Abstract
Purpose
Therapeutic inhibition of angiogenesis has a benefit in the treatment of neoplastic diseases. Cisplatin is a widely used anti-cancer agent; however, it has serious side effects on non-tumor cells and causes nephrotoxicity due to its reactive oxygen species–mediated effect. Thus, a combination between cisplatin and angiogenesis inhibitors may be useful in cancer treatment. In the present study, the effect of sunitinib, a multi-targeted receptor tyrosine kinase inhibitor, on the antitumor activity as well as the nephrotoxic side effect of cisplatin was examined.
Methods
The antitumor activity was evaluated both in vitro using cultured Ehrlich ascites carcinoma (EAC) cells and in vivo using a mouse model of solid tumor. In addition, the effect of cisplatin and/or sunitinib on the angiogenic marker, VEGF, was examined. Nephrotoxicity was induced in rats by single i.p. injection of cisplatin (6 mg/kg).
Results
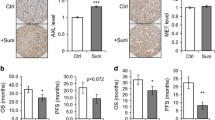
Sunitinib significantly potentiated the cytotoxic effect of cisplatin in vitro and in vivo. The nephrotoxicity of cisplatin was evidenced by decrease in the body weight, increase in kidney/body weight ratio and decrease in the percent survival of rats. The toxicity was also confirmed biochemically by measuring some kidney function parameters and oxidative stress markers. Sunitinib significantly decreased cisplatin-induced changes in serum creatinine, blood urea nitrogen, creatinine clearance and micro total protein in urine, renal malondialdehyde levels and reduced glutathione contents. In addition, sunitinib effectively blunted cisplatin-induced proximal and distal tubules necrosis.
Conclusion
The potential for sunitinib to ameliorate the cisplatin-evoked toxicity as well as to improve the chemotherapeutic effect could have beneficial implications for patients undergoing chemotherapy with cisplatin.


Similar content being viewed by others
References
Atessahim A, Yilmaz S, Karahan I, Ceribasi AO, Karaoglu A (2005) Effects of lycopene against cisplatin-induced nephrotoxicity and oxidative stress in rats. Toxicol 212:116–123
Badary OA, Abdel-Maksoud S, Ahmed WA, Owieda GH (2005) Naringenin attenuates cisplatin nephrotoxicity in rats. Life Sci 76:2125–2135
Badary OA, Sharaby SA, Kenawy SA, El-Denshary EE, Hamada FM (2000) Evaluation of cisplatin combined with ondansetron in Ehrlich ascites carcinoma in vitro and in vivo. Tumori 86:153–156
Baliga R, Zhang Z, Baliga M, Ueda N, Shah SV (1998) In vitro and in vivo evidence suggesting a role for iron in cisplatin-induced nephrotoxicity. Kidney Int 53:394–401
Blokhina OB, Virolainen E, Fagerstedt KV, Hoikkala A, Wähälä K, Chirkova TV (2000) Antioxidant status of anoxia-tolerant and -intolerant plant species under anoxia and reaeration. Physiol Plantarum 109:396–403
Bompart G (1989) Cisplatin-induced changes in cytochrome P-450, lipid peroxidation and drug-metabolizing enzyme activities in rat kidney cortex. Toxicol Lett 48:193–199
Boulikas T, Vougiouka M (2004) Recent clinical trials using cisplatin, carboplatin and their combination chemotherapy drug. Oncol Rep 11:559–595
Cathcart RF 3rd (1985) Vitamin C: the nontoxic, nonrate-limited, antioxidant free radical scavenger. Med Hypotheses 18:61–77
Chirino YI, Hernandez-Pando R, Pedraza-Chaverri J (2004) Peroxynitrite decomposition catalyst ameliorates renal damage and protein nitration in cisplatin-induced nephrotoxicity in rats. BMC Pharmacol 4:20
Conklin KA (2000) Dietary antioxidants during cancer chemotherapy: impact on chemotherapeutic effectiveness and development of side effects. Nutr Cancer 37:1–18
Carmeliet P, Jain RK (2000) Angiogenesis in cancer and other diseases. Nature 407:249–257
Courjault-Gautier F, Le Grimellec C, Giocondi MC, Toutain HJ (1995) Modulation of sodium-coupled uptake and membrane fluidity by cisplatin in renal proximal tubular cells in primary culture and brush-border membrane vesicles. Kidney Int 47:1048–1056
Daniel WW (1991) Hypothesis testing. In: Biostatistics: a foundation for analysis in the health sciences, 5th edn. Wiley, New York, p 191
Draper HH, Hadley M (1990) Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol 186:421–431
Duque I, Garcia-Escribano C, Rodriguez-Puyol M, Diez-Marques ML, Lopez-Novoa JM, Arribas I, Hernando L, Rodriguez-Puyol D (1992) Effects of reactive oxygen species on cultured rat glomeruli. Am J Physiol 263:F466–F473
Ebos JM, Lee CR, Chritensen JG, Mutsaers AJ, Kerbel RS (2007) Multiple circulating proangiogenic factors induced by sunitinib malate are tumor-independent and correlate with antitumor efficacy. Proc Natl Acad Sci USA 104:17069–17074
EL-Azab MF, El-Awady E, Moustafa M, Abd El-Hamid S (2009) Effects of thalidomide, rofecoxib and captopril on vascular endothelial growth factor in Ehrlich ascites carcinoma. Haematologica 94:234 abs. 0572
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77
Eskens FA, Verweij J (2006) The clinical toxicity profile of vascular endothelial growth factor (VEGF) and vascular endothelial growth factor receptor (VEGFR) targeting angiogenesis inhibitors; a review. Eur J Cancer 42:3127–3139
George D, Moore C (2006) Angiogenesis inhibitors in clinical oncology. Update Cancer Ther 1:429–434
Goncalves S, Fernandez-Sanchez R, Sanchez-Nino MD, Tejedor A, Neria F, Egido J, Ruiz-Ortega M, Ortiz A (2010) Tyrphostins as potential therapeutic agents for acute kidney injury. Curr Med Chem 17:974–986
Gulec M, Iraz M, Yilmaz HR, Ozyurtd H, Temel I (2006) The effects of ginkgo biloba extract on tissue adenosine deaminase, xanthine oxidase, myeloperoxidase, malondialdehyde and nitric oxide in cisplatin-induced nephrotoxicity. Toxicol Ind Health 22:125–130
Halliwell B, Whiteman M (2004) Measuring reactive species and oxidative damage in vivo and in cell culture: how should you do it and what do the results mean? Br J Pharmacol 142:231–255 (review)
Henry JB (1974) Todd Sanford Davidsohn: clinical diagnosis and management by laboratory methods, 16th edn. WB Saunders and Co., Philadelphia, p 260
Henry RJ, Cannon DC, Winkelman JW (1974) Clinical chemistry, principles and techniques, 2nd ed edn. Harper and Row, USA, p 525
Jurado R, Lopez-Flores A, Alvarez A, Garcia-Lopez P (2009) Cisplatin cytotoxicity is increased by mifepristone in cervical carcinoma. An in vitro and in vivo study. Oncol Rep 22:1237–1245
Kadikoylu G, Bolaman Z, Demir S, Balkaya M, Akalin N, Enli Y (2004) The effects of desferrioxamine on cisplatin-induced lipid peroxidation and the activities of antioxidant enzymes in rat kidneys. Hum Exp Toxicol 23:29–34
Kellen M, Aronson S, Roizen MF, Barnard J, Thisted RA (1994) Predictive and diagnostic tests of renal failure: a review. Anesth Analg 78:134–142
Kharbanda S, Ren R, Pandey P, Shafman TD, Feller SM, Weichselbaum RR, Kufe DW (1995) Activation of the c-Abl tyrosine kinase in the stress response to DNA-damaging agents. Nature 376:785–788
Kim KJ, Li B, Winer J, Armanini M, Gillett N, Phillips HS, Ferrara N (1993) Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumour growth in vivo. Nature 362:841–844
Kontovinis LF, Papazisis KT, Touplikioti P, Andreadis C, Mouratidou D, Kortsaris AH (2009) Sunitinib treatment for patients with clear-cell metastatic renal cell carcinoma: clinical outcomes and plasma angiogenesis markers. BMC Cancer 9:82
Kuhlmann MK, Burkhardt G, Kohler H (1997) Insights into potential cellular mechanisms of cisplatin nephrotoxicity and their clinical application. Nephrol Dial Transplant 12:2478–2480
Leung DW, Cachianes G, Kuang WJ, Goeddel DV, Ferrara N (1989) Vascular endothelial growth factor is a secreted angiogenic mitogen. Science 246:1306–1309
Marklund SL (1985) Superoxide dismutase isoenzymes in tissues and plasma from New Zealand black mice, nude mice and normal BALB/c mice. Mutat Res 148:129–134
Masuda H, Tanaka T, Takahama UT (1994) Cisplatin generates superoxide anion by interaction with DNA in a cell free system. Biochem Biophys Res Commun 203:175–180
Mueller PW, Lash L, Price RG, Stolte H, Gelpi E, Maack T, Berndt WO (1997) Urinary biomarkers to detect significant effects of environmental and occupational exposure to nephrotoxins. I. Categories of tests for detecting effects of nephrotoxins. Renal Fail 19:505–521
Norden-Zfoni A, Desai J, Manola J, Beaudry P, Force J, Maki R, Folkman J, Bello C, Baumc DePrimo SE, Shalinsky DR, Demetri GD, Heymach JV (2007) Blood-based biomarkers of SU11248 activity and clinical outcome in patients with metastatic imatinib-resistant gastrointestinal stromal tumor. Clin Cancer Res 13:2643–2650
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Oncu M, Gultekin F, Karaöz E, Altuntas I, Delibas N (2002) Nephrotoxicity in rats induced by chlorpryfos-ethyl and ameliorating effects of antioxidants. Hum Exp Toxicol 21:223–230
Plate KH, Breier G, Weich HA, Risau W (1992) Vascular endothelial growth factor is a potential tumour angiogenesis factor in human gliomas in vivo. Nature 359:845–848
Raja Naresh RA, Ndupa N, Uma Devi P (1996) Effect of macrophage activation on niosome encapsulated bleomycin in tumor- bearing mice. Ind J Pharamcol 28:175–180
Ries F, Klastersky J (1986) Nephrotoxicity induced by cancer chemotherapy with special emphasis on cisplatin toxicity. Am J Kidney Dis 8:368–379
Schirner M, Hoffmann J, Menrad A, Schneider MR (1998) Antiangiogenic chemotherapeutic agents: characterization in comparison to their tumor growth inhibition in human renal cell carcinoma models. Clin Cancer Res 4:1331–1336
Shimeda Y, Hirotani Y, Akimoto Y, Shindou K, Ijiri Y, Nishihori T, Tanaka K (2005) Protective effects of capsaicin against cisplatin-induced nephrotoxicity in rats. Biol Pharm Bull 28:1635–1638
Sugiyama S, Hayakawa M, Kato T, Hanaki Y, Shimizu K, Ozawa T (1989) Adverse effects of anti-tumor drug, cisplatin, on rat kidney mitochondria: disturbances in glutathione peroxidase activity. Biochem Biophys Res Commun 159:1121–1127
Sung MJ, Kim DH, Jung YJ, Kong KP, Lee AS, Lee S, Kim W, Davaatseren M, Hwang JT, Kim HJ, Kim MS, Kwon DY, Park SK (2008) Genistein protects the kidney from cisplatin-induced injury. Kidney Int 74:1538–1547
To YF, Sun RW, Chen Y, Chan VS, Yu WY, Tam PK, Che CM, Lin CL (2009) Gold (III) porphyrin complex is more potent than cisplatin in inhibiting growth of nasopharyngeal carcinoma in vitro and in vivo. Int J Cancer 124:1971–1979
Tsutsumishita Y, Onda T, Okada K, Takeda M, Endou H, Futaki S, Niwa M (1998) Involvement of H2O2 production in cisplatin-induced nephrotoxicity. Biochem Biophys Res Commun 242:310–312
Watanabe N, Kamei S, Ohkubo A, Yamanaka M, Ohsawa S, Makino K, Tokuda K (1986) Urinary protein as measured with a pyrogallol red-molybdate complex, manually and in a Hitachi 726 automated analyzer. Clin Chem 32:1551–1554
Weisenthal LM, Dill PL, Kunick NB, Lippman ME (1983) Comparison of dye exclusion assays with a cologenic assay in the determination of drug-induced cytotoxicity. Cancer Res 43:258–264
Yee MS, Blakley BW, Begleiter A, Leith M (2008) Delayed sodium thiosulphate administration reduces cisplatin efficacy on mouse EMT6 tumour cells in vitro. J Otolaryngol Head Neck Surg 37:638–641
Yoshida M, Itzuka K, Hara M, Nishijima H, Shimada A, Nakada K, Satoh Y, Kama Y, Terada A (2000) Prevention of nephrotoxicity of cisplatin by repeated oral administration of ebselen in rats. Tohoku J Exp Med 191:209–220
Zhang N, Erjala K, Kulmala J, Qiu X, Sundvall M, Elenius K, Grenman R (2009) Concurrent cetuximab, cisplatin and radiation for squamous cell carcinoma of the head and neck in vitro. Radiother-Oncol 92:388–392
Acknowledgments
This work was supported by grants from Mansoura University, Egypt. The funder has no role in study design, data collection and analysis or preparation of the manuscript. Disclosures: None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Suddek, G.M. Sunitinib improves chemotherapeutic efficacy and ameliorates cisplatin-induced nephrotoxicity in experimental animals. Cancer Chemother Pharmacol 67, 1035–1044 (2011). https://doi.org/10.1007/s00280-010-1402-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-010-1402-1