Abstract
Purpose
Following a previously published pre-clinical validation, this phase I study evaluated the safety, maximum tolerated dose, anti-tumour activity and immune status of a gemcitabine–chloroquine combination as a first- or late-line treatment in patients with metastatic or unresectable pancreatic cancer.
Methods
In this 3 + 3 dose escalation study, patients received a single weekly standard dose of intravenous gemcitabine, followed by single weekly oral intake of 100, 200 or 300 mg of chloroquine. Tumour response was assessed using the Response Evaluation Criteria in Solid Tumors version 1.1. Immune status was evaluated by RT-PCR to measure the relative expression of immune-related genes in peripheral blood mononuclear cells (PBMCs).
Results
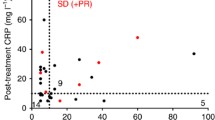
Overall, nine patients [median age 72 years; interquartile range (IQR), 68–78 years] were treated. No dose-limiting toxicities as defined in the protocol were observed. Three patients experienced partial response, and two patients had stable disease. The median time to progression was 4 months (95% CI 0.8–7.2), and the median overall survival was 7.6 months (95% CI 5.3–9.9). Among 86 assayed immune genes, three were significantly differentially expressed in PBMCs from responding versus non-responding patients: interferon-gamma receptor-1, toll-like receptor 2, and beta-2 microglobulin.
Conclusions
The addition of chloroquine to gemcitabine was well tolerated and showed promising effects on the clinical response to the anti-cancer chemotherapy. Based on these initial results, the efficacy of the gemcitabine–chloroquine combination should be further assessed.


Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A. Cancer Statistics (2017) CA Cancer J Clin 67(1):7–30
Burris HA 3rd, Moore MJ, Andersen J, Green MR, Rothenberg ML, Modiano MR et al (1997) Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol 15(6):2403–2413
Conroy T, Desseigne F, Ychou M, Bouche O, Guimbaud R, Becouarn Y et al (2011) FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 364(19):1817–1825
Von Hoff DD, Ervin T, Arena FP, Chiorean EG, Infante J, Moore M et al (2013) Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med 369(18):1691–1703
Schlitzer M (2007) Malaria chemotherapeutics part I: History of antimalarial drug development, currently used therapeutics, and drugs in clinical development. ChemMedChem 2(7):944–986
Lee SJ, Silverman E, Bargman JM (2011) The role of antimalarial agents in the treatment of SLE and lupus nephritis. Nat Rev Nephrol 7(12):718–729
Pascolo S (2016) Time to use a dose of Chloroquine as an adjuvant to anti-cancer chemotherapies. Eur J Pharmacol 771:139–144
Accapezzato D, Visco V, Francavilla V, Molette C, Donato T, Paroli M et al (2005) Chloroquine enhances human CD8 + T cell responses against soluble antigens in vivo. J Exp Med 202(6):817–828
Solomon VR, Lee H (2009) Chloroquine and its analogs: a new promise of an old drug for effective and safe cancer therapies. Eur J Pharmacol 625(1–3):220–233
Briceno E, Calderon A, Sotelo J (2007) Institutional experience with chloroquine as an adjuvant to the therapy for glioblastoma multiforme. Surg Neurol 67(4):388–391
Sotelo J, Briceno E, Lopez-Gonzalez MA (2006) Adding chloroquine to conventional treatment for glioblastoma multiforme: a randomized, double-blind, placebo-controlled trial. Ann Intern Med 144(5):337–343
Buch IN, Munz H, Pascolo C (2016) S. Schedule-dependent synergy of chloroquine with chemotherapy for anti-cancer treatment. Cancer Res Oncol 2(2):8
Zitvogel L, Apetoh L, Ghiringhelli F, Kroemer G (2008) Immunological aspects of cancer chemotherapy. Nat Rev Immunol 8(1):59–73
Jang CH, Choi JH, Byun MS, Jue DM (2006) Chloroquine inhibits production of TNF-alpha, IL-1beta and IL-6 from lipopolysaccharide-stimulated human monocytes/macrophages by different modes. Rheumatology 45(6):703–710
Thome R, Moraes AS, Bombeiro AL, Farias Ados S, Francelin C, da Costa TA et al (2013) Chloroquine treatment enhances regulatory T cells and reduces the severity of experimental autoimmune encephalomyelitis. PLoS One 8(6):e65913
Abiko K, Matsumura N, Hamanishi J, Horikawa N, Murakami R, Yamaguchi K et al (2015) IFN-gamma from lymphocytes induces PD-L1 expression and promotes progression of ovarian cancer. Br J Cancer 112(9):1501–1509
Alberts DS, Marth C, Alvarez RD, Johnson G, Bidzinski M, Kardatzke DR et al (2008) Randomized phase 3 trial of interferon gamma-1b plus standard carboplatin/paclitaxel versus carboplatin/paclitaxel alone for first-line treatment of advanced ovarian and primary peritoneal carcinomas: results from a prospectively designed analysis of progression-free survival. Gynecol Oncol 109(2):174–181
Yang HZ, Cui B, Liu HZ, Mi S, Yan J, Yan HM et al (2009) Blocking TLR2 activity attenuates pulmonary metastases of tumor. PLoS One 4(8):e6520
Acknowledgements
This work was supported by the Julius Müller Stiftung and the Kurt und Senta Hermann Stiftung.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by the University Hospital of Zürich, the Julius Müller Stiftung and the Kurt und Senta Hermann Stiftung.
Conflict of interest
Authors Samaras, Nguyen-Kim, Seifert, Bachmann, Knuth and Pascolo declare that they have no conflict of interest. Author von Moos is a participant in an advisory board of Elli Lilly.
Animals
This article does not contain any studies with animals performed by any of the authors.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the local Ethics Committee and Swissmedic (ClinicalTrials.gov identifier: NCT01777477) and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Samaras, P., Tusup, M., Nguyen-Kim, T.D.L. et al. Phase I study of a chloroquine–gemcitabine combination in patients with metastatic or unresectable pancreatic cancer. Cancer Chemother Pharmacol 80, 1005–1012 (2017). https://doi.org/10.1007/s00280-017-3446-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-017-3446-y