Abstract
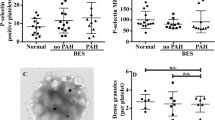
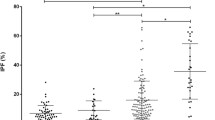
Patients with thalassemia, an inherited hemolytic anemia, have increased risk of hypercoagulable complications. A whole blood flow cytometric (FCM) method has been used for studies of platelet activation and platelet–leukocyte aggregation in these patients. However, this FCM method presents technical difficulties because of the high proportion of immature red blood cells (RBCs) in these patients. A protocol for the simultaneous measurement of platelet activation and their aggregation with leukocyte populations in whole blood using four-color FCM which excluded immature RBC was devised, and evaluated for the evaluation of platelet function in patients with β-thalassemia/hemoglobin E (HbE). Whole blood from these patients and from healthy volunteers was stained for platelet activation and platelet–leukocyte aggregates using anti-CD42a, anti-CD62P, anti-CD45 and glycophorin A (GPA) conjugated with different fluorochromes. Our FCM method is simple, effective and based on the assumption that GPA is present on all immature RBCs, but is not expressed on CD45+ leukocytes. Results from the studies showed that blood samples from these patients contained a high frequency of circulating activated platelets (CD42a+/CD62P+) when compared to samples from healthy individuals. The percentage of platelet–neutrophil, platelet–monocyte—but not platelet–lymphocyte—aggregates were also elevated in both thalassemia genotypes with marked increase in patients who had undergone splenectomy. These findings suggest that platelets adhere to neutrophils and monocytes are activated which support the clinical observation that splenectomized thalassemia patients have an increased risk of arterial or venous thrombotic manifestations.





Similar content being viewed by others
References
Weatherall DJ (2001) Towards molecular medicine; reminiscences of the haemoglobin field, 1960–2000. Br J Haematol 115:729–738. doi:10.1046/j.1365-2141.2001.03227
Weatherall DJ, Clegg JB (2004) The thalassaemia syndromes, 4th edn. Blackwell, Oxford
WHO working group (1982) Hereditary anaemias: genetic basis, clinical features, diagnosis, and treatment. Bull World Health Org 60:643–808
Fucharoen S, Winichagoon P (1987) Hemoglobinopathies in Southeast Asia. Hemoglobin 11:65–88. doi:10.3109/03630268709036587
Wasi P, Na-nakorn S, Pootrakul S, Sookanek M, Pornpatkula M, Panich V (1969) Alpha and beta thalassemia in Thailand. Ann N Y Acad Sci 165:60–82. doi:10.1111/j.1749-6632.1969.tb27777
Eldor A, Rachmilewitz EA (2002) The hypercoagulable state in thalassemia. Blood 99:36–43. doi:10.1182/blood.V99.1.36
Joseph M (1995) The generation of free radicals by blood platelets. Immunopharmacology of platelets. Academic Press, San Diego, pp 209–223
Michelson AD (2003) How platelets work: platelet function and dysfunction. J Thromb Thrombolysis 16:7–12. doi:10.1023/B:THRO.0000014586.77684.82
Neumann FJ, Zohlnhofer D, Fakhoury L, Ott I, Gawaz M, Schomig A (1999) Effect of glycoprotein IIb/IIIa receptor blockade on platelet–leukocyte interaction and surface expression of the leukocyte integrin Mac-1 in acute myocardial infarction. J Am Coll Cardiol 34:1420–1426. doi:10.1016/S0735-1097(99)00350-2
Khairy M, Lasne D, Brohard-Bohn, Aiach M, Rendu F, Bachelot-Loza C (2001) A new approach in the study of the molecular and cellular events implicated in heparin-induced thrombocytopenia. Formation of leukocyte–platelet aggregates. Thromb Haemost 85:1090–1096
Fisher TC, Meiselmann HJ (1994) Polymorphonuclear leukocytes in ischemic vascular disease. Thromb Res 74(Suppl 1):S21–S34. doi:10.1016/S0049-3848(10)80004-0
Jungi TW, Spycher MO, Nydegger, Uebarandun S (1986) Platelet–leukocyte interaction: selective binding of thrombin-stimulated platelets to human monocytes, polymorphonuclear leukocytes, and related cell lines. Blood 67:629–636
Ault KA, Rinder HM, Mitchell JG, Rinder CS, Lambrew CT, Hillman RS (1989) Correlated measurement of platelet release and aggregation in whole blood. Cytometry 10:448–455. doi:10.1002/cyto.990100413
Michelson AD, Ellis PA, Barnard MR, Matic GB, Viles AF, Kestin AS (1991) Downregulation of the platelet surface glycoprotein Ib–IX complex in whole blood stimulated by thrombin, ADP or an in vivo wound. Blood 77:770–779
Pattanapanyasat K, Kyle DE, Tongtawe P, Yongvanitchit K, Fucharoen S (1994) Flow cytometric immunophenotyping of lymphocyte subsets in samples that contain a high proportion of non-lymphoid cells. Cytometry 18:199–208. doi:10.1002/cyto.990180403
Eldor A, Lellouche F, Goldfarb A, Rachmilewitz EA, Maclouf J (1991) In vivo platelet activation in beta thalassemia major reflected by increased platelet thromboxane urinary metabolites. Blood 77:1749–1753
Del Principe D, Menichelli A, Di Giulio S, De Matteis W, Cianciulli P, Papa G (1993) PADGEM/GMP-140 expression on platelet membranes from homozygous β-thalassaemic patients. Br J Haematol 84:111–117. doi:10.1111/j.1365-2141.1993.tb03033.x
Opartkiattikul N, Funahara Y, Fucharoen S, Talalak P (1992) Increase in spontaneous platelet aggregation in β-thalassaemia/haemoglobin E disease: a consequence of splenectomy. Southeast Asian J Trop Med Publ Health 23:36–41
Eldor A (1978) Abnormal platelet functions in beta thalassaemia. Scand J Haematol 20:447–452
Hussain MA, Hutton RA, Pavlidou O, Hoffbrand AV (1979) Platelet function in beta-thalassaemia major. J Clin Pathol 32:429–433. doi:10.1136/jcp.32.5.429
Michelson AD, Barnard MR, Hechtman HB, MacGregor H, Connolly RJ, Loscalzo J, Valeri CR (2001) In vivo tracking of platelet: circulating degranulated platelets rapidly loss surface P-selectin but continue to circulate and function. Proc Natl Acad Sci USA 93:11877–11882
Simon DI, Chen Z, Xu H, Li CQ, Dong J–f, McIntire LV, Ballantyne CM, Zhang L, Furman MI, Berndt MC (2000) Platelet glycoprotein Ibα is a counterreceptor for the leukocyte integrin Mac-1(CD11b/CD18). J Exp Med 192:193–204. doi:10.1084/jem/192.2.193
Sentoso S, Sachs UJH, Kroll H, Linder M, Ruf A, Preissner KT, Chavakis T (2002) The junctional adhesion molecule 3 (JAM-3) on human platelets is a counterreceptor for the leukocyte integrin Mac-1. J Exp Med 196:679–691. doi:10.1084/jem.20020267
McEver RP (2007) P-selectin/PSGL-1 and other interactions between platelets, leukocytes, and endothelium. In: Michelson AD (ed) Platelets, 2nd edn. Elsevier/Academic Press, San Diego, pp 231–249
Muller WA (2003) Leukocyte–endothelial-cell interactions in leukocyte transmigration and the inflammatory response. Trends Immunol 24:326–333. doi:10.1016/S1471-4906(03)00117-0
Michelson AD, Barnard MR, Krueger LA, Valeri CR, Furman MI (2001) Circulating monocyte–platelet aggregates are a more sensitive marker of in vivo platelet activation than platelet surface P-selectin: studies in baboons, human coronary intervention, and human acute myocardial infarction. Circulation 104:1533–1537. doi:10.1161/hc3801.095588
Winichagoon P, Fucharoen S, Wasi P (1981) Increased circulating platelet aggregates in thalassaemia. Southeast Asian J Trop Med Public Health 12:556–560
Nagasue N, Inokuchi K, Kobayashi M, Kanashima R (1978) Platelet aggregability after splenectomy in patients with normosplenism and hypersplenism. Am J Surg 136:260–264. doi:10.1016/0002-9610(78)90242-8
Furman MI, Benoit SE, Barnard MR, Valeri CR, Borbone ML, Becker RC, Hechtman HB, Michelson AD (1998) Increased platelet reactivity and circulating monocyte–platelet aggregates in patients with stable coronary artery disease. J Am Coll Cardiol 31:352–358. doi:10.1016/S0735-1097(97)00510-X
Peyton BD, Rohrer MJ, Furman MI, Barnard MR, Rodino LJ, Benoit SE, Hechtman HB, Valeri CR, Michelson AD (1998) Patients with venous stasis ulceration have increased monocyte–platelet aggregation. J Vasc Surg 27:1109–1116
Grau AJ, Ruf A, Vogt A, Lichy C, Buggle F, Patscheke H, Hacke W (1998) Increased fraction of circulating activated platelets in acute and previous cerebrovascular ischemia. Thromb Haemost 80:298–301
McCabe DJ, Harrison P, Mackie IJ, Sidhu PS, Purdy G, Lawrie AS, Watt H, Brown MM, Machin SJ (2004) Platelet degranulation and monocyte–platelet complex formation are increased in the acute and convalescent phases after ischaemic stroke or transient ischaemic attack. Br J Haematol 125:777–787. doi:10.1111/j.1365-2141.2004.04983
Esposito CJ, Popescu WM, Rinder HM, Schwartz JJ, Smith BR, Rinder CS (2003) Increased leukocyte–platelet adhesion in patients with graft occlusion after peripheral vascular surgery. Thromb Haemost 90:1128–1134. doi:10.1160/TH03-04-0226
Acknowledgments
We sincerely thank Professor Aftab A. Ansari of the Department of Pathology and Laboratory Medicine, Emory University School of Medicine, USA, for valuable discussions. Support for this study was provided by the Thailand Research Fund – Senior Research Scholar Award, Chalermprakiat Grant, Faculty of Medicine Siriraj Hospital, Mahidol University, and Office of the Higher Education Commission and Mahidol University under the National Research Universities Initiative. S.F. and K.P. are presently Thailand Research Fund – Senior Research Scholar.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Keawvichit, R., Khowawisetsut, L., Chaichompoo, P. et al. Platelet activation and platelet–leukocyte interaction in β-thalassemia/hemoglobin E patients with marked nucleated erythrocytosis. Ann Hematol 91, 1685–1694 (2012). https://doi.org/10.1007/s00277-012-1522-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-012-1522-2