Abstract
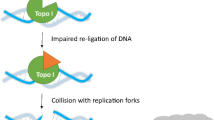
The effect of 7-alkyl substitutions on growth inhibition in seven Camptothecin (CPT) ring systems with various groups at the ten position was evaluated in three human breast cancer cell lines that model (1) hormone-sensitive (MCF-7/wt), (2) hormone insensitive (MDA-MB-231), or (3) alkylator-resistant (MCF-7/4-hc) forms of disease. To assess the impact of persistence of cleavage complexes on antiproliferative activity, a post-exposure recovery period in drug-free medium was incorporated into the growth inhibition assay. This modification produced on average a twofold reduction in the growth inhibition endpoint (the IC50), suggesting a greater apoptotic response. The results further revealed a three log range in potency from a mean IC50 of 2 nM (7-butyl-10,11-methylenedioxy-CPT) to 2.5 μM (7-bromomethyl-10-hydryoxy-CPT). Increasing 7-alkyl chain length in six of the ten-substituted CPTs enhanced potency, which was directly correlated with persistence of topoisomerase I-induced DNA cleavage complexes in 10-hydroxy, 10-methoxy, and 10,11-methylenedioxy substituted CPTs. Modeling of the binding mode of 7-butyl-10-amino-CPT revealed a direct hydrogen bond contact for the 10-amino to the side chain of Glu-356 of Core Subdomain I of top1 in addition to known contacts found for other camptothecins. More important, residues 350–356 and 425–431 of Core Subdomain I may provide induced fit stabilization to the lipophilic alkyl moiety at the seven position.







Similar content being viewed by others
Abbreviations
- top1:
-
Topoisomerase I
- CPT:
-
Camptothecin
- MD or MDO:
-
10,11-Methylenedioxy
- ED:
-
10,11-Ethylenedioxy
- MeO:
-
Methoxy
- DFMD:
-
Difluoromethylenedioxy
- CMMDC:
-
7-Chloromethyl-10,11-methylenedioxy-camptothecin
- SN-38:
-
7-Ethyl-10-hydroxy-camptothecin
- BACPT:
-
7-Butyl-10-amino-camptothecin
References
Bellarosa D, Ciucci A, Bullo A, Nardelli F, Manzini S, Maggi CA, Goso C (2001) Apoptotic events in a human ovarian cancer cell line exposed to anthracyclines. J Pharmacol Exp Ther 296:276–283
Bom D, Curran DP, Kruszewski S, Zimmer SG, Thompson Strode J, Kohlhagen G, Du W, Chavan AJ, Fraley KA, Bingcang AL, Latus LJ, Pommier Y, Burke TG (2000) The novel silatecan 7-tert-butyldimethylsilyl-10-hydroxycamptothecin displays high lipophilicity, improved human blood stability, and potent anticancer activity. J Med Chem 43:3970–3980
Flowers JL, Ludeman SM, Gamcsik MP, Colvin OM, Shao KL, Boal JH, Springer JB, Adams DJ (2000) Evidence for a role of chloroethylaziridine in the cytotoxicity of cyclophosphamide. Cancer Chemother Pharmacol 45:335–344
Gamcsik M, Kasibhatla M, Adams D, Flowers J, Colvin O, Manikumar G, Wani M, Wall M, Kohlhagen G, Pommier Y (2001) Dual role of glutathione in modulating camptothecin activity: depletion potentiates activity, but conjugation enhances the stability of the topoisomerase I-DNA cleavage complex. Mol Cancer Ther 1:11–20
Giovanella B, Stehlin J, Wall M, Wani M, Nicholas A, Liu L, Silber R, Potmesil M (1989) DNA topoisomerase I-targeted chemotherapy of human colon cancer in xenografts. Science 246:1046–1048
Hirabayashi N, Kim R, Nishiyama M, Aogi K, Saeki S, Toge T, Okada K (1992) Tissue expression of topoisomerase I and II in digestive tract cancers and adjacent normal tissues (abstract). Proc Am Assoc Cancer Res 33:436
Hsiang Y-H, Hertzberg R, Hecht S, Lui R (1985) Camptothecin induces protein-linked DNA breaks via mammalian DNA topoisomerase I. J Biol Chem 260:14873–14878
Husain I, Mohler J, Seigler H, Besterman J (1994) Elevation of topoisomerase I messenger RNA, protein and catalytic activity in human tumors: demonstration of tumor-type specificity and implications for cancer chemotherapy. Cancer Res 54:539–546
Jaxel C, Kohn KW, Wani MC, Wall ME, Pommier Y (1989) Structure-activity study of the actions of camptothecin derivatives on mammalian topoisomerase I: evidence for a specific receptor site and a relation to antitumor activity. Cancer Res 49:1465–1469
Kehrer DFS, Soepenberg O, Loos WJ, Verweij J, Sparreboom A (2001) Modulation of camptothecin analogs in the treatment of cancer: a review. Anticancer Drugs 12:89–105
Li TK, Liu LF (2001) Tumor cell death induced by topoisomerase-targeting drugs. Annu Rev Pharmacol Toxicol 41:53–77
Matsui S, Endo W, Wrzosek C, Haridas K, Seetharamulu P, Hausheer FH, Rustum YM (1999) Characterisation of a synergistic interaction between a thymidylate synthase inhibitor, ZD1694, and a novel lipophilic topoisomerase I inhibitor karenitecin, BNP1100: mechanisms and clinical implications. Eur J Cancer 35:984–993
Paull K, Shoemaker R, Hodes L, Monks A, Scudiero D, Rubinstein L, Plowman J, Boyd M (1989) Display and analysis of patterns of differential activity of drugs against human tumor cell lines: development of mean graph and COMPARE algorithm. J Natl Cancer Inst 81:1088–1092
Pommier Y, Kohlhagen G, Kohn KW, Leteurtre F, Wani MC, Wall ME (1995) Interaction of an alkylating camptothecin derivative with a DNA base at topoisomerase I-DNA cleavage sites. Proc Natl Acad Sci USA 92:8861–8865
Pommier Y, Kohlhagen G, Laco G, Kroth H, Sayer J, Jerina D (2002) Different effects on human topoisomerase I by minor groove and intercalated deoxyguanosine adducts derived from two polycyclic aromatic hydrocarbon diol epoxides at or near a normal cleavage site. J Biol Chem 277:13666–13672
Pommier Y, Pourquier P, Fan Y, Strumberg D (1998) Mechanism of action of eukaryotic DNA topoisomerase I and drugs targeted to the enzyme. Biochim Biophys Acta 1400:83–105
Potmesil M, Hsiang Y-H, Liu L et al (1988) Topoisomerase I (topo-I) and topoisomerase II (topo-II) levels in high and low grade lymphomas. Proc Am Assoc Cancer Res 29:176
Pourquier P, Takebayashi Y, Urasaki Y, Gioffre C, Kohlhagen G, Pommier Y (2000) Induction of topoisomerase I cleavage complexes by 1-beta-D- arabinofuranosylcytosine (ara-C) in vitro and in ara-C-treated cells. Proc Natl Acad Sci USA 97:1885–1890
Redinbo MR, Stewart L, Kuhn P, Champoux JJ, Hol WGJ (1998) Crystal structures of human topoisomerase I in covalent and noncovalent complexes with DNA. Science 279:1504–1513
Sawada S, Nokata K, Furuta T, Yokokura T, Miyasake T (1991) Chemical modification of an antitumor alkaloid camptothecin: Synthesis and antitumor activity of 7-C-substituted camptothecins. Chem Pharm Bull 39:2574–2580
Staker BL, Hjerrild K, Feese MD, Behnke CA, Burgin AB, Stewart L (2002) The mechanism of topoisomerase I poisoning by a camptothecin analog. Proc Natl Acad Sci USA 99:15387–15392
Valenti M, Nieves-Neira W, Kohlhagen G, Kohn K, Wall M, Wani M, Pommier Y (1997) Novel 7-alkyl methylenedioxy-camptothecin derivatives exhibit increased cytotoxicity and induce persistent cleavable complexes both with purified mammalian topoisomerase I and in human colon carcinoma SW620 cells. Mol Pharm 52:82–87
Van der Zee A, Hollema H, DeJong S, Boonstra H, Gouw A, Willemse P, Zijlstra J, de Vries E (1991) P-glycoprotein expression and DNA topoisomerase I and II activity in benign tumors of the ovary and in malignant tumors of the ovary, before and after platinum/cyclophosphamide chemotherapy. Cancer Res 51:5915–5920
Van Hattum AH, Pinedo HM, Schluper HM, Hausheer FH, Boven E (2000) New highly lipophilic camptothecin BNP1350 is an effective drug in experimental human cancer. Int J Cancer 88:260–266
Vladu B, Woynarowski JM, Manikumar G, Wani MC, Wall ME, Von Hoff DD, Wadkins RM (2000) 7- and 10-substituted camptothecins: dependence of topoisomerase I-DNA cleavable complex formation and stability on the 7- and 10-substituents. Mol Pharmacol 57:243–251
Wall M, Wani M, Cook C, Palmer K, McPhail H, Sim G (1966) Plant antitumor agents. I. The isolation and structure of camptothecin, a novel alkaloidal leukemia and tumor inhibitor from Camptotheca acuminata. J Am Chem Soc 88:3888–3890
Wall ME, Wani MC, Nicholas AW, Manikumar G, Tele C, Moore L, Truesdale A, Leitner P, Besterman JM (1993) Plant antitumor agents. 30. Synthesis and structure activity of novel camptothecin analogs. J Med Chem 36:2689–2700
Wang XY, Wang LK, Kingsbury WD, Johnson RK, Hecht SM (1998) Differential effects of camptothecin derivatives on topoisomerase I-mediated DNA structure modification. Biochemistry 37:9399–9408
Acknowledgements
This work was supported by NIH grant UO1 CA68697-02 and is dedicated to the memory of Dr. Monroe E. Wall, who inspired this research team and many other investigators committed to creating useful anticancer drugs from natural products.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Adams, D.J., Silva, M.W.d., Flowers, J.L. et al. Camptothecin analogs with enhanced activity against human breast cancer cells. I. Correlation of potency with lipophilicity and persistence in the cleavage complex. Cancer Chemother Pharmacol 57, 135–144 (2006). https://doi.org/10.1007/s00280-005-0007-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-005-0007-6