Abstract
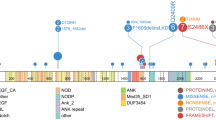
T cell acute lymphoblastic leukemia (T-ALL) is an aggressive neoplasm for which there are currently no adequate biomarkers for developing risk-adapted therapeutic regimens to improve the treatment outcome. In this prospective study of 83 Chinese patients (54 children and 29 adults) with de novo T-ALL, we analyzed mutations in 11 T-ALL genes: NOTCH1, FBXW7, PHF6, PTEN, N-RAS, K-RAS, WT1, IL7R, PIK3CA, PIK3RA, and AKT1. NOTCH1 mutations were identified in 51.9 and 37.9 % of pediatric and adult patients, respectively, and these patients showed improved overall survival (OS) and event-free survival (EFS). The FBXW7 mutant was present in 25.9 and 6.9 % of pediatric and adult patients, respectively, and was associated with inferior OS and EFS in pediatric T-ALL. Multivariate analysis revealed that mutant FBXW7 was an independent prognostic indicator for inferior EFS (hazard ratio [HR] 4.38; 95 % confidence interval [CI] 1.15–16.71; p = 0.03) and tended to be associated with reduced OS (HR 2.81; 95 % CI 0.91–8.69; p = 0.074) in pediatric T-ALL. Mutant PHF6 was present in 13 and 20.7 % of our childhood and adult cohorts, respectively, while PTEN mutations were noted in 11.1 % of the pediatric patients. PTEN and NOTCH1 mutations were almost mutually exclusive, while IL7R and WT1 mutations were rare in pediatric T-ALL and PTPN11 and AKT1 mutations were infrequent in adult T-ALL. This study revealed differences in the mutational profiles of pediatric and adult T-ALL and suggests mutant FBXW7 as an independent prognostic indicator for inferior survival in pediatric T-ALL.


Similar content being viewed by others
References
Pui CH, Carroll WL, Meshinchi S, Arceci RJ (2011) Biology, risk stratification, and therapy of pediatric acute leukemias: an update. J Clin Oncol 29(5):551–565. doi:10.1200/JCO.2010.30.7405
Marks DI, Paietta EM, Moorman AV, Richards SM, Buck G, DeWald G, Ferrando A, Fielding AK, Goldstone AH, Ketterling RP, Litzow MR, Luger SM, McMillan AK, Mansour MR, Rowe JM, Tallman MS, Lazarus HM (2009) T-cell acute lymphoblastic leukemia in adults: clinical features, immunophenotype, cytogenetics, and outcome from the large randomized prospective trial (UKALL XII/ECOG 2993). Blood 114(25):5136–5145. doi:10.1182/blood-2009-08-231217
Grossmann V, Haferlach C, Weissmann S, Roller A, Schindela S, Poetzinger F, Stadler K, Bellos F, Kern W, Haferlach T, Schnittger S, Kohlmann A (2013) The molecular profile of adult T-cell acute lymphoblastic leukemia: mutations in RUNX1 and DNMT3A are associated with poor prognosis in T-ALL. Genes Chromosom Cancer 52(4):410–422. doi:10.1002/gcc.22039
Pui CH, Robison LL, Look AT (2008) Acute lymphoblastic leukaemia. Lancet 371(9617):1030–1043. doi:10.1016/S0140-6736(08)60457-2
Ferrando AA, Neuberg DS, Staunton J, Loh ML, Huard C, Raimondi SC, Behm FG, Pui CH, Downing JR, Gilliland DG, Lander ES, Golub TR, Look AT (2002) Gene expression signatures define novel oncogenic pathways in T cell acute lymphoblastic leukemia. Cancer Cell 1(1):75–87
Radtke F, Wilson A, Mancini SJ, MacDonald HR (2004) Notch regulation of lymphocyte development and function. Nat Immunol 5(3):247–253. doi:10.1038/ni1045
Weng AP, Ferrando AA, Lee W, Morris JP, Silverman LB, Sanchez-Irizarry C, Blacklow SC, Look AT, Aster JC (2004) Activating mutations of NOTCH1 in human T cell acute lymphoblastic leukemia. Science 306(5694):269–271. doi:10.1126/science.1102160
Asnafi V, Buzyn A, Le Noir S, Baleydier F, Simon A, Beldjord K, Reman O, Witz F, Fagot T, Tavernier E, Turlure P, Leguay T, Huguet F, Vernant JP, Daniel F, Bene MC, Ifrah N, Thomas X, Dombret H, Macintyre E (2009) NOTCH1/FBXW7 mutation identifies a large subgroup with favorable outcome in adult T-cell acute lymphoblastic leukemia (T-ALL): a Group for Research on Adult Acute Lymphoblastic Leukemia (GRAALL) study. Blood 113(17):3918–3924. doi:10.1182/blood-2008-10-184069
O’Neil J, Grim J, Strack P, Rao S, Tibbitts D, Winter C, Hardwick J, Welcker M, Meijerink JP, Pieters R, Draetta G, Sears R, Clurman BE, Look AT (2007) FBW7 mutations in leukemic cells mediate NOTCH pathway activation and resistance to gamma-secretase inhibitors. J Exp Med 204(8):1813–1824. doi:10.1084/jem.20070876
Zhu YM, Zhao WL, Fu JF, Shi JY, Pan Q, Hu J, Gao XD, Chen B, Li JM, Xiong SM, Gu LJ, Tang JY, Liang H, Jiang H, Xue YQ, Shen ZX, Chen Z, Chen SJ (2006) NOTCH1 mutations in T-cell acute lymphoblastic leukemia: prognostic significance and implication in multifactorial leukemogenesis. Clin Cancer Res 12(10):3043–3049. doi:10.1158/1078-0432.CCR-05-2832
Zuurbier L, Homminga I, Calvert V, te Winkel ML, Buijs-Gladdines JG, Kooi C, Smits WK, Sonneveld E, Veerman AJ, Kamps WA, Horstmann M, Petricoin EF 3rd, Pieters R, Meijerink JP (2010) NOTCH1 and/or FBXW7 mutations predict for initial good prednisone response but not for improved outcome in pediatric T-cell acute lymphoblastic leukemia patients treated on DCOG or COALL protocols. Leukemia 24(12):2014–2022. doi:10.1038/leu.2010.204
Gao C, Liu SG, Zhang RD, Li WJ, Zhao XX, Cui L, Wu MY, Zheng HY, Li ZG (2014) NOTCH1 mutations are associated with favourable long-term prognosis in paediatric T-cell acute lymphoblastic leukaemia: a retrospective study of patients treated on BCH-2003 and CCLG-2008 protocol in China. Br J Haematol 166(2):221–228. doi:10.1111/bjh.12866
Van Vlierberghe P, Patel J, Abdel-Wahab O, Lobry C, Hedvat CV, Balbin M, Nicolas C, Payer AR, Fernandez HF, Tallman MS, Paietta E, Melnick A, Vandenberghe P, Speleman F, Aifantis I, Cools J, Levine R, Ferrando A (2011) PHF6 mutations in adult acute myeloid leukemia. Leukemia 25(1):130–134. doi:10.1038/leu.2010.247
Gutierrez A, Sanda T, Grebliunaite R, Carracedo A, Salmena L, Ahn Y, Dahlberg S, Neuberg D, Moreau LA, Winter SS, Larson R, Zhang J, Protopopov A, Chin L, Pandolfi PP, Silverman LB, Hunger SP, Sallan SE, Look AT (2009) High frequency of PTEN, PI3K, and AKT abnormalities in T-cell acute lymphoblastic leukemia. Blood 114(3):647–650. doi:10.1182/blood-2009-02-206722
Zenatti PP, Ribeiro D, Li W, Zuurbier L, Silva MC, Paganin M, Tritapoe J, Hixon JA, Silveira AB, Cardoso BA, Sarmento LM, Correia N, Toribio ML, Kobarg J, Horstmann M, Pieters R, Brandalise SR, Ferrando AA, Meijerink JP, Durum SK, Yunes JA, Barata JT (2011) Oncogenic IL7R gain-of-function mutations in childhood T-cell acute lymphoblastic leukemia. Nat Genet 43(10):932–939. doi:10.1038/ng.924
Shochat C, Tal N, Bandapalli OR, Palmi C, Ganmore I, te Kronnie G, Cario G, Cazzaniga G, Kulozik AE, Stanulla M, Schrappe M, Biondi A, Basso G, Bercovich D, Muckenthaler MU, Izraeli S (2011) Gain-of-function mutations in interleukin-7 receptor-alpha (IL7R) in childhood acute lymphoblastic leukemias. J Exp Med 208(5):901–908. doi:10.1084/jem.20110580
Perentesis JP, Bhatia S, Boyle E, Shao Y, Shu XO, Steinbuch M, Sather HN, Gaynon P, Kiffmeyer W, Envall-Fox J, Robison LL (2004) RAS oncogene mutations and outcome of therapy for childhood acute lymphoblastic leukemia. Leukemia 18(4):685–692. doi:10.1038/sj.leu.2403272
Renneville A, Kaltenbach S, Clappier E, Collette S, Micol JB, Nelken B, Lepelley P, Dastugue N, Benoit Y, Bertrand Y, Preudhomme C, Cave H (2010) Wilms tumor 1 (WT1) gene mutations in pediatric T-cell malignancies. Leukemia 24(2):476–480. doi:10.1038/leu.2009.221
Gao C, Zhao XX, Li WJ, Cui L, Zhao W, Liu SG, Yue ZX, Jiao Y, Wu MY, Li ZG (2012) Clinical features, early treatment responses, and outcomes of pediatric acute lymphoblastic leukemia in China with or without specific fusion transcripts: a single institutional study of 1,004 patients. Am J Hematol 87(11):1022–1027. doi:10.1002/ajh.23307
Forbes SA, Bhamra G, Bamford S, Dawson E, Kok C, Clements J, Menzies A, Teague JW, Futreal PA, Stratton MR (2008) The Catalogue of Somatic Mutations in Cancer (COSMIC). Current protocols in human genetics/editorial board, Jonathan L Haines [et al.] Chapter 10:Unit 10 11. doi:10.1002/0471142905.hg1011s57
Song JH, Schnittke N, Zaat A, Walsh CS, Miller CW (2008) FBXW7 mutation in adult T-cell and B-cell acute lymphocytic leukemias. Leuk Res 32(11):1751–1755. doi:10.1016/j.leukres.2008.03.040
Huh HJ, Lee SH, Yoo KH, Sung KW, Koo HH, Jang JH, Kim K, Kim SJ, Kim WS, Jung CW, Lee KO, Kim SH, Kim HJ (2013) Gene mutation profiles and prognostic implications in Korean patients with T-lymphoblastic leukemia. Ann Hematol 92(5):635–644. doi:10.1007/s00277-012-1664-2
Park MJ, Taki T, Oda M, Watanabe T, Yumura-Yagi K, Kobayashi R, Suzuki N, Hara J, Horibe K, Hayashi Y (2009) FBXW7 and NOTCH1 mutations in childhood T cell acute lymphoblastic leukaemia and T cell non-Hodgkin lymphoma. Br J Haematol 145(2):198–206. doi:10.1111/j.1365-2141.2009.07607.x
Lee SY, Kumano K, Masuda S, Hangaishi A, Takita J, Nakazaki K, Kurokawa M, Hayashi Y, Ogawa S, Chiba S (2005) Mutations of the Notch1 gene in T-cell acute lymphoblastic leukemia: analysis in adults and children. Leukemia 19(10):1841–1843. doi:10.1038/sj.leu.2403896
Thompson BJ, Buonamici S, Sulis ML, Palomero T, Vilimas T, Basso G, Ferrando A, Aifantis I (2007) The SCFFBW7 ubiquitin ligase complex as a tumor suppressor in T cell leukemia. J Exp Med 204(8):1825–1835. doi:10.1084/jem.20070872
Breit S, Stanulla M, Flohr T, Schrappe M, Ludwig WD, Tolle G, Happich M, Muckenthaler MU, Kulozik AE (2006) Activating NOTCH1 mutations predict favorable early treatment response and long-term outcome in childhood precursor T-cell lymphoblastic leukemia. Blood 108(4):1151–1157. doi:10.1182/blood-2005-12-4956
Mansour MR, Sulis ML, Duke V, Foroni L, Jenkinson S, Koo K, Allen CG, Gale RE, Buck G, Richards S, Paietta E, Rowe JM, Tallman MS, Goldstone AH, Ferrando AA, Linch DC (2009) Prognostic implications of NOTCH1 and FBXW7 mutations in adults with T-cell acute lymphoblastic leukemia treated on the MRC UKALLXII/ECOG E2993 protocol. J Clin Oncol 27(26):4352–4356. doi:10.1200/JCO.2009.22.0996
Fujii Y, Yada M, Nishiyama M, Kamura T, Takahashi H, Tsunematsu R, Susaki E, Nakagawa T, Matsumoto A, Nakayama KI (2006) Fbxw7 contributes to tumor suppression by targeting multiple proteins for ubiquitin-dependent degradation. Cancer Sci 97(8):729–736. doi:10.1111/j.1349-7006.2006.00239.x
Mao JH, Kim IJ, Wu D, Climent J, Kang HC, DelRosario R, Balmain A (2008) FBXW7 targets mTOR for degradation and cooperates with PTEN in tumor suppression. Science 321(5895):1499–1502. doi:10.1126/science.1162981
Van Vlierberghe P, Ferrando A (2012) The molecular basis of T cell acute lymphoblastic leukemia. J Clin Invest 122(10):3398–3406. doi:10.1172/JCI61269
Lower KM, Turner G, Kerr BA, Mathews KD, Shaw MA, Gedeon AK, Schelley S, Hoyme HE, White SM, Delatycki MB, Lampe AK, Clayton-Smith J, Stewart H, van Ravenswaay CM, de Vries BB, Cox B, Grompe M, Ross S, Thomas P, Mulley JC, Gecz J (2002) Mutations in PHF6 are associated with Borjeson-Forssman-Lehmann syndrome. Nat Genet 32(4):661–665. doi:10.1038/ng1040
Van Vlierberghe P, Palomero T, Khiabanian H, Van der Meulen J, Castillo M, Van Roy N, De Moerloose B, Philippe J, Gonzalez-Garcia S, Toribio ML, Taghon T, Zuurbier L, Cauwelier B, Harrison CJ, Schwab C, Pisecker M, Strehl S, Langerak AW, Gecz J, Sonneveld E, Pieters R, Paietta E, Rowe JM, Wiernik PH, Benoit Y, Soulier J, Poppe B, Yao X, Cordon-Cardo C, Meijerink J, Rabadan R, Speleman F, Ferrando A (2010) PHF6 mutations in T-cell acute lymphoblastic leukemia. Nat Genet 42(4):338–342. doi:10.1038/ng.542
Chao MM, Todd MA, Kontny U, Neas K, Sullivan MJ, Hunter AG, Picketts DJ, Kratz CP (2010) T-cell acute lymphoblastic leukemia in association with Borjeson-Forssman-Lehmann syndrome due to a mutation in PHF6. Pediatr Blood Cancer 55(4):722–724. doi:10.1002/pbc.22574
Wang Q, Qiu H, Jiang H, Wu L, Dong S, Pan J, Wang W, Ping N, Xia J, Sun A, Wu D, Xue Y, Drexler HG, Macleod RA, Chen S (2011) Mutations of PHF6 are associated with mutations of NOTCH1, JAK1 and rearrangement of SET-NUP214 in T-cell acute lymphoblastic leukemia. Haematologica 96(12):1808–1814. doi:10.3324/haematol.2011.043083
Jiang Q, Li WQ, Aiello FB, Mazzucchelli R, Asefa B, Khaled AR, Durum SK (2005) Cell biology of IL-7, a key lymphotrophin. Cytokine Growth Factor Rev 16(4–5):513–533. doi:10.1016/j.cytogfr.2005.05.004
Palomero T, Sulis ML, Cortina M, Real PJ, Barnes K, Ciofani M, Caparros E, Buteau J, Brown K, Perkins SL, Bhagat G, Agarwal AM, Basso G, Castillo M, Nagase S, Cordon-Cardo C, Parsons R, Zuniga-Pflucker JC, Dominguez M, Ferrando AA (2007) Mutational loss of PTEN induces resistance to NOTCH1 inhibition in T-cell leukemia. Nat Med 13(10):1203–1210. doi:10.1038/nm1636
Bandapalli OR, Zimmermann M, Kox C, Stanulla M, Schrappe M, Ludwig WD, Koehler R, Muckenthaler MU, Kulozik AE (2013) NOTCH1 activation clinically antagonizes the unfavorable effect of PTEN inactivation in BFM-treated children with precursor T-cell acute lymphoblastic leukemia. Haematologica 98(6):928–936. doi:10.3324/haematol.2012.073585
Trinquand A, Tanguy-Schmidt A, Ben Abdelali R, Lambert J, Beldjord K, Lengline E, De Gunzburg N, Payet-Bornet D, Lhermitte L, Mossafa H, Lheritier V, Bond J, Huguet F, Buzyn A, Leguay T, Cahn JY, Thomas X, Chalandon Y, Delannoy A, Bonmati C, Maury S, Nadel B, Macintyre E, Ifrah N, Dombret H, Asnafi V (2013) Toward a NOTCH1/FBXW7/RAS/PTEN-based oncogenetic risk classification of adult T-cell acute lymphoblastic leukemia: a Group for Research in Adult Acute Lymphoblastic Leukemia study. J Clin Oncol 31(34):4333–4342. doi:10.1200/JCO.2012.48.5292
Gandre-Babbe S, Paluru P, Aribeana C, Chou ST, Bresolin S, Lu L, Sullivan SK, Tasian SK, Weng J, Favre H, Choi JK, French DL, Loh ML, Weiss MJ (2013) Patient-derived induced pluripotent stem cells recapitulate hematopoietic abnormalities of juvenile myelomonocytic leukemia. Blood 121(24):4925–4929. doi:10.1182/blood-2013-01-478412
Irving J, Matheson E, Minto L, Blair H, Case M, Halsey C, Swidenbank I, Ponthan F, Kirschner-Schwabe R, Groeneveld-Krentz S, Hof J, Allan J, Harrison C, Vormoor J, von Stackelberg A, Eckert C (2014) Ras pathway mutations are prevalent in relapsed childhood acute lymphoblastic leukemia and confer sensitivity to MEK inhibition. Blood 124(23):3420–3430. doi:10.1182/blood-2014-04-531871
Xu D, Liu X, Yu WM, Meyerson HJ, Guo C, Gerson SL, Qu CK (2011) Non-lineage/stage-restricted effects of a gain-of-function mutation in tyrosine phosphatase Ptpn11 (Shp2) on malignant transformation of hematopoietic cells. J Exp Med 208(10):1977–1988. doi:10.1084/jem.20110450
Tartaglia M, Martinelli S, Cazzaniga G, Cordeddu V, Iavarone I, Spinelli M, Palmi C, Carta C, Pession A, Arico M, Masera G, Basso G, Sorcini M, Gelb BD, Biondi A (2004) Genetic evidence for lineage-related and differentiation stage-related contribution of somatic PTPN11 mutations to leukemogenesis in childhood acute leukemia. Blood 104(2):307–313. doi:10.1182/blood-2003-11-3876
Bains T, Heinrich MC, Loriaux MM, Beadling C, Nelson D, Warrick A, Neff TL, Tyner JW, Dunlap J, Corless CL, Fan G (2012) Newly described activating JAK3 mutations in T-cell acute lymphoblastic leukemia. Leukemia 26(9):2144–2146. doi:10.1038/leu.2012.74
Yamamoto T, Isomura M, Xu Y, Liang J, Yagasaki H, Kamachi Y, Kudo K, Kiyoi H, Naoe T, Kojma S (2006) PTPN11, RAS and FLT3 mutations in childhood acute lymphoblastic leukemia. Leuk Res 30(9):1085–1089. doi:10.1016/j.leukres.2006.02.004
Paschka P, Marcucci G, Ruppert AS, Whitman SP, Mrozek K, Maharry K, Langer C, Baldus CD, Zhao W, Powell BL, Baer MR, Carroll AJ, Caligiuri MA, Kolitz JE, Larson RA, Bloomfield CD (2008) Wilms’ tumor 1 gene mutations independently predict poor outcome in adults with cytogenetically normal acute myeloid leukemia: a cancer and leukemia group B study. J Clin Oncol 26(28):4595–4602. doi:10.1200/JCO.2007.15.2058
Virappane P, Gale R, Hills R, Kakkas I, Summers K, Stevens J, Allen C, Green C, Quentmeier H, Drexler H, Burnett A, Linch D, Bonnet D, Lister TA, Fitzgibbon J (2008) Mutation of the Wilms’ tumor 1 gene is a poor prognostic factor associated with chemotherapy resistance in normal karyotype acute myeloid leukemia: the United Kingdom Medical Research Council Adult Leukaemia Working Party. J Clin Oncol 26(33):5429–5435. doi:10.1200/JCO.2008.16.0333
Tosello V, Mansour MR, Barnes K, Paganin M, Sulis ML, Jenkinson S, Allen CG, Gale RE, Linch DC, Palomero T, Real P, Murty V, Yao X, Richards SM, Goldstone A, Rowe J, Basso G, Wiernik PH, Paietta E, Pieters R, Horstmann M, Meijerink JP, Ferrando AA (2009) WT1 mutations in T-ALL. Blood 114(5):1038–1045. doi:10.1182/blood-2008-12-192039
Acknowledgments
We are grateful to the patients, families, and physicians for their participation in the study. The study was supported in part by the Children’s Hospital of Chongqing Medical University (Grant No. ZHYX2013-7).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Lan Yuan and Ling Lu contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
PCR primers and annealing temperatures (PDF 180 kb)
Table S2
Genetic aberrations identified in the pediatric T-ALL cohort (PDF 55 kb)
Table S3
Genetic aberrations identified in the adult T-ALL cohort (PDF 22 kb)
Fig. S1
Survival analyses according to the presence of NOTCH1 mutations in adult T-ALL and the NOTCH1 and FBXW7 mutants in pediatric T-ALL. (a and b) Kaplan-Meier plots of OS and EFS for pediatric T-ALL patients with mutated NOTCH1 and/or mutated FBXW7 and wild-type NOTCH1 and FBXW7. (c) Kaplan-Meier plots of OS for adult T-ALL patients with mutated and wild-type NOTCH1. (JPEG 46 kb)
Rights and permissions
About this article
Cite this article
Yuan, L., Lu, L., Yang, Y. et al. Genetic mutational profiling analysis of T cell acute lymphoblastic leukemia reveal mutant FBXW7 as a prognostic indicator for inferior survival. Ann Hematol 94, 1817–1828 (2015). https://doi.org/10.1007/s00277-015-2474-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-015-2474-0