Abstract
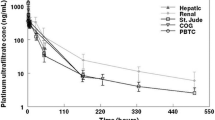
The inter- and intraindividual variabilities in topotecan clearance (CL) were explored using a population pharmacokinetic approach. Total (lactone + hydroxy acid) topotecan plasma concentrations were obtained in 31 women with metastatic epithelial ovarian cancer treated by the 30-min intravenous infusion on 5 subsequent days. The data corresponding to three occasions (days 1 and 5 of cycle 1, and day 1 of cycle 2), were analyzed using the nonlinear mixed effect model program. A large interindividual variability was observed, with CL varying from 9.1 to 42.5 l per hour (mean 21.0). Topotecan CL was related to serum creatinine level, and age. A close relationship was also observed between topotecan CL and creatinine clearance. Intraindividual variability both within cycle 1 and between the two first cycles was limited, with a mean variation of −2 ± 17%, and +5 ± 20%, respectively. A limited sampling strategy using Bayesian estimation based on two samples (5 min before the end of the 30-min infusion, and 4 h after the end of infusion) was developed. The results of this study combine relationships between topotecan pharmacokinetic parameters and patient covariates that may be useful for a priori dose adjustment, and convenient sampling procedure that can be used for further studies and drug monitoring.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 23 February 2000 / Accepted: 22 May 2000
Rights and permissions
About this article
Cite this article
Montazeri, A., Boucaud, M., Lokiec, F. et al. Population pharmacokinetics of topotecan: intraindividual variability in total drug. Cancer Chemother Pharmacol 46, 375–381 (2000). https://doi.org/10.1007/s002800000161
Issue Date:
DOI: https://doi.org/10.1007/s002800000161