Abstract
Purpose
Standard therapy for advanced small bowel adenocarcinoma (SBA) has not yet been established. The present study assessed the efficacy and safety of chemotherapy (CT) in association with molecular targeting approaches for SBA.
Methods
The histories of 33 advanced SBA patients from six different institutions in Japan, who received CT from 2008 to 2016, were retrospectively examined for background, clinical course and outcome.
Results
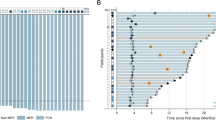
Median patient age was 65 years (range 39–83). Primary tumor was located in the duodenum in 21 patients (67%), the ampulla of Vater in three patients (9%), the jejunum in seven patients (21%) and the ileum in one patient (3%). Histologically, well-to-moderately and poorly differentiated adenocarcinoma were identified in 20 (61%) and nine (27%) patients, respectively. Thirteen patients received a single CT regimen, seven patients received two types of CT regimen, and 13 patients received three or more CT regimens. As first-line CT, modified FOLFOX6, capecitabine plus oxaliplatin, and S-1 plus cisplatin were employed in 13, 1, and 4 patients, respectively. The response rate (RR) and median progression-free survival (PFS) were 25% and 6.0 months, respectively. Median overall survival (OS) was 13.0 months. Nine out of the 33 patients received bevacizumab-containing CT and three received cetuximab-containing CT. Median OS of bevacizumab-containing CT patients was 21.9 months. No unexpected serious adverse events were observed.
Conclusions
The analysis indicates that combination CT for advanced SBA is associated with modest efficacy and safety, and bevacizumab-containing CT may contribute to favorable outcome in these patients.

Similar content being viewed by others
References
Radhav K, Overman MJ (2013) Small bowel adenocarcinoma-existing evidence and evolving paradigm. Nat Rev Clin Oncol 10:534–544
Bilimoria KY, Bentrem DJ, Wayne JD, Ko CY, Bennett CL, Talamonti MS (2009) Small bowel cancer in the United States: changes in epidemiology, treatment, and survival over the last 20 years. Ann Surg 249:63–71. doi:10.1097/SLA.0b013e31818e4641
Dabaja BS, Suki D, Pro B, Bonnen M, Ajani J (2004) Adenocarcinoma of the small bowel: presentation, prognostic factors, and outcome of 217 patients. Cancer 101:518–526
Talamonti MS, Goetz LH, Rao S, Joehl RJ (2002) Primary cancers of the small bowel: analysis of prognostic factors and results of surgical management. Arch Surg 137:564–570
Howe JR, Karnell LH, Menck HR, Scott-Conner C (1999) The American College of Surgeons Commission on Cancer and the American Cancer Society. Adenocarcinoma of the small bowel: review of the National Cancer Data Base, 1985–1995. Caner 86:2693–2706
Overman MJ, Varadhachary GR, Kopetz S, Adinin R, Lin E, Morris JS, Eng C, Abbruzzese JL, Wolff RA (2009) Phase II study of capecitabine and oxaliplatin for advanced adenocarcinoma of the small bowel and ampulla of Vater. J Clin Oncol 27:2598–2603. doi:10.1200/JCO.2008.19.7145
Xiang XJ, Liu YW, Zhang L, Qiu F, Yu F, Zhan ZY, Feng M, Yan J, Zhao JG, Xiong JP (2012) A phase II study of modified FOLFOX as first-line chemotherapy in advanced small bowel adenocarcinoma. Anticancer Drugs 23:561–566. doi:10.1097/CAD.0b013e328350dd0d
Zaanan A, Gauthier M, Malka D, Locher C, Gornet JM, Thirot-Bidault A, Tougeron D, Taïeb J, Bonnetain F, Aparicio T (2011) Second-line chemotherapy with fluorouracil, leucovorin, and irinotecan (FOLFIRI regimen) in patients with advanced small bowel adenocarcinoma after failure of first-line platinum-based chemotherapy: a multicentre AGEO study. Cancer 117:1422–1428. doi:10.1002/cncr.25614
Tsushima T, Taguri M, Honma Y et al (2012) Multicenter retrospective study of 132 patients with unresectable small bowel adenocarcinoma treated with chemotherapy. Oncologist 17:1163–1170
Zaanan A, Costes L, Gauthier M et al (2010) Chemotherapy of advanced small-bowel adenocarcinoma: a multicenter AGEO study. Ann Oncol 21:1786–1793. doi:10.1093/annonc/mdq038
Douillard JY, Siena S, Cassidy J et al (2010) Randomized, phase III trial of panitumumab with infusional fluorouracil, leucovorin, and oxaliplatin (FOLFOX4) versus FOLFOX4 alone as first-line treatment in patients with previously untreated metastatic colorectal cancer: the PRIME study. J Clin Oncol 28:4697–4705. doi:10.1200/JCO.2009.27.4860
Saltz LB, Clarke S, Díaz-Rubio E et al (2008) Bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: a randomized phase III study. J Clin Oncol 26:2013–2019. doi:10.1200/JCO.2007.14.9930
Bang YJ, Van Cutsem E, Feyereislova A, ToGA Trial Investigators et al (2010) Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomized controlled trial. Lancet 376:687–697. doi:10.1016/S0140-6736(10)61121-X
Wilke H, Muro K, Van Cutsem E, RAINBOW Study Group et al (2014) Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol 15:1224–1235. doi:10.1016/S1470-2045(14)70420-6
Aparicio T, Svrcek M, Zaanan A et al (2013) Small bowel adenocarcinoma phenotyping, a clinicobiological prognostic study. Br J Cancer 109:3057–3061. doi:10.1038/bjc.2013.677
Gulhati P, Raghav K, Shroff RT et al (2017) Bevacizumab combined with capecitabine and oxaliplatin in patients with advanced adenocarcinoma of the small bowel or ampulla of Vater: a single-center, open-label, phase 2 study. Cancer 123:1011–1017
Aydin D, Sendur MA, Kefeli U et al (2016) Evaluation of bevacizumab in advanced small bowel adenocarcinoma. Clin Corolectal Cancer. doi:10.1016/j.clcc.2016.04.013
Santini D, Fratto ME, Spoto C, Russo A, Galluzzo S, Zoccoli A, Vincenzi B, Tonini G (2010) Cetuximab in small bowel adenocarcinoma: a new friend? Br J Cancer 103:1305. doi:10.1038/sj.bjc.6605898
Eisenhauer EA, Therasse P, Bogaerts J et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45:228–247. doi:10.1016/j.ejca.2008.10.026
National Cancer Institute: Common Terminology Criteria for Adverse Events (CTCAE), version 4.0. http://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference_5x7.pdf. Accessed 10 Feb 2017
Cassidy J, Clarke S, Díaz-Rubio E et al (2008) Randomized phase III study of capecitabine plus oxaliplatin compared with fluorouracil/folinic acid plus oxaliplatin as first-line therapy for metastatic colorectal cancer. J Clin Oncol 26:2006–2012
Halfdanarson TR, McWilliams RR, Donohue JH, Quevedo JF (2010) A single-institution experience with 491 cases of small bowel adenocarcinoma. Am J Surg 199:797–803. doi:10.1016/j.amjsurg.2009.05.037
Nicholl MB, Ahuja V, Conway WC, Vu VD, Sim MS, Singh G (2010) Small bowel adenocarcinoma: understaged and undertreated? Ann Surg Oncol 17:2728–2732. doi:10.1245/s10434-010-1109-x
Overman MJ, Hu CY, Wolff RA, Chang GJ (2010) Prognostic value of lymph node evaluation in small bowel adenocarcinoma: analysis of the surveillance, epidemiology, and end results database. Cancer 116:5374–5382. doi:10.1002/cncr.25324
Kim IJ, Park JH, Kang HC, Shin Y, Park HW, Park HR, Ku JL, Lim SB, Park JG (2003) Mutational analysis of BRAF and K-ras in gastric cancers: absence of BRAF mutations in gastric cancers. Hum Genet 114:118–120
Moehler M, Mueller A, Trarbach T, Lordick F, Seufferlein T, Kubicka S, Geissler M, Schwarz S, Galle PR (2011) Cetuximab with irinotecan folinic acid and 5-fluorouracil as first-line treatment in advanced gastroesophageal cancer: a prospective multi-center biomarker-oriented phase II study. Ann Oncol 22:1358–1366. doi:10.1093/annonc/mdq591
Lordick F, Luber B, Lorenzen S et al (2010) Cetuximab plus oxaliplatin/leucovorin/5-fluorouracil in first-line metastatic gastric cancer: a phase II study of the Arbeitsgemeinschaft Internistische Onkologie (AIO). Br J Cancer 102:500–505. doi:10.1038/sj.bjc.6605521
Koornstra JJ, Kleibeuker JH, Vasen HF (2008) Small-bowel cancer in Lynch syndrome: is it time for surveillance? Lancet Oncol 9:901–905. doi:10.1016/S1470-2045(08)70232-8
Watson P, Vasen HF, Mecklin JP, Bernstein I, Aarnio M, Järvinen HJ, Myrhøj T, Sunde L, Wijnen JT, Lynch HT (2008) The risk of extra-colonic, extra-endometrial cancer in the Lynch syndrome. Int J Cancer 123:444–449. doi:10.1002/ijc.23508
Vasen HF, Watson P, Mecklin JP, Lynch HT (1999) New clinical criteria for hereditary nonpolyposis colorectal cancer (HNPCC, Lynch syndrome) proposed by the International Collaborative Group on HNPCC. Gastroenterology 116:1453–1456
Ohtsu A, Shah MA, Van Cutsem E et al (2011) Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a randomized, double-blind, placebo-controlled phase III study. J Clin Oncol 29:3968–3976. doi:10.1200/JCO.2011.36.2236
Sartore-Bianchi A, Trusolino L, Martino C et al (2016) Dual-targeted therapy with trastuzumab and lapatinib in treatment-refractory, KRAS codon 12/13 wild-type, HER2-positive metastatic colorectal cancer (HERACLES): a proof-of-concept, multicentre, open-label, phase 2 trial. Lancet Oncol 17:738–746. doi:10.1016/S1470-2045(16)00150-9
Nagaraj G, Zarbalian Y, Flora K, Tan BR Jr (2014) Complete response and prolonged disease-free survival in a patient with recurrent duodenal adenocarcinoma treated with bevacizumab plus FOLFOX6. J Gastrointest Oncol 5:E1–E6. doi:10.3978/j.issn.2078-6891.2013.038
Okubo K, Yoshioka S, Asukai K et al (2010) A case report of primary adenocarcinoma of small intestine. Gan To Kagaku Ryoho 37:2792–2794
Overman MJ, Pozadzides J, Kopetz S, Wen S, Abbruzzese JL, Wolff RA, Wang H (2010) Immunophenotype and molecular characterisation of adenocarcinoma of the small intestine. Br J Cancer 102:144–150. doi:10.1038/sj.bjc.6605449
Hapani S, Chu D, Wu S (2009) Risk of gastrointestinal perforation in patients with cancer treated with bevacizumab: a meta-analysis. Lancet Oncol 10:559–568. doi:10.1016/S1470-2045(09)70112-3
Kozloff M, Yood MU, Berlin J, Flynn PJ, Kabbinavar FF, Purdie DM, Ashby MA, Dong W, Sugrue MM, Grothey A (2009) Investigators of the BRiTE study. Clinical outcomes associated with bevacizumab-containing treatment of metastatic colorectal cancer: the BRiTE observational cohort study. Oncologist 14:862–870. doi:10.1634/theoncologist.2009-0071
Garcia AA, Hirte H, Fleming G et al (2008) Phase II clinical trial of bevacizumab and low-dose metronomic oral cyclophosphamide in recurrent ovarian cancer: a trial of the California, Chicago, and Princess Margaret Hospital phase II consortia. J Clin Oncol 26:76–82. doi:10.1200/JCO.2007.12.1939
Acknowledgements
The authors thank the patients and their families for participating in this study, and the medical staff for their respective contributions to the treatment of patients.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Baba E. has received research Grants from Chugai and Merck Serono. Akashi K. has received research Grants from Chugai, Yakult and Merck Serono and an honorarium from Chugai. Esaki T. has received a research Grant from Merck Serono and honoraria from Chugai and Merck Serono.
Ethical approval
The study was performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. The study was approved by the ethics committee of each participating institution. For this type of study formal consent is not required.
Rights and permissions
About this article
Cite this article
Takayoshi, K., Kusaba, H., Uenomachi, M. et al. Suggestion of added value by bevacizumab to chemotherapy in patients with unresectable or recurrent small bowel cancer. Cancer Chemother Pharmacol 80, 333–342 (2017). https://doi.org/10.1007/s00280-017-3371-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-017-3371-0