Abstract
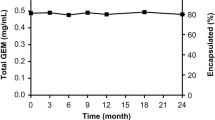
Despite its rapid enzymatic inactivation and therefore limited activity in vivo, Gemcitabine is the standard drug for pancreatic cancer treatment. To protect the drug, and achieve passive tumor targeting, we developed a liposomal formulation of Gemcitabine, GemLip (∅: 36 nm: 47% entrapment). Its anti-tumoral activity was tested on MIA PaCa-2 cells growing orthotopically in nude mice. Bioluminescence measurement mediated by the stable integration of the luciferase gene was employed to randomize the mice, and monitor tumor growth. GemLip (4 and 8 mg/kg), Gemcitabine (240 mg/kg), and empty liposomes (equivalent to 8 mg/kg GemLip) were injected intravenously once weekly for 5 weeks. GemLip (8 mg/kg) stopped tumor growth, as measured via in vivo bioluminescence, reducing the primary tumor size by 68% (SD ± 8%; p < 0.02), whereas Gemcitabine hardly affected tumor size (-7%; ± 1.5%). In 80% of animals, luciferase activity in the liver indicated the presence of metastases. All treatments, including the empty liposomes, reduced the metastatic burden. Thus, GemLip shows promising antitumoral activity in this model. Surprisingly, empty liposomes attenuate the spread of metastases similar to Gemcitabine and GemLip. Further, luciferase marked tumor cells are a powerful tool to observe tumor growth in vivo, and to detect and quantify metastases.






Similar content being viewed by others
References
Jemal A, Tiwari RC, Murray T, Ghafoor A, Samuels A, Ward E, Feuer EJ, Thun MJ (2004) CA Cancer J Clin 54(1):8–29
Noble S, Goa KL (1997) Drugs 54(3):447–472
Axelson J, Lindell M, Horlin K, Ohlsson B (2005) Pancreatology 5(2–3):251–258
Zagon IS, Jaglowski JR, Verderame MF, Smith JP, Leure-Dupree AE, McLaughlin PJ (2005) Cancer Chemother Pharmacol 56(5):510–520
Abbruzzese JL, Grunewald R, Weeks EA, Gravel D, Adams T, Nowak B, Mineishi S, Tarassoff P, Satterlee W, Raber MN et al (1991) J Clin Oncol 9(3):491–498
Wang LR, Huang MZ, Xu N, Shentu JZ, Liu J, Cai J (2005) J Zhejiang Univ Sci B 6(5):446–450
Reid JM, Qu W, Safgren SL, Ames MM, Krailo MD, Seibel NL, Kuttesch J, Holcenberg J (2004) J Clin Oncol 22(12):2445–2451
Moog R, Burger AM, Brandl M, Schuler J, Schubert R, Unger C, Fiebig HH, Massing U (2002) Cancer Chemother Pharmacol 49(5):356–366
Shipley LA, Brown TJ, Cornpropst JD, Hamilton M, Daniels WD, Culp HW (1992) Drug Metab Dispos 20(6):849–855
van Borssum Waalkes M, Kuipers F, Havinga R, Scherphof GL (1993) Biochim Biophys Acta 1176(1–2):43–50
van Borssum Waalkes M, van Galen M, Morselt H, Sternberg B, Scherphof GL (1993) Biochim Biophys Acta 1148(1):161–172
Yuan F, Dellian M, Fukumura D, Leunig M, Berk DA, Torchilin VP, Jain RK (1995) Cancer Res 55(17):3752–3756
Massing U (1997) Int J Clin Pharmacol Ther 35(2):87–90
Massing U, Fuxius S (2000) Drug Resist Updat 3(3):171–177
Brandl M, Massing U (2003) Vesicular phospholipid gels. In: New RW, Torchillin V (eds) Liposomes practical approaches. IRL-Press at Oxford University Press, Oxford
Schueler J (1998) Entwicklung und Charakterisierung humaner Tumormodelle durch orthotope Implantation. In: Inst. für Veterinärpathologie, Freie Universität, Berlin
Sipos B, Moser S, Kalthoff H, Torok V, Lohr M, Kloppel G (2003) Virchows Arch 442(5):444–452
Bold RJ, Chandra J, McConkey DJ (1999) Ann Surg Oncol 6(3):279–285
Katz MH, Bouvet M, Takimoto S, Spivack D, Moossa AR, Hoffman RM (2003) Cancer Res 63(17):5521–5525
Hotz HG, Reber HA, Hotz B, Yu T, Foitzik T, Buhr HJ, Cortina G, Hines OJ (2003) Pancreas 26(4):e89–e98
Tomioka D, Maehara N, Kuba K, Mizumoto K, Tanaka M, Matsumoto K, Nakamura T (2001) Cancer Res 61(20):7518–7524
Tsutsumi S, Yanagawa T, Shimura T, Kuwano H, Raz A (2004) Clin Cancer Res 10(22):7775–7784
Bouvet M, Yang M, Nardin S, Wang X, Jiang P, Baranov E, Moossa AR, Hoffman RM (2000) Clin Exp Metastasis 18(3):213–218
Shah K, Weissleder R (2005) NeuroRx 2(2):215–225
Tardi C (1999) Vesikuläre Phospholipidgele: in vitro Charakterisierung, Autoklavierbarkeit, Anwendung als Depotarzneiform. In. Dept. Pharmaceutical Technology, Albert-Ludwigs-University of Freiburg, Freiburg
Gossen M, Bujard H (1992) Proc Natl Acad Sci USA 89(12):5547–5551
Workman P, Balmain A, Hickman JA, McNally NJ, Rohas AM, Mitchison NA, Pierrepoint CG, Raymond R, Rowlatt C, Stephens TC et al (1988) Lab Anim 22(3):195–201
Giovannetti E, Mey V, Danesi R, Mosca I, Del Tacca M (2004) Clin Cancer Res 10(9):2936–2943
Braakhuis BJ, van Dongen GA, Vermorken JB, Snow GB (1991) Cancer Res 51(1):211–214
Sener SF, Fremgen A, Menck HR, Winchester DP (1999) J Am Coll Surg 189(1):1–7
Eibl G, Reber HA (2005) Pancreas 31(3):258–262
Keller R (1985) Invasion Metastasis 5(5):295–308
Nagami H, Nakano K, Ichihara H, Matsumoto Y, Ueoka R (2006) Bioorg Med Chem Lett 16(4):782–785
Acknowledgment
We are extremely grateful to Marta Rodriguez-Franco and Gunter Neuhaus (Institute for Biology II; Cell Biology;University Freiburg) for the permission to use their CCD camera, and their lab-members for hosting us. Furthermore, we would like to thank Sandra Pöllath and Bianca Giesen for excellent technical assistance, and Lenka Taylor for critically reading the manuscript. This work was funded, in part, by grants from the Clotten Stiftung and Dietmar Hopp Stiftung GmbH.
Author information
Authors and Affiliations
Corresponding author
Additional information
C. Bornmann and R. Graeser have equally contributed to this article.
Rights and permissions
About this article
Cite this article
Bornmann, C., Graeser, R., Esser, N. et al. A new liposomal formulation of Gemcitabine is active in an orthotopic mouse model of pancreatic cancer accessible to bioluminescence imaging. Cancer Chemother Pharmacol 61, 395–405 (2008). https://doi.org/10.1007/s00280-007-0482-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-007-0482-z