Abstract
Purpose
BO-1051 is an N-mustard derivative that is conjugated with DNA-affinic 9-anilinoacridine. Since BO-1051 was reported to have strong anticancer activity, we investigated the effect and underlying mechanism of BO-1051 in human glioma cell lines.
Methods
Human glioma cell lines U251MG and U87MG were studied with BO-1051 or the combination of BO-1051 and autophagic inhibitors. Growth inhibition was assessed by MTT assay. Apoptosis was measured by annexin V staining followed by flow cytometry and immunoblotting for apoptosis-related molecules. Induction of autophagy was detected by acridine orange labeling, electron microscopy, LC3 localization and its conversion. Transfection of shRNA was used to determine the involvement of Beclin1 in apoptotic cell death.
Results
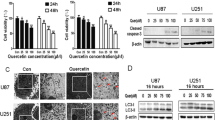
MTT assay showed that BO-1051 suppressed the viability of four glioma cell lines (U251MG, U87MG, GBM-3 and DBTRG-05MG) in a dose-dependent manner. The IC50 values of BO-1051 for the glioma cells were significantly lower than the values for primary neurons cultures and normal fibroblast cells. Moreover, BO-1051 not only induced apoptotic cell death, but also enhanced autophagic flux via inhibition of Akt/mTOR and activation of Erk1/2. Importantly, suppression of autophagy by 3-methyladenine or bafilomycin A1 significantly increased BO-1051-induced apoptotic cell death in U251MG and U87MG cells. In addition, the proportion of apoptotic cells after BO-1051 treatment was enhanced by co-treatment with shRNA against Beclin1.
Conclusions
BO-1051 induced both apoptosis and autophagy, and inhibition of autophagy significantly augmented the cytotoxic effect of BO-1051. Thus, a combination of BO-1051 and autophagic inhibitors offers a potentially new therapeutic modality for the treatment of malignant glioma.






Similar content being viewed by others
References
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Rajski SR, Williams RM (1998) DNA cross-linking agents as antitumor drugs. Chem Rev 98:2723–2796
Maze R, Carney JP, Kelley MR, Glassner BJ, Williams DA et al (1996) Increasing DNA repair methyltransferase levels via bone marrow stem cell transduction rescues mice from the toxic effects of 1, 3-bis(2-chloroethyl)-1-nitrosourea, a chemotherapeutic alkylating agent. Proc Natl Acad Sci USA 93:206–210
Kapuriya N, Kapuriya K, Zhang X, Chou TC, Kakadiya R et al (2008) Synthesis and biological activity of stable and potent antitumor agents, aniline nitrogen mustards linked to 9-anilinoacridines via a urea linkage. Bioorg Med Chem 16:5413–5423
Su TL, Lin YW, Chou TC, Zhang X, Bacherikov VA et al (2006) Potent antitumor 9-anilinoacridines and acridines bearing an alkylating N-mustard residue on the acridine chromophore: synthesis and biological activity. J Med Chem 49:3710–3718
Su TL (2002) Development of DNA topoisomerase II-mediated anticancer agents, 3-(9-acridinylamino)-5-hydroxymethylanilines (AHMAs) and related compounds. Curr Med Chem 9:1677–1688
Chu PM, Chiou SH, Su TL, Lee YJ, Chen LH et al (2011) Enhancement of radiosensitivity in human glioblastoma cells by the DNA N-mustard alkylating agent BO-1051 through augmented and sustained DNA damage response. Radiat Oncol 6:7
Chen S, Rehman SK, Zhang W, Wen A, Yao L et al (2010) Autophagy is a therapeutic target in anticancer drug resistance. Biochim Biophys Acta 1806:220–229
Tiwari M, Bajpai VK, Sahasrabuddhe AA, Kumar A, Sinha RA et al (2008) Inhibition of N-(4-hydroxyphenyl)retinamide-induced autophagy at a lower dose enhances cell death in malignant glioma cells. Carcinogenesis 29:600–609
Shingu T, Fujiwara K, Bogler O, Akiyama Y, Moritake K et al (2009) Inhibition of autophagy at a late stage enhances imatinib-induced cytotoxicity in human malignant glioma cells. Int J Cancer 124:1060–1071
Kanzawa T, Germano IM, Komata T, Ito H, Kondo Y et al (2004) Role of autophagy in temozolomide-induced cytotoxicity for malignant glioma cells. Cell Death Differ 11:448–457
Lomonaco SL, Finniss S, Xiang C, Decarvalho A, Umansky F et al (2009) The induction of autophagy by gamma-radiation contributes to the radioresistance of glioma stem cells. Int J Cancer 125:717–722
Liu WT, Lin CH, Hsiao M, Gean PW (2011) Minocycline inhibits the growth of glioma by inducing autophagy. Autophagy 7:166–175
Alonso MM, Jiang H, Yokoyama T, Xu J, Bekele NB et al (2008) Delta-24-RGD in combination with RAD001 induces enhanced anti-glioma effect via autophagic cell death. Mol Ther 16:487–493
Chao AC, Hsu YL, Liu CK, Kuo PL (2011) alpha-Mangostin, a dietary xanthone, induces autophagic cell death by activating the AMP-activated protein kinase pathway in glioblastoma cells. J Agric Food Chem 59:2086–2096
Kondo Y, Kanzawa T, Sawaya R, Kondo S (2005) The role of autophagy in cancer development and response to therapy. Nat Rev Cancer 5:726–734
Eisenberg-Lerner A, Bialik S, Simon HU, Kimchi A (2009) Life and death partners: apoptosis, autophagy and the cross-talk between them. Cell Death Differ 16:966–975
Fu YS, Lin YY, Chou SC, Tsai TH, Kao LS et al (2008) Tetramethylpyrazine inhibits activities of glioma cells and glutamate neuro-excitotoxicity: potential therapeutic application for treatment of gliomas. Neuro Oncol 10:139–152
Paglin S, Hollister T, Delohery T, Hackett N, McMahill M et al (2001) A novel response of cancer cells to radiation involves autophagy and formation of acidic vesicles. Cancer Res 61:439–444
Chen LH, Loong CC, Su TL, Lee YJ, Chu PM et al (2011) Autophagy inhibition enhances apoptosis triggered by BO-1051, an N-mustard derivative, and involves the ATM signaling pathway. Biochem Pharmacol 81:594–605
Jinno-Oue A, Shimizu N, Hamada N, Wada S, Tanaka A et al (2010) Irradiation with carbon ion beams induces apoptosis, autophagy, and cellular senescence in a human glioma-derived cell line. Int J Radiat Oncol Biol Phys 76:229–241
Kanzawa T, Kondo Y, Ito H, Kondo S, Germano I (2003) Induction of autophagic cell death in malignant glioma cells by arsenic trioxide. Cancer Res 63:2103–2108
Bursch W, Ellinger A, Kienzl H, Torok L, Pandey S et al (1996) Active cell death induced by the anti-estrogens tamoxifen and ICI 164 384 in human mammary carcinoma cells (MCF-7) in culture: the role of autophagy. Carcinogenesis 17:1595–1607
Fu J, Shao CJ, Chen FR, Ng HK, Chen ZP (2010) Autophagy induced by valproic acid is associated with oxidative stress in glioma cell lines. Neuro Oncol 12:328–340
Mizushima N, Yoshimori T, Levine B (2010) Methods in mammalian autophagy research. Cell 140:313–326
Pankiv S, Clausen TH, Lamark T, Brech A, Bruun JA et al (2007) p62/SQSTM1 binds directly to Atg8/LC3 to facilitate degradation of ubiquitinated protein aggregates by autophagy. J Biol Chem 282:24131–24145
Shinojima N, Yokoyama T, Kondo Y, Kondo S (2007) Roles of the Akt/mTOR/p70S6K and ERK1/2 signaling pathways in curcumin-induced autophagy. Autophagy 3:635–637
Scarlatti F, Granata R, Meijer AJ, Codogno P (2009) Does autophagy have a license to kill mammalian cells? Cell Death Differ 16:12–20
Maiuri MC, Zalckvar E, Kimchi A, Kroemer G (2007) Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nat Rev Mol Cell Biol 8:741–752
Levy JM, Thorburn A (2011) Targeting autophagy during cancer therapy to improve clinical outcomes. Pharmacol Ther 131:130–141
Das A, Banik NL, Patel SJ, Ray SK (2004) Dexamethasone protected human glioblastoma U87MG cells from temozolomide induced apoptosis by maintaining Bax:Bcl-2 ratio and preventing proteolytic activities. Mol Cancer 3:36
Haga N, Fujita N, Tsuruo T (2005) Involvement of mitochondrial aggregation in arsenic trioxide (As2O3)-induced apoptosis in human glioblastoma cells. Cancer Sci 96:825–833
Qian W, Liu J, Jin J, Ni W, Xu W (2007) Arsenic trioxide induces not only apoptosis but also autophagic cell death in leukemia cell lines via up-regulation of Beclin-1. Leuk Res 31:329–339
Zhang H, Kong X, Kang J, Su J, Li Y et al (2009) Oxidative stress induces parallel autophagy and mitochondria dysfunction in human glioma U251 cells. Toxicol Sci 110:376–388
Abedin MJ, Wang D, McDonnell MA, Lehmann U, Kelekar A (2007) Autophagy delays apoptotic death in breast cancer cells following DNA damage. Cell Death Differ 14:500–510
Ito H, Daido S, Kanzawa T, Kondo S, Kondo Y (2005) Radiation-induced autophagy is associated with LC3 and its inhibition sensitizes malignant glioma cells. Int J Oncol 26:1401–1410
Herman-Antosiewicz A, Johnson DE, Singh SV (2006) Sulforaphane causes autophagy to inhibit release of cytochrome C and apoptosis in human prostate cancer cells. Cancer Res 66:5828–5835
Harhaji-Trajkovic L, Vilimanovich U, Kravic-Stevovic T, Bumbasirevic V, Trajkovic V (2009) AMPK-mediated autophagy inhibits apoptosis in cisplatin-treated tumor cells. J Cell Mol Med 13:3644–3654
Sotelo J, Briceno E, Lopez-Gonzalez MA (2006) Adding chloroquine to conventional treatment for glioblastoma multiforme: a randomized, double-blind, placebo-controlled trial. Ann Intern Med 144:337–343
Dewaele M, Maes H, Agostinis P (2010) ROS-mediated mechanisms of autophagy stimulation and their relevance in cancer therapy. Autophagy 6:838–854
Acknowledgments
This study was supported by research grants from the National Science Council (NSC97-3111-B-075-001-MY3, NSC98-2320-B-075-003-MY3, NSC99-2628-B- 016-014-MY3 and NSC99-2811-B-016-007-MY3), Taipei Veterans General Hospital (V97B1-006 and E1-008, F-001), Tri-Service General Hospital (TSGH-C100-047), the Joint Projects of UTVGH (VGHUST 98-p1-01), Yen-Tjing-Ling Medical Foundation (96/97/98), National Yang-Ming University (Ministry of Education, Aim for the Top University Plan) & Genomic Center Project, Institute of Biological medicine, Academia Sinica (IBMS-CRC99-p01), and Center of Excellence for Cancer Research at Taipei Veterans General Hospital (DOH99-TD-C-111-007), Taiwan.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chu, PM., Chen, LH., Chen, MT. et al. Targeting autophagy enhances BO-1051-induced apoptosis in human malignant glioma cells. Cancer Chemother Pharmacol 69, 621–633 (2012). https://doi.org/10.1007/s00280-011-1747-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-011-1747-0