Abstract
Purpose
Normal white blood cell counts (WBC) are unknown in children with acute lymphoblastic leukemia (ALL). Accordingly, 6-mercaptopurine (6MP) and methotrexate (MTX) maintenance therapy is adjusted by a common WBC target of 1.5–3.0 × 109/L. Consequently, the absolute degree of myelosuppression is unknown for the individual child and we wanted to evaluate this.
Methods
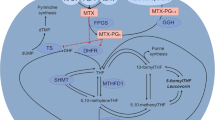
A median of 22 (range 8–27) 6MP/MTX metabolite samples and 100 (range 25–130) blood counts during therapy and 10 (range 2–15) off therapy were collected in 50 children with ALL. Differences between off-therapy and on-therapy WBCs [including absolute neutrophil (ANC) and lymphocyte counts (ALC)] were used to retrospectively approximate the absolute myelosuppression (=“delta-”) and association with age, sex and 6MP/MTX doses explored. We applied linear mixed models to estimate on-therapy counts by 6MP/MTX metabolites: DNA-incorporated thioguanine nucleotides (DNA-TGN), erythrocyte thioguanine nucleotides (ery-TGN), erythrocyte-methylated 6MP metabolites (ery-MeMP) and erythrocyte MTX polyglutamates with 2–6 glutamate residues (ery-MTXpg2–6).
Results
On-therapy WBC was correlated with ANC and ALC (r s = 0.84 and r s = 0.33, p values <0.001), whereas ANC was weakly correlated with ALC (r s = −0.11, p < 0.001), and neither significantly correlated with age. Off-therapy ALC, but not ANC, was strongly correlated with age (r s = −0.68 and −0.18, p < 0.001 and p = 0.22). Delta-ALC decreased with increasing age (r s = −0.69, p < 0.001). Incorporation of DNA-TGN was positively associated with ery-TGN (p < 0.001), ery-MeMP (p < 0.001) and ery-MTXpg2–6 (p = 0.047). On-therapy ALC decreased with increasing DNA-TGN level (p < 0.001, model adjusted for off-therapy ALC), whereas on-therapy ANC could not be modeled reliably.
Conclusion
Measurements of 6MP/MTX metabolites could supplement blood counts in assessing therapy intensity, but require prospective validation.




Similar content being viewed by others
References
Van Eys J, Berry D, Crist W et al (1989) Treatment intensity and outcome for children with acute lymphocytic leukemia of standard risk: A Pediatric Oncology group study. Cancer 63:1466–1471
Schmiegelow K, Bjork O, Glomstein A et al (2003) Intensification of mercaptopurine/methotrexate maintenance chemotherapy may increase the risk of relapse for some children with acute lymphoblastic leukemia. J Clin Oncol 21:1332–1339. doi:10.1200/JCO.2003.04.039
Relling MV, Hancock ML, Boyett JM et al (1999) Prognostic importance of 6-mercaptopurine dose intensity in acute lymphoblastic leukemia. Blood 93:2817–2823
Schmiegelow K, Nielsen SN, Frandsen TL, Nersting J (2014) Mercaptopurine/methotrexate maintenance therapy of childhood acute lymphoblastic leukemia: clinical facts and fiction. J Pediatr Hematol Oncol 00:1–15. doi:10.1097/MPH.0000000000000206
Aricó M, Baruchel A, Bertrand Y et al (2005) The seventh international childhood acute lymphoblastic leukemia workshop report: Palermo, Italy, January 29–30, 2005. Leukemia 19:1145–1152. doi:10.1038/sj.leu.2403783
Schmiegelow K, Pulczynska MK, Seip M (1988) White cell count during maintenance chemotherapy for standard-risk childhood acute lymphoblastic leukemia: relation to relapse rate. Pediatr Hematol Oncol 5:259–267. doi:10.3109/08880018809037365
Dolan G, Lilleyman JS, Richards SM (1989) Prognostic importance of myelosuppression during maintenance treatment of lymphoblastic leukaemia. Leukaemia in Childhood Working Party of the Medical Research Council. Arch Dis Child 64:1231–1234
Chessells JM, Harrison G, Lilleyman JS et al (1997) Continuing (maintenance) therapy in lymphoblastic leukaemia: lessons from MRC UKALL X. Medical Research Council Working Party in Childhood Leukaemia. Br J Haematol 98:945–951
Schmiegelow K, Nersting J, Nielsen SN et al (2016) Maintenance therapy of childhood acute lymphoblastic leukemia revisited—should drug doses be adjusted by white blood cell, neutrophil, or lymphocyte counts? Pediatr Blood Cancer. doi:10.1002/pbc.26139
Aldrimer M, Ridefelt P, Rödöö P et al (2013) Population-based pediatric reference intervals for hematology, iron and transferrin. Scand J Clin Lab Invest 73:253–261. doi:10.3109/00365513.2013.769625
Radtke S, Zolk O, Renner B et al (2013) Germline genetic variations in methotrexate candidate genes are associated with pharmacokinetics, toxicity, and outcome in childhood acute lymphoblastic leukemia. Blood 121:5145–5153. doi:10.1182/blood-2013-01-480335
Schrøder H, Fogh K (1988) Methotrexate and its polyglutamate derivatives in erythrocytes during and after weekly low-dose oral methotrexate therapy of children with acute lymphoblastic leukemia. Cancer Chemother Pharmacol 21:145–149
Lafolie P, Hayder S, Bj O, Peterson C (1991) Intraindividual variation in 6-mercaptopurine pharmacokinetics during oral maintenance therapy of children with acute lymphoblastic leukaemia. Eur J Clin Pharmacol 40:599–601
Lennard L (1992) The clinical pharmacology of 6-mercaptopurine. Eur J Clin Pharmacol 43:329–339
Karran P, Attard N (2008) Thiopurines in current medical practice: molecular mechanisms and contributions to therapy-related cancer. Nat Rev Cancer 8:24–36. doi:10.1038/nrc2292
Hedeland RL, Hvidt K, Nersting J et al (2010) DNA incorporation of 6-thioguanine nucleotides during maintenance therapy of childhood acute lymphoblastic leukaemia and non-Hodgkin lymphoma. Cancer Chemother Pharmacol 66:485–491. doi:10.1007/s00280-009-1184-5
Ebbesen MS, Nersting J, Jacobsen JH et al (2013) Incorporation of 6-thioguanine nucleotides into DNA during maintenance therapy of childhood acute lymphoblastic leukemia—the influence of thiopurine methyltransferase genotypes. J Clin Pharmacol 53:670–674. doi:10.1002/jcph.81
Vang SI, Schmiegelow K, Frandsen T et al (2015) Mercaptopurine metabolite levels are predictors of bone marrow toxicity following high-dose methotrexate therapy of childhood acute lymphoblastic leukaemia. Cancer Chemother Pharmacol 75:1089–1093. doi:10.1007/s00280-015-2717-8
Toft N, Birgens H, Abrahamsson J et al (2013) Risk group assignment differs for children and adults 1–45 yr with acute lymphoblastic leukemia treated by the NOPHO ALL-2008 protocol. Eur J Haematol 90:404–412. doi:10.1111/ejh.12097
Frandsen TL, Heyman M, Abrahamsson J et al (2014) Complying with the European Clinical Trials directive while surviving the administrative pressure—an alternative approach to toxicity registration in a cancer trial. Eur J Cancer 50:251–259. doi:10.1016/j.ejca.2013.09.027
Jacobsen JH, Schmiegelow K, Nersting J (2012) Liquid chromatography-tandem mass spectrometry quantification of 6-thioguanine in DNA using endogenous guanine as internal standard. J Chromatogr B Analyt Technol Biomed Life Sci 881–882:115–118. doi:10.1016/j.jchromb.2011.11.032
Shipkova M, Armstrong VW, Wieland E, Oellerich M (2003) Differences in nucleotide hydrolysis contribute to the differences between erythrocyte 6-thioguanine nucleotide concentrations determined by two widely used methods. Clin Chem 49:260–268. doi:10.1373/49.2.260
Chabner BA, Allegra CJ, Curt GA et al (1985) Polyglutamation of methotrexate. Is methotrexate a prodrug? J Clin Invest 76:907–912. doi:10.1172/JCI112088
Den Boer E, Meesters RJW, Van Zelst BD et al (2013) Measuring methotrexate polyglutamates in red blood cells: a new LC-MS/MS-based method. Anal Bioanal Chem 405:1673–1681. doi:10.1007/s00216-012-6581-7
Weinshilboum R (2001) Thiopurine pharmacogenetics: clinical and molecular studies of thiopurine methyltransferase. Drug Metab Dispos 29:601–605
Karim H, Ghalali A, Lafolie P et al (2013) Differential role of thiopurine methyltransferase in the cytotoxic effects of 6-mercaptopurine and 6-thioguanine on human leukemia cells. Biochem Biophys Res Commun 437:280–286. doi:10.1016/j.bbrc.2013.06.067
Korell J, Duffull SB, Dalrymple JM et al (2014) Comparison of intracellular methotrexate kinetics in red blood cells with the kinetics in other cell types. Br J Clin Pharmacol 77:493–497. doi:10.1111/bcp.12209
Karran P (2006) Thiopurines, DNA damage, DNA repair and therapy-related cancer. Br Med Bull 79–80:153–170. doi:10.1093/bmb/ldl020
Kolaczkowska E, Kubes P (2013) Neutrophil recruitment and function in health and inflammation. Nat Rev Immunol 13:159–175. doi:10.1038/nri3399
Sprent J (1993) Lifespans of naive, memory and effector lymphocytes. Curr Opin Immunol 5:433–438
Jacobsen JH (2012) Quantification of DNA-thioguanine in children with acute lymphoblastic leukemia. Thesis, University of Copenhagen
Möricke A, Zimmermann M, Reiter A et al (2010) Long-term results of five consecutive trials in childhood acute lymphoblastic leukemia performed by the ALL-BFM study group from 1981 to 2000. Leukemia 24:265–284. doi:10.1038/leu.2009.257
Toyoda Y, Manabe A, Tsuchida M et al (2000) Six months of maintenance chemotherapy after intensified treatment for acute lymphoblastic leukemia of childhood. J Clin Oncol 18:1508–1516
Richards S, Gray R, Peto R et al (1996) Duration and intensity of maintenance chemotherapy in acute lymphoblastic leukaemia: Overview of 42 trials involving 12,000 randomised children. Lancet 347:1783–1788. doi:10.1016/S0140-6736(96)91615-3
Eyrich M, Wiegering V, Lim A et al (2009) Immune function in children under chemotherapy for standard risk acute lymphoblastic leukaemia—a prospective study of 20 paediatric patients. Br J Haematol 147:360–370. doi:10.1111/j.1365-2141.2009.07862.x
El-Chennawi FA, Al-Tonbary YA, Mossad YM, Ahmed MA (2008) Immune reconstitution during maintenance therapy in children with acute lymphoblastic leukemia, relation to co-existing infection. Hematology 13:203–209. doi:10.1179/102453308X316086
O’Connor D, Bate J, Wade R et al (2014) Infection-related mortality in children with acute lymphoblastic leukemia: an analysis of infectious deaths on UKALL2003. Blood 124:1056–1061. doi:10.1182/blood-2014-03-560847
Gupta S, Antillon FA, Bonilla M et al (2011) Treatment-related mortality in children with acute lymphoblastic leukemia in Central America. Cancer 117:4788–4795. doi:10.1002/cncr.26107
Adam de Beaumais T, Fakhoury M, Medard Y et al (2011) Determinants of mercaptopurine toxicity in paediatric acute lymphoblastic leukemia maintenance therapy. Br J Clin Pharmacol 71:575–584. doi:10.1111/j.1365-2125.2010.03867.x
Rousseau A, Marquet P (2002) Application of pharmacokinetic modelling to the routine therapeutic drug monitoring of anticancer drugs. Fundam Clin Pharmacol 16:253–262. doi:10.1046/j.1472-8206.2002.00086.x
Nersting J, Schmiegelow K (2009) Pharmacogenomics of methotrexate: moving towards individualized therapy. Pharmacogenomics 10:1887–1889. doi:10.2217/pgs.09.148
Acknowledgments
The authors thank the staff at the laboratory of Pediatric Oncology, Bonkolab, Copenhagen, for their dedicated work.
Funding
This study received financial support from The Danish Cancer Society (R56-A3093-12-S2), The Danish Childhood Cancer Foundation and The Nordic Cancer Union (79400).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflicts of interest.
Ethical approval
Both oral and written informed consent to participate in the NOPHO ALL-2008 maintenance study were provided by both the parents (and when appropriate by the patient) according to the ICH/GCP guidelines and the Helsinki II Declaration. Further, all procedures performed involving human participants were in accordance with the ethical standards of the Danish Regional Ethical Committee (Study Approval: H-2-2010-002) and the Helsinki II declaration.
Electronic supplementary material
Below is the link to the electronic supplementary material.
280_2016_3151_MOESM2_ESM.pdf
Off-therapy hematology after termination of 6-mercaptopurine and methotrexate maintenance therapy of 50 children with acute lypmhoblastic leukemia. WBC: white blood cell count. ANC: absolute neutrophil count. ALC: absolute lymphocyte count (PDF 29 kb)
280_2016_3151_MOESM3_ESM.pdf
The mean weighted on-therapy leukocyte subsets of children treated for acute lymphoblastic leukemia in relation to the mean weighted off-therapy counts, one data point represents one child: a. The on-therapy absolute neutrophil count (ANC) plotted against the off-therapy ANC. b. The on-therapy absolute lymphocyte count (ALC) plotted against the off-therapy ALC (PDF 6 kb)
280_2016_3151_MOESM4_ESM.pdf
Early and late mean white blood cell counts (WBCs) after completion of maintenance therapy of childhood acute lymphoblastic leukemia. The off-therapy WBC measured day 200 to 360 after termination of therapy is plotted against the off-therapy WBC measured day 30 to 360 after termination of therapy. Each data point represents one child (PDF 5 kb)
Rights and permissions
About this article
Cite this article
Nielsen, S.N., Grell, K., Nersting, J. et al. Measures of 6-mercaptopurine and methotrexate maintenance therapy intensity in childhood acute lymphoblastic leukemia. Cancer Chemother Pharmacol 78, 983–994 (2016). https://doi.org/10.1007/s00280-016-3151-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-016-3151-2