Abstract
Purpose
To evaluate the efficiency of radiomics signatures in predicting the response of transarterial chemoembolization (TACE) therapy based on preoperative contrast-enhanced computed tomography (CECT).
Materials
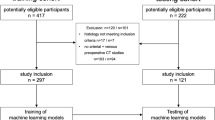
This study consisted of 111 patients with intermediate-stage hepatocellular carcinoma who underwent CECT at both the arterial phase (AP) and venous phase (VP) before and after TACE. According to mRECIST 1.1, patients were divided into an objective-response group (n = 38) and a non-response group (n = 73). Among them, 79 patients were assigned as the training dataset, and the remaining 32 cases were assigned as the test dataset.
Methods
Radiomics features were extracted from CECT images. Two feature ranking methods and three classifiers were used to find the best single-phase radiomics signatures for both AP and VP on the training set. Meanwhile, multi-phase radiomics signatures were built upon integration of images from two CECT phases by decision-level fusion and feature-level fusion. Finally, multivariable logistic regression was used to develop a nomogram by combining radiomics signatures and clinic-radiologic characteristics. The prediction performance was evaluated by AUC on the test dataset.
Results
The multi-phase radiomics signature (AUC = 0.883) performed better in predicting TACE therapy response compared to the best single-phase radiomics signature (AUC = 0.861). The nomogram (AUC = 0.913) showed better performance than any radiomics signatures.
Conclusion
The radiomics signatures and nomogram were developed and validated for predicting responses to TACE therapy, and the radiomics model may play a positive role in identifying patients who may benefit from TACE therapy in clinical practice.




Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424.
Villanueva A. Hepatocellular carcinoma. N Engl J Med. 2019;380(15):1450–62.
Chang Y, Jeong SW, Young JJ, Jae KY. Recent updates of transarterial chemoembolilzation in hepatocellular carcinoma. Int J Mol Sci. 2020;21(21):8165.
Qiu G, Jin Z, Chen X, Huang J. Interpretation of guidelines for the diagnosis and treatment of primary liver cancer (2019 edition) in China. Glob Health Med. 2020;2(5):306–11.
Haywood N, Gennaro K, Obert J, Sauer PF Jr, Redden DT, Zarzour J, et al. Does the degree of hepatocellular carcinoma tumor necrosis following transarterial chemoembolization impact patient survival? J Oncol. 2016;2016:4692139.
Lencioni R, Petruzzi P, Crocetti L. Chemoembolization of hepatocellular carcinoma. Semin Intervent Radiol. 2013;30(1):3–11.
Kudo M, Matsui O, Izumi N, Kadoya M, Okusaka T, Miyayama S, et al. Transarterial chemoembolization failure/refractoriness: JSH-LCSGJ criteria 2014 update. Oncology. 2014;87(Suppl 1):22–31.
Bruix J, Reig M, Sherman M. Evidence-based diagnosis, staging, and treatment of patients with hepatocellular carcinoma. Gastroenterology. 2016;150(4):835–53.
Kudo M. Proposal of primary endpoints for TACE combination trials with systemic therapy: lessons learned from 5 negative trials and the positive TACTICS trial. Liver Cancer. 2018;7(3):225–34.
Kloeckner R, Pitton MB, Dueber C, Schmidtmann I, Galle PR, Koch S, et al. Validation of clinical scoring systems ART and ABCR after transarterial chemoembolization of hepatocellular carcinoma. J Vasc Interv Radiol. 2017;28(1):94–102.
Chen L, Ni CF, Chen SX, Cao JM, Zhu HD, Guo JH, et al. A modified model for assessment for retreatment with transarterial chemoembolization in Chinese hepatocellular carcinoma patients. J Vasc Interv Radiol. 2016;27(9):1288–97.
Xia W, Hu B, Li H, Geng C, Wu Q, Yang L, et al. Multiparametric-MRI-based radiomics model for differentiating primary central nervous system lymphoma from glioblastoma: development and cross-vendor validation. J Magn Reson Imaging. 2021;53(1):242–50.
Meng X, Xia W, Xie P, Zhang R, Li W, Wang M, et al. Preoperative radiomic signature based on multiparametric magnetic resonance imaging for noninvasive evaluation of biological characteristics in rectal cancer. Eur Radiol. 2019;29(6):3200–9.
Xu X, Zhang HL, Liu QP, Sun SW, Zhang J, Zhu FP, et al. Radiomic analysis of contrast-enhanced CT predicts microvascular invasion and outcome in hepatocellular carcinoma. J Hepatol. 2019;70(6):1133–44.
Jian J, Li Y, Pickhardt PJ, Xia W, He Z, Zhang R, et al. MR image-based radiomics to differentiate type Iota and type IotaIota epithelial ovarian cancers. Eur Radiol. 2021;31(1):403–10.
Wang X, Zhao X, Li Q, Xia W, Peng Z, Zhang R, et al. Can peritumoral radiomics increase the efficiency of the prediction for lymph node metastasis in clinical stage T1 lung adenocarcinoma on CT? Eur Radiol. 2019;29(11):6049–58.
Sun Y, Bai H, Xia W, Wang D, Zhou B, Zhao X, et al. Predicting the outcome of transcatheter arterial embolization therapy for unresectable hepatocellular carcinoma based on radiomics of preoperative multiparameter MRI. J Magn Reson Imaging. 2020;52(4):1083–90.
Sieghart W, Hucke F, Peck-Radosavljevic M. Transarterial chemoembolization: modalities, indication, and patient selection. J Hepatol. 2015;62(5):1187–95.
Peng J, Kang S, Ning Z, Deng H, Shen J, Xu Y, et al. Residual convolutional neural network for predicting response of transarterial chemoembolization in hepatocellular carcinoma from CT imaging. Eur Radiol. 2020;30(1):413–24.
Chapiro J, Duran R, Lin M, Schernthaner RE, Wang Z, Gorodetski B, et al. Identifying staging markers for hepatocellular carcinoma before transarterial chemoembolization: comparison of three-dimensional quantitative versus non–three-dimensional imaging markers. Radiology. 2015;275(2):438–47.
Zhao W, Yang J, Sun Y, Li C, Wu W, Jin L, et al. 3D deep learning from CT scans predicts tumor invasiveness of subcentimeter pulmonary adenocarcinomas. Cancer Res. 2018;78(24):6881–9.
Zhao X, Wang X, Xia W, Li Q, Zhou L, Li Q, et al. A cross-modal 3D deep learning for accurate lymph node metastasis prediction in clinical stage T1 lung adenocarcinoma. Lung Cancer. 2020;145:10–7.
Liu D, Liu F, Xie X, Su L, Liu M, Xie X, et al. Accurate prediction of responses to transarterial chemoembolization for patients with hepatocellular carcinoma by using artificial intelligence in contrast-enhanced ultrasound. Eur Radiol. 2020;30(4):2365–76.
Van Griethuysen JJ, Fedorov A, Parmar C, Hosny A, Aucoin N, Narayan V, et al. Computational radiomics system to decode the radiographic phenotype. Cancer Res. 2017;77(21):e104–7.
Chang Y-W, Lin C-J, editors. Feature ranking using linear SVM. Causation and prediction challenge; 2008: PMLR.
Parmar C, Grossmann P, Bussink J, Lambin P, Aerts HJJSr. Machine learning methods for quantitative radiomic biomarkers. Sci Rep. 2015;5:13087.
Nie K, Shi L, Chen Q, Hu X, Jabbour SK, Yue N, et al. Rectal cancer: assessment of neoadjuvant chemoradiation outcome based on radiomics of multiparametric MRI Prediction of pathological response for LARC Using MRI. Clin Cancer Res. 2016;22(21):5256–64.
Qi Y. Random forest for bioinformatics. Ensemble machine learning. Springer; 2012. p. 307–23.
Fernández-Delgado M, Cernadas E, Barro S, Amorim DJT. Do we need hundreds of classifiers to solve real world classification problems? J Mach Learn Res. 2014;15(1):3133–81.
Huang Y-q, Liang C-h, He L, Tian J, Liang C-s, Chen X, et al. Development and validation of a radiomics nomogram for preoperative prediction of lymph node metastasis in colorectal cancer. J Clin Oncol. 2016;34(18):2157–64.
Gunatilaka AH, Baertlein BAJ. Feature-level and decision-level fusion of noncoincidently sampled sensors for land mine detection. IEEE Transact Pattern Anal Mach Intell. 2001;23(6):577–89.
Planet S, Iriondo I, editors. Comparison between decision-level and feature-level fusion of acoustic and linguistic features for spontaneous emotion recognition. In: 7th Iberian conference on information systems and technologies (CISTI 2012) ; 2012: IEEE.
Hanley JA, McNeil BJJR. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology. 1982;143(1):29–36.
Huang Y, Liu Z, He L, Chen X, Pan D, Ma Z, et al. Radiomics signature: a potential biomarker for the prediction of disease-free survival in early-stage (I or II) non—small cell lung cancer. 2016;281(3) :947–57.
Mundra PA. Rajapakse JCJIton. SVM-RFE with MRMR filter for gene selection. 2009;9(1):31–7.
Ramírez-Gallego S, Lastra I, Martínez-Rego D, Bolón-Canedo V, Benítez JM, Herrera F, et al. Fast-mRMR: fast minimum redundancy maximum relevance algorithm for high-dimensional big data. Int J Intell Syst. 2017;32(2):134–52.
Belgiu M, Drăguţ LJI. Random forest in remote sensing: a review of applications and future directions. J Photogramm Remote Sens. 2016;114:24–31.
Cherkassky V, Ma Y. Practical selection of SVM parameters and noise estimation for SVM regression. J Neural Netw. 2004;17(1):113–26.
Zou HJ. The adaptive lasso and its oracle properties. J Am Stat Assoc. 2006;101(476):1418–29.
Yasui Y, Tsuchiya K, Kurosaki M, Takeguchi T, Takeguchi Y, Okada M, et al. Up-to-seven criteria as a useful predictor for tumor downstaging to within Milan criteria and Child-Pugh grade deterioration after initial conventional transarterial chemoembolization. Hepatol Res. 2018;48(6):442–50.
Wang Q, Xia D, Bai W, Wang E, Sun J, Huang M, et al. Development of a prognostic score for recommended TACE candidates with hepatocellular carcinoma: a multicentre observational study. J Hepatol. 2019;70(5):893–903.
Tacher V, Lin M, Duran R, Yarmohammadi H, Lee H, Chapiro J, et al. Comparison of existing response criteria in patients with hepatocellular carcinoma treated with transarterial chemoembolization using a 3D quantitative approach. Radiology. 2016;278(1):275–84.
Funding
The Key Research and Development Program of Shandong Province (Grant Number 2021SFGC0104). The Key Research and Development Program of Jiangsu [Grant Number: BE2021663]. The National Natural Science Foundation of China [Grant Number: 81871439]. The Science and Technology Planning Project of Suzhou [Grant Number: SJC2021014]. Jiangsu Province Engineering Research Center of Diagnosis and Treatment of Children’s Malignant Tumor.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
Institutional Review Board approval was obtained in concordance with the standards of the First Affiliated Hospital of Soochow University Ethics Committee (2020; approval no. 064). All procedures performed in this study, which involved human participants, were in accordance with the ethical standards of the institutional and/or nation research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Consent for Publication
The manuscript, or part of it, have not been and will not be submitted elsewhere for publication.
Informed Consent
Approved by the ethics committee of the First Affiliated Hospital of Soochow University Ethics Committee, this retrospective analysis was involving CECT of patients in and informed consent was waived for all patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bai, H., Meng, S., Xiong, C. et al. Preoperative CECT-based Radiomic Signature for Predicting the Response of Transarterial Chemoembolization (TACE) Therapy in Hepatocellular Carcinoma. Cardiovasc Intervent Radiol 45, 1524–1533 (2022). https://doi.org/10.1007/s00270-022-03221-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-022-03221-z