Abstract
Purpose
To identify the optimal schedule for intraperitoneal (i.p.) infusion of floxuridine (FUDR) against peritoneal micrometastases from gastric cancer.
Methods
The efficacy of continuous i.p. infusion of FUDR was compared with that of bolus i.p. administration in peritoneal gastric cancer (MKN45) xenografts. The FUDR continuous delivery system in this study was in the form of injectable poly(lactic-coglycolic) acid (PLGA) microspheres intended for i.p. injection. Animals were treated by continuous i.p. infusion using FUDR-loaded microspheres or bolus i.p. administration of FUDR.
Results
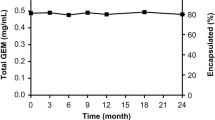
In vitro testing demonstrated that FUDR was released slowly from the microspheres at a rate of approximately 5% of the total encapsulated drug per day. In in vivo studies, the peritoneal level was found to persist and was approximately 5- to 50-fold higher than that of plasma for more than 2 weeks following a single injection of the microspheres. An in vitro MTT assay showed that exposure time clearly influenced the cytotoxic potency of FUDR. In vivo, continuous infusion was more effective against peritoneal tumor than bolus administration at equivalent doses. However, compared with bolus administration, toxicity was increased, resulting in a reduced maximum tolerated dose (MTD) with continuous infusion. When the treatment was carried out at each MTD (continuous 1 mg/kg, bolus 600 mg/kg), continuous infusion had no advantage in inhibiting tumor growth.
Conclusions
Owing to the higher toxicity and the equal efficacy of continuous infusion compared with bolus administration, continuous infusion is not recommended in i.p. FUDR treatment.





Similar content being viewed by others
References
Parkin DM, Bray FI, Devesa SS (2001) Cancer burden in the year 2000. The global picture. Eur J Cancer 37 [Suppl 8]:S4
Shimada K, Ajani JA (1999) Adjuvant therapy for gastric carcinoma patients in the past 15 years: a review of western and oriental trials. Cancer 86:1657
Earle CC, Maroun JA (1999) Adjuvant chemotherapy after curative resection for gastric cancer in non-Asian patients: revisiting a meta-analysis of randomised trials. Eur J Cancer 35:1059
Mari E, Floriani I, Tinazzi A, Buda A, Belfiglio M, Valentini M, Cascinu S, Barni S, Labianca R, Torri V (2000) Efficacy of adjuvant chemotherapy after curative resection for gastric cancer: a meta-analysis of published randomised trials. A study of the GISCAD (Gruppo Italiano per lo Studio dei Carcinomi dell’Apparato Digerente). Ann Oncol 11:837
Janunger KG, Hafstrom L, Nygren P, Glimelius B (2001) A systematic overview of chemotherapy effects in gastric cancer. Acta Oncol 40:309
Panzini I, Gianni L, Fattori PP, Tassinari D, Imola M, Fabbri P, Arcangeli V, Drudi G, Canuti D, Fochessati F, Ravaioli A (2002) Adjuvant chemotherapy in gastric cancer: a meta-analysis of randomized trials and a comparison with previous meta-analyses. Tumori 88:21
Yoo CH, Noh SH, Shin DW, Choi SH, Min JS (2000) Recurrence following curative resection for gastric carcinoma. Br J Surg 87:236
Maehara Y, Hasuda S, Koga T, Tokunaga E, Kakeji Y, Sugimachi K (2000) Postoperative outcome and sites of recurrence in patients following curative resection of gastric cancer. Br J Surg 87:353
Markman M (2001) Intraperitoneal drug delivery of antineoplastics. Drugs 61:1057
Muggia FM, Chan KK, Russell C, Colombo N, Speyer JL, Sehgal K, Jeffers S, Sorich J, Leichman L, Beller U (1991) Phase I and pharmacologic evaluation of intraperitoneal 5-fluoro-2′-deoxyuridine. Cancer Chemother Pharmacol 28:241
Leichman L, Silberman H, Leichman CG, Spears CP, Ray M, Muggia FM, Kiyabu M, Radin R, Laine L, Stain S (1992) Preoperative systemic chemotherapy followed by adjuvant postoperative intraperitoneal therapy for gastric cancer: a University of Southern California pilot program. J Clin Oncol 10:1933
Kelsen DP, Saltz L, Cohen AM, Yao TJ, Enker W, Tong W, Tao Y, Bertino JR (1994) A phase I trial of immediate postoperative intraperitoneal floxuridine and leucovorin plus systemic 5-fluorouracil and levamisole after resection of high risk colon cancer. Cancer 74:2224
Muggia FM, Liu PY, Alberts DS, Wallace DL, O’Toole RV, Terada KY, Franklin EW, Herrer GW, Goldberg DA, Hannigan EV (1996) Intraperitoneal mitoxantrone or floxuridine: effects on time-to-failure and survival in patients with minimal residual ovarian cancer after second-look laparotomy—a randomized phase II study by the Southwest Oncology Group. Gynecol Oncol 61:395
Crookes P, Leichman CG, Leichman L, Tan M, Laine L, Stain S, Baranda J, Casagrande Y, Groshen S, Silberman H (1997) Systemic chemotherapy for gastric carcinoma followed by postoperative intraperitoneal therapy: a final report. Cancer 79:1767
Culliford AT 4th, Brooks AD, Sharma S, Saltz LB, Schwartz GK, O’Reilly EM, Ilson DH, Kemeny NE, Kelsen DP, Guillem JG, Wong WD, Cohen AM, Paty PB (2001) Surgical debulking and intraperitoneal chemotherapy for established peritoneal metastases from colon and appendix cancer. Ann Surg Oncol 8:787
Okada H, Toguchi H (1995) Biodegradable microspheres in drug delivery. Crit Rev Ther Drug Carrier Syst 12:1
Smith-Rogers JA, Tong WP, Duafala ME, Markman M, Bertino JR (1991) High-performance liquid chromatographic method for the simultaneous measurement of floxuridine and fluorouracil in human body fluids. J Chromatogr 566:147
Poorter RL, Bakker PJ, Veenhof CH (1998) Continuous infusion of chemotherapy: focus on 5-fluorouracil and fluorodeoxyuridine. Pharm World Sci 20:45
Makhija S, Leitao M, Sabbatini P, Bellin N, Almadrones L, Leon L, Spriggs DR, Barakat R (2001) Complications associated with intraperitoneal chemotherapy catheters. Gynecol Oncol 81:77
Markman M (1991) Intraperitoneal chemotherapy. Semin Oncol 18:248
Link KH, Aigner KR, Peschau K, Warthona M, Schwemmle K, Danenberg PV (1988) Concentration and time dependence of the toxicity of fluorinated pyrimidines to HT 29 colorectal carcinoma cells. Cancer Chemother Pharmacol 22:58
Sullivan RD, Miller E (1965) The clinical effects of prolonged intravenous infusion of 5-fluoro-2′-deoxyuridine. Cancer Res 25:1025
Lokich JJ, Sonneborn H, Paul S, Zipoli T (1983) Phase I study of continuous venous infusion of floxuridine (5-FUDR) chemotherapy. Cancer Treat Rep 67:791
Moertel CG, Reitemeier RJ, Hahn RG (1967) A controlled comparison of 5-fluoro-2′-deoxyuridine therapy administered by rapid intravenous injection and by continuous intravenous infusion. Cancer Res 27:549
Meta-analysis Group In Cancer (1998) Efficacy of intravenous continuous infusion of fluorouracil compared with bolus administration in advanced colorectal cancer. J Clin Oncol 16:301
Ensminger WD, Rosowsky A, Raso V, Levin DC, Glode M, Come S, Steele G, Frei E (1978) A clinical-pharmacological evaluation of hepatic arterial infusions of 5-fluoro-2′-deoxyuridine and 5-fluorouracil. Cancer Res 38:3784
Meta-Analysis Group In Cancer (1996) Reappraisal of hepatic arterial infusion in the treatment of nonresectable liver metastases from colorectal cancer. J Natl Cancer Inst 88:252
Kemeny N, Huang Y, Cohen AM, Shi W, Conti JA, Brennan MF, Bertino JR, Turnbull AD, Sullivan D, Stockman J, Blumgart LH, Fong Y (1999) Hepatic arterial infusion of chemotherapy after resection of hepatic metastases from colorectal cancer. N Engl J Med 341:2039
Kemeny MM, Adak S, Gray B, Macdonald JS, Smith T, Lipsitz S, Sigurdson ER, O’Dwyer PJ, Benson AB (2002) Combined-modality treatment for resectable metastatic colorectal carcinoma to the liver: surgical resection of hepatic metastases in combination with continuous infusion of chemotherapy—an intergroup study. J Clin Oncol 20:1499
Acknowledgements
We thank Dr. Teiichi Motoyama for providing the human gastric cancer cell line (MKN45).
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported in part by a grant from the Japan Society for the Promotion of Science and by a grant from the Setsuro Fujii Memorial Osaka Foundation for Promotion of Fundamental Medical Research.
Rights and permissions
About this article
Cite this article
Inoue, K., Onishi, H., Kato, Y. et al. Comparison of intraperitoneal continuous infusion of floxuridine and bolus administration in a peritoneal gastric cancer xenograft model. Cancer Chemother Pharmacol 53, 415–422 (2004). https://doi.org/10.1007/s00280-003-0748-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-003-0748-z