Abstract
Purpose
Thalidomide, originally developed as a sedative, was subsequently identified to have antiangiogenic properties. Lenalidomide is an antiangiogenic and immunomodulatory agent that has been utilized in the treatment of patients with brain tumors. We studied the pharmacokinetics and cerebrospinal fluid (CSF) penetration of thalidomide and lenalidomide in a nonhuman primate model.
Methods
A dose of 50 mg of thalidomide or 20 mg of lenalidomide was administered once orally to each of three rhesus monkeys. Plasma and CSF samples were obtained at specified intervals, and the thalidomide or lenalidomide concentrations were determined by high-performance liquid chromatography with tandem mass spectrometry. Pharmacokinetic parameters were estimated using noncompartmental methods. CSF penetration was calculated as area under the concentration–time curve (AUC) CSF/AUC plasma.
Results
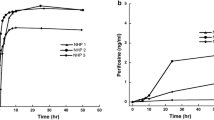
For thalidomide, the median apparent clearance (Cl/F) was 2.9 mL/min/kg, the median plasma AUC was 80 μM h, and the median terminal half-life (t½) was 13.3 h. For lenalidomide, the median Cl/F was 8.7 mL/min/kg, the median AUC was 9 μM h, and the median t½ was 5.6 h. Thalidomide was detected in the CSF of all animals, with a median penetration of 42%. Lenalidomide was detected in the CSF of 2 of 3 animals, with a CSF penetration of 11% in each.
Conclusion
Thalidomide and lenalidomide penetrate into the CSF after oral administration of clinically relevant doses. Plasma exposure to lenalidomide was similar in our model to that observed in studies involving children who have brain tumors. These results support further development of lenalidomide for the treatment of central nervous system malignancies.


Similar content being viewed by others
References
Arora RS, Alston RD, Eden TO, Estlin EJ, Moran A, Birch JM (2009) Age-incidence patterns of primary CNS tumors in children, adolescents, and adults in England. Neuro Oncol 11(4):403–413
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, Thun MJ (2008) Cancer statistics, 2008. CA Cancer J Clin 58(2):71–96
Wisoff JH, Boyett JM, Berger MS, Brant C, Li H, Yates AJ, McGuire-Cullen P, Turski PA, Sutton LN, Allen JC, Packer RJ, Finlay JL (1998) Current neurosurgical management and the impact of the extent of resection in the treatment of malignant gliomas of childhood: a report of the Children’s Cancer Group trial no. CCG-945. J Neurosurg 89(1):52–59
Freeman CR, Kepner J, Kun LE, Sanford RA, Kadota R, Mandell L, Friedman H (2000) A detrimental effect of a combined chemotherapy-radiotherapy approach in children with diffuse intrinsic brain stem gliomas? Int J Radiat Oncol Biol Phys 47(3):561–564
Nightingale SL (1998) From the Food and Drug Administration. JAMA 280(10):872
List AF (2007) Lenalidomide–the phoenix rises. New Engl J Med 357(21):2183–2186
Melchert M, List A (2007) The thalidomide saga. Int J Biochem Cell Biol 39(7–8):1489–1499
Palumbo A, Dimopoulos M, San Miguel J, Harousseau JL, Attal M, Hussein M, Knop S, Ludwig H, von Lilienfeld-Toal M, Sonneveld P (2009) Lenalidomide in combination with dexamethasone for the treatment of relapsed or refractory multiple myeloma. Blood Rev 23(2):87–93
Corral LG, Haslett PA, Muller GW, Chen R, Wong LM, Ocampo CJ, Patterson RT, Stirling DI, Kaplan G (1999) Differential cytokine modulation and T cell activation by two distinct classes of thalidomide analogues that are potent inhibitors of TNF-alpha. J Immunol 163(1):380–386
Haslett PA, Corral LG, Albert M, Kaplan G (1998) Thalidomide costimulates primary human T lymphocytes, preferentially inducing proliferation, cytokine production, and cytotoxic responses in the CD8+ subset. J Exp Med 187(11):1885–1892
McHugh SM, Rifkin IR, Deighton J, Wilson AB, Lachmann PJ, Lockwood CM, Ewan PW (1995) The immunosuppressive drug thalidomide induces T helper cell type 2 (Th2) and concomitantly inhibits Th1 cytokine production in mitogen- and antigen-stimulated human peripheral blood mononuclear cell cultures. Clin Exp Immunol 99(2):160–167
Dredge K, Horsfall R, Robinson SP, Zhang LH, Lu L, Tang Y, Shirley MA, Muller G, Schafer P, Stirling D, Dalgleish AG, Bartlett JB (2005) Orally administered lenalidomide (CC-5013) is anti-angiogenic in vivo and inhibits endothelial cell migration and Akt phosphorylation in vitro. Microvasc Res 69(1–2):56–63
Gandhi AK, Kang J, Naziruddin S, Parton A, Schafer PH, Stirling DI (2006) Lenalidomide inhibits proliferation of Namalwa CSN.70 cells and interferes with Gab1 phosphorylation and adaptor protein complex assembly. Leuk Res 30(7):849–858
Drappatz J, Wong ET, Schiff D, Kesari S, Batchelor TT, Doherty L, Lafrankie DC, Ramakrishna N, Weiss S, Smith ST, Ciampa A, Zimmerman J, Ostrowsky L, David K, Norden A, Barron L, Sceppa C, Black PM, Wen PY (2009) A pilot safety study of lenalidomide and radiotherapy for patients with newly diagnosed glioblastoma multiforme. Int J Radiat Oncol Biol Phys 73(1):222–227
Fine HA, Kim L, Albert PS, Duic JP, Ma H, Zhang W, Tohnya T, Figg WD, Royce C (2007) A phase I trial of lenalidomide in patients with recurrent primary central nervous system tumors. Clin Cancer Res 13(23):7101–7106
Berg SL, Cairo MS, Russell H, Ayello J, Ingle AM, Lau H, Chen N, Adamson PC, Blaney SM (2011) Safety, pharmacokinetics, and immunomodulatory effects of lenalidomide in children and adolescents with relapsed/refractory solid tumors or myelodysplastic syndrome: a Children’s Oncology Group phase I Consortium Report. J Clin Oncol 29(3):316–323
McCully CL, Balis FM, Bacher J, Phillips J, Poplack DG (1990) A rhesus monkey model for continuous infusion of drugs into cerebrospinal fluid. Lab Anim Sci 40(5):520–525
National Research Council (1996) Guide for the care and use of laboratory animals. National Academy Press, Washington
Gibladi M, Perrier D (1982) Phamacokinetics, 2nd edn. Marcel Dekker, Inc., New York, Basel
Calabrese L, Fleischer AB (2000) Thalidomide: current and potential clinical applications. Am J Med 108(6):487–495
Warren KE, Goldman S, Pollack IF, Fangusaro J, Schaiquevich P, Stewart CF, Wallace D, Blaney SM, Packer R, Macdonald T, Jakacki R, Boyett JM, Kun LE (2011) Phase I trial of lenalidomide in pediatric patients with recurrent, refractory, or progressive primary CNS tumors: Pediatric Brain Tumor Consortium Study PBTC-018. J Clin Oncol 29(3):324–329
Chintagumpala M, Blaney SM, Bomgaars LR, Aleksic A, Kuttesch JF, Klenke RA, Berg SL (2004) Phase I and pharmacokinetic study of thalidomide with carboplatin in children with cancer. J Clin Oncol 22(21):4394–4400
Berg SL, Chamberlain MC (2003) Systemic chemotherapy, intrathecal chemotherapy, and symptom management in the treatment of leptomeningeal metastasis. Curr Oncol Rep 5(1):29–40
Aweeka F, Trapnell C, Chernoff M, Jayewardene A, Spritzler J, Bellibas SE, Lizak P, Jacobson J (2001) Pharmacokinetics and pharmacodynamics of thalidomide in HIV patients treated for oral aphthous ulcers: ACTG protocol 251 AIDS Clinical Trials Group. J Clin Pharmacol 41(10):1091–1097
Figg WD, Raje S, Bauer KS, Tompkins A, Venzon D, Bergan R, Chen A, Hamilton M, Pluda J, Reed E (1999) Pharmacokinetics of thalidomide in an elderly prostate cancer population. J Pharm Sci 88(1):121–125
Piscitelli SC, Figg WD, Hahn B, Kelly G, Thomas S, Walker RE (1997) Single-dose pharmacokinetics of thalidomide in human immunodeficiency virus-infected patients. Antimicrob Agents Chemother 41(12):2797–2799
Noormohamed FH, Youle MS, Higgs CJ, Kook KA, Hawkins DA, Lant AF, Thomas SD (1999) Pharmacokinetics and hemodynamic effects of single oral doses of thalidomide in asymptomatic human immunodeficiency virus-infected subjects. AIDS Res Hum Retroviruses 15(12):1047–1052
Blum W, Klisovic RB, Becker H, Yang X, Rozewski DM, Phelps MA, Garzon R, Walker A, Chandler JC, Whitman SP, Curfman J, Liu S, Schaaf L, Mickle J, Kefauver C, Devine SM, Grever MR, Marcucci G, Byrd JC (2010) Dose escalation of lenalidomide in relapsed or refractory acute leukemias. J Clin Oncol 28(33):4919–4925
Acknowledgments
The grant support is provided by National Cancer Institute K12 Pediatric Clinical Oncology Research Training Program 5K12CA90433-09 (JA Muscal).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Muscal, J.A., Sun, Y., Nuchtern, J.G. et al. Plasma and cerebrospinal fluid pharmacokinetics of thalidomide and lenalidomide in nonhuman primates. Cancer Chemother Pharmacol 69, 943–947 (2012). https://doi.org/10.1007/s00280-011-1781-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-011-1781-y