Abstract
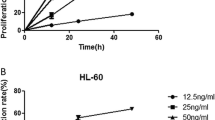
This investigation was designed to assess the effect of DuP-697 on growth and apoptosis in a human chronic myeloid leukemia (CML) cell line (K562 cells) and primary CML cells from CML patient bone marrow. DuP-697 significantly suppressed K562 cells and primary CML cells growth and induced apoptosis in a concentration-dependent manner and the growth-inhibiting effect was independent on Philadelphia chromosome. The IC50 of DuP-697 at 36 h was 31.7 μM. It arrested G1-S phase transmit on cell cycle and its apoptosis activity was partially abrogated by pretreating K562 cells with Z-IETD-fmk, a specific inhibitor of caspase-8. This study suggested that Dup-697 suppresses growth and induces apoptosis on K562 leukemia cells by cell-cycle arrest and caspase-8 activation.





Similar content being viewed by others
References
Pang R, Hayes M, Rests D et al (2004) Pharmakinetics and pharmacodynamics of imatinib in a phase 1 trial with chronic myeloid leukemia patients. J Clin Oncol 22:935–942
Inokuchi K (2006) Chronic myelogenous leukemia: from molecular biology to clinical aspects and novel targeted therapies. J Nippon Med Sch 73:178–192
Talpaz M, Silver RT, Druker BJ et al (2002) Imatinib induces durable hematologic and cytogenetic responses in patients with accelerated phase chronic myeloid leukemia results of a phase 2 study. Blood 99:1928–1937
Marin D, Marktel S, Szydlo R et al (2003) Survival of patients with chronic-phase chronic myeloid leukeamia on imatinib after failure on interferon alfa. Lancet 362:617–619
Druker BJ, Tarnura S, Buchdunger E et al (1996) Effects of a selective inhibitor of the Ahl tyrosine kinase on the growth of Bcr-Abl positive cells. Nat Med 2:561–566
Gorre ME, Mohammed M, Ellwood K et al (2001) Clinical resistance to STI-571 cancer therapy caused by BCR-ABL gene mutation or amplification. Science 293:876–880
Hochhaus A, Kreil S, Corbin AS et al (2002) Molecular and chromosomal mechanisms of resistance to imatinib (STI571) therapy. Leukemia 16:2190–2196
FitzGerald GA, Patrono C (2001) The coxibs, selective inhibitors of cyclooxygenase-2. N Engl J Med 345:433–442
Larkins TL, Nowell M, Singh S et al (2006) Inhibition of cyclooxygenase-2 decreases breast cancer cell motility, invasion and matrix metalloproteinase expression. BMC Cancer 6:181–193
Pai R, Soreghan B, Szabo IL et al (2002) Prostaglandin E2 transactivates EGF receptor: a novel mechanism for promoting colon cancer growth and gastrointestinal hypertrophy. Nat Med 8:289–293
Zweifel BS, Davis TW, Ornberg RL et al (2002) Direct evidence for a role of cyclooxygenase 2-derived prostaglandin E2 in human head and neck xenograft tumors. Cancer Res 62:6706–6711
Dohadwala M, Batra RK, Luo J et al (2002) Autocrine/paracrine prostaglandin E2 production by nonsmall cell lung cancer cells regulates matrix metalloproteinase-2 and CD44 in cyclooxygenase-2-dependent invasion. J Biol Chem 277:50828–50833
Denkert C, Winzer KJ, Muller BM et al (2003) Elevated expression of cyclooxygenase-2 is a negative prognostic factor for disease free survival and overall survival in patients with breast carcinoma. Cancer 97:2978–2987
Gupta GP, Nguyen DX, Chiang AC et al (2007) Mediators of vascular remodeling co-opted for sequential steps in lung metastasis. Nature 446:765–770
Steinbach G, Lynch PM, Phillips RK et al (2000) The effect of celecoxib, a cyclooxygenase-2 inhibitor, in familial adenomatous polyposis. N Engl J Med 342:1946–1952
Zhang GS, Tu CQ, Zhang GY et al (2000) Indomethacin induces apoptosis and inhibits proliferation in chronic myeloid leukemia cells. Leuk Res 24:385–392
Zhang GS, Liu DS, Dai CW et al (2006) Antitumor effects of celecoxib on K562 leukemia cells are mediated by cell-cycle arrest, caspase-3 activation, and downregulation of cox-2 expression and are synergistic with hydroxyurea or imatinib. Am J Hematol 81:242–255
Paul W (2006) Balancing the cyclooxygenase portfolio. Can Med Assoc J 174:1581–1582
Tsujii M, Kawano S, DuBois RN (1997) Cyclooxygenase-2 expression in human colon cancer cells increases metastatic potential. Proc Natl Acad Sci U S A 94:3336–3340
Secchiero P, Barbarotto E, Gonelli A et al (2005) Potential pathogenetic implications of cyclooxygenase-2 overexpres-sion in B chronic lymphoid leukemia cells. Am J Pathol 167:1599–1607
Wun T, McKnighta H, Joseph M et al (2004) Increased cyclooxygenase-2 (COX-2): a potential role in the pathogenesis of lymphoma. Leuk Res 28:179–190
Ladetto M, Vallet S, Trojan A et al (2005) Cyclooxygenase-2 (COX-2) is frequently expressed in multiple myeloma and is an independent predictor of poor outcome. Blood 105:4784–4791
Seibert K, Masferrer JL, Needleman P, Salvemini D (1996) Pharmacological manipulation of cyclo-oxygenase-2 in the inflamed hydronephrotic kidney. Br J Pharmacol 117:1016–1020
Granax, Reddy EP (1995) Cell cycle control in mammalian cells: role of cyclins, cyclin dependent kinesis (CDKs), growth suppressor genes and cyclin-dependent kinase inhibitors (CKIs). Oncogene 11:211–219
Johnson AJ, Song X, HsuA-L et al (2001) Apoptosis signaling pathways mediated by cyclooxygenase-2 inhibitors in prostate cancer cells. Adv Enzyme Regul 41:221–235
Hsu AL, Ching TT, Wang DS et al (2000) The cyclooxygenale-2 inhibitor celecoxib induces apoptosis by blocking AKE activation in human prostate cancer cells independently of BC1-2. J Biochem 275:11397–11403
Liu X, Yue P, Zhou Z et al (2004) Death receptor regulation and celecoxib-induced apoptosis in human lung cancer cells. J Natl Cancer Inst 96:1769–1780
Jendrossek V, Handrick R, Belka C (2003) Celecoxib activates a novel mitochondrial apoptosis signaling pathway. FASEB J 17:1547–1549
Acknowledgement
This work was supported in part by a grant-in-aid from Chinese Medical Board. USA (no. 99-698).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Peng, HL., Zhang, GS., Liu, JH. et al. Dup-697, a specific COX-2 inhibitor, suppresses growth and induces apoptosis on K562 leukemia cells by cell-cycle arrest and caspase-8 activation. Ann Hematol 87, 121–129 (2008). https://doi.org/10.1007/s00277-007-0385-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-007-0385-4