Abstract
Purpose
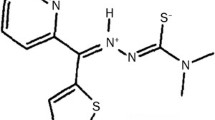
The interaction of p53 with its negative regulators Mdm2/4 has been widely studied (Khoury and Domling in Curr Pharm Des 18(30):4668–4678, 2012). In p53+/+ cells, expression of Mdm2/4 leads to p53 turnover, inhibition of downstream transcription, decreasing cell cycle arrest, or apoptosis. We report in vitro cytotoxicity and in vivo efficacy, pharmacokinetics, and metabolism of YH264, YH263, and WW751, three proposed small molecule inhibitors of the Mdm2/4–p53 interaction.
Methods
MTT cytotoxicity assays were performed, and alterations in proteins were examined using western blots. Mice were dosed 150 mg/kg YH264 or YH263 IV or PO QDx5. Mice were IV dosed 88, 57, or 39 mg/kg WW751 for 3, 5, or 5 days. YH264, YH263, and WW751 and metabolites were quantitated by LC–MS/MS.
Results
IC50 values for YH264, YH263, and WW751 against p53 wild-type HCT 116 cells after 72 h of incubation were 18.3 ± 2.3, 8.9 ± 0.6, and 3.1 ± 0.2 μM, respectively. Only YH264 appeared to affect p53 expression in vitro. None of the compounds affected the growth of HCT 116 xenografts in C.B-17 SCID mice. YH264 plasma half-life was 147 min; YH263 plasma half-life was 263 min; and WW751 plasma half-life was less than 120 min.
Conclusions
Despite dosing the mice at the maximum soluble doses, we could not achieve tumor concentrations equivalent to the intracellular concentrations required to inhibit cell growth in vitro. YH263 and WW751 do not appear to affect p53/Mdm2, and none of the three were active in a subcutaneous HCT 116 p53+/+ xenograft model.





Similar content being viewed by others
Abbreviations
- PBS:
-
Phosphate-buffered saline
- LC–MS/MS:
-
Liquid chromatography/tandem mass spectrometry
- MTT:
-
Methylthiazolyldiphenyl-tetrazolium bromide
- AUC:
-
Area under the drug concentration versus time curve
- C max :
-
Maximum peak concentration
References
Khoury K, Domling A (2012) P53 Mdm2 inhibitors. Curr Pharm Des 18(30):4668–4678
Teodoro JG, Evans SK, Green MR (2007) Inhibition of tumor angiogenesis by p53: a new role for the guardian of the genome. J Mol Med 85(11):1175–1186. doi:10.1007/s00109-007-0221-2
Momand J, Jung D, Wilczynski S, Niland J (1998) The MDM2 gene amplification database. Nucleic Acids Res 26(15):3453–3459
Harris SL, Levine AJ (2005) The p53 pathway: positive and negative feedback loops. Oncogene 24(17):2899–2908. doi:10.1038/sj.onc.1208615
Hunziker A, Jensen MH, Krishna S (2010) Stress-specific response of the p53–Mdm2 feedback loop. BMC Syst Biol 4:94. doi:10.1186/1752-0509-4-94
Zhuo YB, Bernard D, Wang S (2013) Small molecule inhibitors of MDM2–p53 and MDMX = p53 interactions as new cancer therapeutics. BioDiscov 8:1–14
Huang Y (2011) Discovery of small molecule inhibitors of protein–protein interactions (Doctoral Dissertation). University of Pittsburgh, Pittsburgh, PA
Popowicz GM, Czarna A, Wolf S, Wang K, Wang W, Domling A et al (2010) Structures of low molecular weight inhibitors bound to MDMX and MDM2 reveal new approaches for p53–MDMX/MDM2 antagonist drug discovery. Cell Cycle 9(6):1104–1111
Popowicz GM, Domling A, Holak TA (2011) The structure-based design of Mdm2/Mdmx–p53 inhibitors gets serious. Angew Chem 50(12):2680–2688. doi:10.1002/anie.201003863
Bunz F, Dutriaux A, Lengauer C, Waldman T, Zhou S, Brown JP et al (1998) Requirement for p53 and p21 to sustain G2 arrest after DNA damage. Science 282(5393):1497–1501
Domling AH, Inventor TA (2011) NOVEL P53–MDM2/P53–MDM4 antagonists to treat proliferative disease. http://www.google.com.ar/patents/WO2011106650A2?cl=ja
Wei N, Chu E, Wipf P, Schmitz JC (2014) Protein kinase d as a potential chemotherapeutic target for colorectal cancer. Mol Cancer Ther 13(5):1130–1141. doi:10.1158/1535-7163.MCT-13-0880
Bruce RD (1985) An up-and-down procedure for acute toxicity testing. Fundam Appl Toxicol: Off J Soc Toxicol 5(1):151–157
D’Argenio DZ, Schumitzky A, Wang X (2009) ADAPT 5 user’s guide: pharmacokinetic/pharmacodynamic systems analysis software. Biomedical Simulations Resource, Los Angeles
Ding Q, Zhang Z, Liu JJ, Jiang N, Zhang J, Ross TM et al (2013) Discovery of RG7388, a potent and selective p53–MDM2 inhibitor in clinical development. J Med Chem 56(14):5979–5983. doi:10.1021/jm400487c
Koes D, Khoury K, Huang Y, Wang W, Bista M, Popowicz GM et al (2012) Enabling large-scale design, synthesis and validation of small molecule protein–protein antagonists. PLoS ONE 7(3):e32839. doi:10.1371/journal.pone.0032839
Shangary S, Ding K, Qiu S, Nikolovska-Coleska Z, Bauer JA, Liu M et al (2008) Reactivation of p53 by a specific MDM2 antagonist (MI-43) leads to p21-mediated cell cycle arrest and selective cell death in colon cancer. Mol Cancer Ther 7(6):1533–1542. doi:10.1158/1535-7163.MCT-08-0140
Ravizza R, Gariboldi MB, Passarelli L, Monti E (2004) Role of the p53/p21 system in the response of human colon carcinoma cells to Doxorubicin. BMC Cancer 4:92. doi:10.1186/1471-2407-4-92
Ju J, Schmitz JC, Song B, Kudo K, Chu E (2007) Regulation of p53 expression in response to 5-fluorouracil in human cancer RKO cells. Clin Cancer Res: Off J Am Assoc Cancer Res 13(14):4245–4251. doi:10.1158/1078-0432.CCR-06-2890
Mahyar-Roemer M, Roemer K (2001) p21 Waf1/Cip1 can protect human colon carcinoma cells against p53-dependent and p53-independent apoptosis induced by natural chemopreventive and therapeutic agents. Oncogene 20(26):3387–3398. doi:10.1038/sj.onc.1204440
Meijer A, Kruyt FA, van der Zee AG, Hollema H, Le P, ten Hoor KA et al (2013) Nutlin-3 preferentially sensitises wild-type p53-expressing cancer cells to DR5-selective TRAIL over rhTRAIL. Br J Cancer 109(10):2685–2695. doi:10.1038/bjc.2013.636
Acknowledgments
This project used the UPCI Clinical Pharmacology Analytical Facility (CPAF) and was supported in part by Award P30CA047904. We would like to acknowledge the support from the University of Pittsburgh, Department of Pharmaceutical Sciences, School of Pharmacy. We would also like to acknowledge the excellent animal care provided by the Hillman Cancer Center DLAR.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Christner, S.M., Clausen, D.M., Beumer, J.H. et al. In vitro cytotoxicity and in vivo efficacy, pharmacokinetics, and metabolism of pyrazole-based small molecule inhibitors of Mdm2/4–p53 interaction. Cancer Chemother Pharmacol 76, 287–299 (2015). https://doi.org/10.1007/s00280-015-2791-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-015-2791-y