attention. Activated PRRs form molecular complexes called inflammasomes, consisting of related proteins that include procaspase 1[interleukin (IL) 1β converting enzyme (ICE)]. Inflammasomes have been shown to facilitate molecular proximity, stimulate activation of procaspase 1, which consequently produces inflammatory cytokines IL-1β and IL-18 and ultimately lead to the initiation of innate immunity. An adaptor protein, apoptosis-associated speck-like protein containing a CARD (ASC), which recruits PRRs carrying the pyrin homologous domain (PYD) and procaspase 1 through PYD and CARD, respectively, is responsible for the formation of some inflammasomes and also activation of procaspase 1. In this review, our main attention will be directed to PYD region analysis of ASC to understand the interaction between PYD-carrying PRRs and ASC. Taking into consideration the other aspects of the ASC gene in the proapoptotic ability and down-regulation by methylation, the biological function of ASC will be discussed in relation to the epigenetic aspects of infection, inflammation, and cancer.


Similar content being viewed by others
References
Akira S, Takeda K (2004) Toll-like receptor signaling. Nat Rev Immunol 4:499–511
Inohara N, Chamaillard M, McDonald C, Nunez G (2005) NOD-LRR proteins: role in host-microbial interactions and inflammatory disease. Annu Rev Biochem 74:355–383
Ting JP, Kastner DL, Hoffman HM (2006) CATERPILLERs, pyrin and hereditary immunological disorders. Nat Rev Immunol 6:183–195
Philpott DJ, Girardin SE (2004) The role of Toll-like receptors and Nod proteins in bacterial infection. Mol Immunol 41:1099–1108
Strober W, Murray PJ, Kitani A, Watanabe T (2006) Signalling pathways and molecular interactions of NOD1 and NOD2. Nat Rev Immunol 6:9–20
Kufer TA, Fritz JH, Philpott DJ (2005) NACHT-LRR proteins (NLRs) in bacterial infection and immunity. Trends Microbiol 13:381–388
Viala J et al Nod1 responds to peptidoglycan delivered by the Helicobacter pylori cag pathogenicity island. Nature Immunol 5:1166–1174
Meylan E, Tschopp J, Karin M (2006) Intracellular pattern recognition receptors in the host response. Nature 442:39–44
Martinon F, Burns K, Tschopp J (2002) The nflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-1β. Mol Cell 10:417–426
Schmitz J. et al (2005) IL-33, an interleukin-1-like cytokine that signals via the IL-1 receptor-related protein ST2 and induces T helper type 2-associated cytokines. Immunity 23:479–490
Thornberry NA, Molineaux SM (1995) Interleukin-1β converting enzyme: a novel cysteine protease required for IL-1β production and implicated in programmed cell death. Protein Sci 4:3–12
Hoffman HM, Mueller JL, Broide DH, Wanderer AA, Kolodner RD (2001) Mutation of a new gene encoding a putative pyrin-like protein causes familial cold autoinflammatory syndrome and Muckle–Wells syndrome. Nat Genet 29:301–305
McDermott MF, Aksentijevich I (2002) The autoinflammatory syndromes. Curr Opin Allergy Clin Immunol 2:511–516
Feldmann J et al (2002) Chronic infantile neurological cutaneous and articular syndrome is caused by mutations in CIS1, a gene highly expressed in polymorphonuclear cells and chondrocytes. Am J Hum Genet 71:198–203
Martinon F, Tschopp J (2004) Inflammatory caspases: linking an intracellular innate immune system to autoinflammatory diseases. Cell 117:561–574
Solle M et al (2001) Altered cytokine production in mice lacking P2X7 receptors. J Biol Chem 276:125–132
Perregaux D, Gabel CA (1994) Interleukin-1β maturation and release in response to ATP and nigericin. Evidence that potassium depletion mediated by these agents is a necessary and common feature of their activity. J Biol Chem 269:15195–15203
Walev I, Reske K, Palmer M, Valeva A, Bhakdi S (1995) Potassium-inhibited processing of IL-1β in human monocytes. EMBO J 14:1607–1614
Gurcel L, Abrami L, Girardin S, Tschopp J, van der Goot FG (2006) Caspase-1 activation of lipid metabolic pathways in response to bacterial pore-forming toxins promotes cell survival. Cell 126:1135–1145
Andrei C et al (2004) Phospholipases C and A2 control lysosome-mediated IL-1β secretion: implications for inflammatory processes. Proc Natl Acad Sci USA 101:9745–9750
Masumoto J, Taniguchi S, Ayukawa K et al (1999) ASC, a novel 22-kDa protein, aggregates during apoptosis of human promyelocytic leukemia HL-60 cells. J Biol Chem 274(33):835–838
The International FMF Consortium (1997) Ancient missense mutations in a new member of the RoRet gene family are likely to cause familial Mediterranean fever. Cell 90:797–807
Masumoto J, Taniguchi S, Sagara J (2001) Pyrin N-terminal homology domain- and caspase recruitment domain-dependent oligomerization of ASC. Biochem Biophys Res Commun 280:652–655
Moriya M, Taniguchi S, Wu P, Liepinsh E, Otting G, Sagara J (2005) Role of charged and hydrophobic residues in the oligomerization of the PYRIN domain of ASC. Biochemistry 44:575–583
Masumoto J, Taniguchi S, Nakayama J et al (2001) Expression of apoptosis-associated speck-like protein containing a caspase recruitment domain, a pyrin N-terminal homology domain-containing protein, in normal human tissues. J Histochem Cytochem 49:1269–1275
Shiohara M, Taniguchi S, Masumoto J et al (2002) ASC, which is composed of a PYD and a CARD, is up-regulated by inflammation and apoptosis in human neutrophils. Biochem Biophys Res Commun 293:1314–1318
Yamamoto M, Yaginuma K, Tsutsui H et al (2004) ASC is essential for LPS-induced activation of procaspase-1 independently of TLR-associated signal adaptor molecules. Genes Cells 9:1055–1067
Mariathasan S, Newton K, Monack DM et al (2004) Differential activation of the inflammasome by caspase-1 adaptors ASC and Ipaf. Nature 430:213–218
Dinarello CA (1996) Biologic basis for interleukin-1 in disease. Blood 87:2095–2147
Annand RR et al (1999) Caspase-1 (interleukin-1β-converting enzyme) is inhibited by the human serpin analogue proteinase inhibitor 9. Biochem J 342:655–665
Saleh M et al (2006) Enhanced bacterial clearance and sepsis resistance in caspase-12-deficient mice. Nature 440:1064–1068
Razmara M et al (2002) CARD-8 protein, a new CARD family member that regulates caspase-1 activation and apoptosis. J Biol Chem 277:13952–13958
Aksentijevich I et al (1999) Mutation and haplotype studies of familial Mediterranean fever reveal new ancestral relationships and evidence for a high carrier frequency with reduced penetrance in the Ashkenazi Jewish population. Am J Hum Genet 64:949–962
Chae JJ et al (2003) Targeted disruption of pyrin, the FMF protein, causes heightened sensitivity to endotoxin and a defect in macrophage apoptosis. Mol Cell 11:591–604
Inohara N, Nunez G (2003) NODs: intracellular proteins involved in inflammation and apoptosis. Nat Rev Immunol 3(5):371–82 (Review, May)
Chae JJ et al (2006) The B30.2 domain of pyrin, the familial Mediterranean fever protein, interacts directly with caspase-1 to modulate IL-1β production. Proc Natl Acad Sci USA 103:9982–9987
Messud-Petit F et al (1998) Serp2, an inhibitor of the interleukin-1β -converting enzyme, is critical in the pathobiology of myxoma virus. J Virol 72:7830–7839
Johnston JB et al (2005) A poxvirus-encoded pyrin domain protein interacts with ASC-1 to inhibit host inflammatory and apoptotic responses to infection. Immunity 23:587–598
Bertin J, DiStefano PS (2000) The PYRIN domain: a novel motif found in apoptosis and inflammation proteins. Cell Death Differ 7:1273–1274
Fairbrother WJ, Gordon NC, Humke EW, O’Rourke KM, Starovasnik MA, Yin JP, Dixit VM (2001) The PYRIN domain: a member of the death domain-fold superfamily. Protein Sci 10:1911–1918
Pawlowski K, Pio F, Chu Z, Reed JC, Godzik A (2001) PAAD—a new protein domain associated with apoptosis, cancer and autoimmune diseases. Trends Biochem Sci 26:85–87
Staub, E., Dahl, E. and Rosenthal, A. (2001) The DAPIN family: a novel domain links apoptotic and interferon response proteins. Trends Biochem Sci 26:83–85
Xiao T, Towb P, Wasserman SA, Sprang SR (1999) Three-dimensional structure of a complex between the death domains of Pelle and Tube. Cell 99:545–555
Qin H, Srinivasula SM, Wu G, Fernandes-Alnemri T, Alnemri ES, Shi Y (1999) Structural basis of procaspase-9 recruitment by the apoptotic protease-activating factor 1. Nature 399:549–557
Weber CH, Vincenz C (2001) The death domain superfamily: a tale of two interfaces? Trends Biochem Sci 26:475–481
Liepinish E, Barbals R, Dahl E, Sharipo A, Staub E, Otting G (2003) The death-domain fold of the ASC PYRIN domain, presenting a basis for PYRIN/PYRIN recognition. J Mol Biol 332:1155–1163
Hiller S, Kohl A, Fiorito F, Herrmann T, Wider G, Tschopp J, Grütter MG, Wüthrich K (2003) NMR structure of the apoptosis- and inflammation-related NALP1 pyrin domain. Structure 11:1199–1205
Siegel RM, Martin DA, Zheng L, Ng S, Bertin J, Cohen J, Lenardo MJ (1998) Death-effector filaments: novel cytoplasmic structures that recruit caspases and trigger apoptosis. J Cell Biol 141:1243–1253
Perez D, White E (1998) E1B 19K inhibits Fas-mediated apoptosis through FADD-dependent sequestration of FLICE. J Cell Biol 141:1255–1266
Yan M, Lee J, Schilbach S, Godard A, Dexit V (1999) mE10, a novel caspase recruitment domain-containing proapoptotic molecule. J Biol Chem 274:10287–10292
Guiet C, Vito P (2000) Caspase recruitment domain (CARD)-dependent cytoplasmic filaments mediate Bcl 10-induced NF-κB activation. J Cell Biol 148:1131–1139
Hull KM, Shoham N, Chae JJ, Aksentijevich I, Kastner DL (2003) The expanding spectrum of systemic autoinflammatory disorders and their rheumatic manifestations. Curr Opin Rheumatol 15:61–69
Mariathasan S, Monack DM (2007) Inflammasome adaptors and sensors:intracellular regulatetors of infection and inflammation. Nat Immunol 70:31–40
Conway KE, McConnell BB, Bowring CE, Donald CD, Warren ST, Vertino PM (2000) TMS1, a novel proapoptotic caspase recruitment domain protein, is a target of methylation-induced gene silencing in human breast cancers. Cancer Res 60:6236–42
Guan X, Sagara J, Yokoyama T et al (2003) ASC/TMS1, a caspase-1 activating adaptor, is downregulated by aberrant methylation in human melanoma. Int J Cancer 107:202–208
Machida EO, Brock MV, Hooker CM, Nakayama J, Ishida A, Amano J, Picchi MA, Belinsky SA, Herman JG, Taniguchi S, Baylin SB (2006) Hypermethylation of ASC/TMS1 is a sputum marker for late-stage lung cancer. Cancer Res 66(12):6210–6218 (Jun 15)
Masumoto J, Dowds TA, Schaner P, Chen FF, Ogura Y, Li M et al (2003) ASC is an activating adaptor for NF-kappa B and caspase-8-dependent apoptosis. Biochem Biophys Res Commun 303:69–73
Ohtsuka T, Ryu H, Minamishima YA et al (2004) ASC is a Bax adaptor and regulates the p53-Bax mitochondrial apoptosis pathway. Nat Cell Biol 6:121–128
Hasegawa M, Kawase K, Inohara N, Imamura R, Yeh W-C, Kinoshita T, Suda T (2007) Mechanism of ASC-mediated apoptosis:Bid-dependent apoptosis in type II cells. Oncogene 26:1748–1756
Grenier JM et al (2002) Functional screening of five PYPAF family members identifies PYPAF5 as a novel regulator of NF-κB and caspase-1. FEBS Lett 530:73–78
Wang L et al (2002) PYPAF7, a novel PYRIN-containing Apaf1-like protein that regulates activation of NF-κB and caspase-1-dependent cytokine processing. J Biol Chem 277:29874–29880
Mariathasan S, Weiss DS, Dixit VM, Monack DM (2005) Innate immunity against Francisella tularensis is dependent on the ASC/caspase-1 axis. Exp Med 202:1043–1049
Gavrilin MA et al (2006) Internalization and phagosome escape required for Francisella to induce human monocyte IL-1β processing and release. Proc Natl Acad Sci USA 103:141–146
Zamboni DS et al (2006) The Birc1e cytosolic patternrecognition receptor contributes to the detection and control of Legionella pneumophila infection. Nature Immunol 7:318–325
Ren T, Zamboni DS, Roy CR, Dietrich WF, Vance RE (2006) Flagellin-deficient Legionella mutants evade caspase-1- and Naip5-mediated macrophage immunity. PLoS Pathog 2:e18
Molofsky AB et al (2006) Cytosolic recognition of flagellin by mouse macrophages restricts Legionella pneumophila infection. J Exp Med 203:1093–1104
Mariathasan S. et al (2006) Cryopyrin activates the inflammasome in response to toxins and ATP. Nature 440:228–232
Kanneganti TD et al (2006) Bacterial RNA and small antiviral compounds activate caspase-1 through cryopyrin/Nalp3. Nature 440:233–236
Martinon F, Petrilli V, Mayor A, Tardivel A, Tschopp J (2006) Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440:237–241
Acknowledgements
We thank our collaborators and all graduate students who have been involved in the study of ASC. This work was partially supported by a grant-in-aid from Novartis Pharma K.K.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Taniguchi, S., Sagara, J. Regulatory molecules involved in inflammasome formation with special reference to a key mediator protein, ASC. Semin Immunopathol 29, 231–238 (2007). https://doi.org/10.1007/s00281-007-0082-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00281-007-0082-3
Keywords
- Pattern recognition receptors (PRRs)
- Leucine-rich repeats (LRRs)
- Pathogen-associated molecular patterns (PAMPs)
- A PRR family of cytosolic NOD-like receptors (NLRs)
- Apoptosis-associated speck-like protein containing a CARD (ASC)
- Pyrin homologous domain (PYD)
- NACHT-, LRR- and PYD-containing proteins (NALPs)
- Caspase 1
- Inflammasome
- IL-1β
- IL-18
- IL-33
- Apoptosis