Abstract
Purpose
Unresectable intrahepatic cholangiocarcinoma (ICC) signifies a poor prognosis with limited treatment options beyond systemic chemotherapy. This study’s purpose was to evaluate the safety, efficacy, and potential for downstaging to resection of yttrium-90 (Y90) radioembolization for treatment of unresectable ICC.
Materials and Methods
From 2004 to 2020, 136 patients with unresectable ICC were treated with radioembolization at a single institution. Retrospective review was performed of a prospectively collected database. Outcomes were (1) biochemical and clinical toxicities, (2) local tumor response, (3) time to progression, and (4) overall survival (OS) after Y90. Univariate/multivariate survival analyses were performed. A subgroup analysis was performed to calculate post-resection recurrence and OS in patients downstaged to resection after Y90.
Results
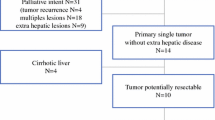
Grade 3+ clinical and biochemical toxicities were 7.6% (n = 10) and 4.9% (n = 6), respectively. Best index lesion response was complete response in 2 (1.5%), partial response in 42 (32.1%), stable disease in 82 (62.6%), and progressive disease in 5 (3.8%) patients. Median OS was 14.2 months. Solitary tumor (P < 0.001), absence of vascular involvement (P = 0.009), and higher serum albumin (P < 0.001) were independently associated with improved OS. Eleven patients (8.1%) were downstaged to resection and 2 patients (1.5%) were bridged to transplant. R0-resection was achieved in 8/11 (72.7%). Post-resection median recurrence and OS were 26.3 months and 39.9 months, respectively.
Conclusion
Y90 has an acceptable safety profile and high local disease control rates for the treatment of unresectable ICC. Downstaging to resection with > 3 years survival supports the therapeutic role of Y90 for unresectable ICC.
Level of Evidence: Level 3, single-arm single-center cohort study.



Similar content being viewed by others
Abbreviations
- CA:
-
Carbohydrate antigen
- CT:
-
Computed tomography
- ICC:
-
Intrahepatic cholangiocarcinoma
- MR:
-
Magnetic resonance
- OS:
-
Overall survival
- TTP:
-
Time to progression
- WHO:
-
World Health Organization
- Y90:
-
Yttrium-90
References
Tang R, Latchana N, Rahnemai-Azar AA, Pawlik TM. Guidelines for Resection of Intrahepatic Cholangiocarcinoma. In: Cardona K, Maithel SK, editors. Primary and Metastatic Liver Tumors: Treatment Strategy and Evolving Therapies. Cham: Springer International Publishing; 2018. p. 99–110.
Banales JM, Marin JJG, Lamarca A, et al. Cholangiocarcinoma 2020: the next horizon in mechanisms and management. Nat Rev Gastroenterol Hepatol. 2020;17(9):557–88.
Bridgewater J, Galle PR, Khan SA, et al. Guidelines for the diagnosis and management of intrahepatic cholangiocarcinoma. J Hepatol. 2014;60(6):1268–89.
Lamarca A, Ross P, Wasan HS, et al. Advanced Intrahepatic Cholangiocarcinoma: Post Hoc Analysis of the ABC-01, -02, and -03 Clinical Trials. J Natl Cancer Inst. 2020;112(2):200–10.
Javle MM, Roychowdhury S, Kelley RK, et al. (2021) Final results from a phase II study of infigratinib (BGJ398), an FGFR-selective tyrosine kinase inhibitor, in patients with previously treated advanced cholangiocarcinoma harboring an FGFR2 gene fusion or rearrangement. Journal of Clinical Oncology, 39(3_suppl):265–265
Park J, Kim MH, Kim KP, et al. Natural History and Prognostic Factors of Advanced Cholangiocarcinoma without Surgery, Chemotherapy, or Radiotherapy: A Large-Scale Observational Study. Gut Liver. 2009;3(4):298–305.
Valle JW, Borbath I, Khan SA, et al. Biliary cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2016;27(suppl 5):v28–37.
National Comprehensive Cancer Network (2020) NCCN Clinical Practice Guidelines in Oncology; Hepatobiliary Cancers (Version 5.2020),
Mosconi C, Solaini L, Vara G, et al. Transarterial Chemoembolization and Radioembolization for Unresectable Intrahepatic Cholangiocarcinoma-a Systemic Review and Meta-Analysis. Cardiovasc Intervent Radiol. 2021;44(5):728–38.
White J, Carolan-Rees G, Dale M, et al. Yttrium-90 Transarterial Radioembolization for Chemotherapy-Refractory Intrahepatic Cholangiocarcinoma: A Prospective. Observational Study J Vasc Interv Radiol. 2019;30(8):1185–92.
Edeline J, Touchefeu Y, Guiu B, et al. (2019) Radioembolization Plus Chemotherapy for First-line Treatment of Locally Advanced Intrahepatic Cholangiocarcinoma: A Phase 2 Clinical Trial. JAMA Oncol
Ibrahim SM, Mulcahy MF, Lewandowski RJ, et al. Treatment of unresectable cholangiocarcinoma using yttrium-90 microspheres: results from a pilot study. Cancer. 2008;113(8):2119–28.
Mouli S, Memon K, Baker T, et al. Yttrium-90 radioembolization for intrahepatic cholangiocarcinoma: safety, response, and survival analysis. J Vasc Interv Radiol. 2013;24(8):1227–34.
Salem R, Thurston KG (2006) Radioembolization with 90Yttrium microspheres: a state-of-the-art brachytherapy treatment for primary and secondary liver malignancies. Part 1: Technical and methodologic considerations. J Vasc Interv Radiol, 17(8):1251–1278
Salem R, Lewandowski RJ, Atassi B, et al. Treatment of unresectable hepatocellular carcinoma with use of 90Y microspheres (TheraSphere): safety, tumor response, and survival. J Vasc Interv Radiol. 2005;16(12):1627–39.
Salem R, Thurston KG (2006) Radioembolization with 90yttrium microspheres: a state-of-the-art brachytherapy treatment for primary and secondary liver malignancies. Part 2: special topics. J Vasc Interv Radiol, 17(9):1425–1439
Chung YE, Kim MJ, Park YN, et al. Varying appearances of cholangiocarcinoma: radiologic-pathologic correlation. Radiographics. 2009;29(3):683–700.
Fabrega-Foster K, Ghasabeh MA, Pawlik TM, Kamel IR. Multimodality imaging of intrahepatic cholangiocarcinoma. Hepatobiliary Surg Nutr. 2017;6(2):67–78.
Riaz A, Miller FH, Kulik LM, et al. Imaging response in the primary index lesion and clinical outcomes following transarterial locoregional therapy for hepatocellular carcinoma. JAMA. 2010;303(11):1062–9.
Patel AH, Harnois DM, Klee GG, LaRusso NF, Gores GJ. The utility of CA 19–9 in the diagnoses of cholangiocarcinoma in patients without primary sclerosing cholangitis. Am J Gastroenterol. 2000;95(1):204–7.
Edeline J, Du FL, Rayar M, et al. Glass Microspheres 90Y Selective Internal Radiation Therapy and Chemotherapy as First-Line Treatment of Intrahepatic Cholangiocarcinoma. Clin Nucl Med. 2015;40(11):851–5.
Levillain H, Duran Derijckere I, Ameye L, et al. Personalised radioembolization improves outcomes in refractory intra-hepatic cholangiocarcinoma: a multicenter study. Eur J Nucl Med Mol Imaging. 2019;46(11):2270–9.
Bargellini I, Mosconi C, Pizzi G, et al. Yttrium-90 Radioembolization in Unresectable Intrahepatic Cholangiocarcinoma: Results of a Multicenter Retrospective Study. Cardiovasc Intervent Radiol. 2020;43(9):1305–14.
Helmberger T, Golfieri R, Pech M, et al. Clinical Application of Trans-Arterial Radioembolization in Hepatic Malignancies in Europe: First Results from the Prospective Multicentre Observational Study CIRSE Registry for SIR-Spheres Therapy (CIRT). Cardiovasc Intervent Radiol. 2021;44(1):21–35.
Wu L, Tsilimigras DI, Farooq A, et al. Potential survival benefit of radiofrequency ablation for small solitary intrahepatic cholangiocarcinoma in nonsurgically managed patients: A population-based analysis. J Surg Oncol. 2019;120(8):1358–64.
Xiang X, Hu D, Jin Z, Liu P, Lin H (2020) Radiofrequency Ablation vs. Surgical Resection for Small Early-Stage Primary Intrahepatic Cholangiocarcinoma. Front Oncol, 10:540662
Gaba RC, Lewandowski RJ, Kulik LM, et al. Radiation lobectomy: preliminary findings of hepatic volumetric response to lobar yttrium-90 radioembolization. Ann Surg Oncol. 2009;16(6):1587–96.
Mavros MN, Economopoulos KP, Alexiou VG, Pawlik TM. Treatment and Prognosis for Patients With Intrahepatic Cholangiocarcinoma: Systematic Review and Meta-analysis. JAMA Surg. 2014;149(6):565–74.
Bourien H, Palard X, Rolland Y, et al. Yttrium-90 glass microspheres radioembolization (RE) for biliary tract cancer: a large single-center experience. Eur J Nucl Med Mol Imaging. 2019;46(3):669–76.
Riby D, Mazzotta AD, Bergeat D, et al. Downstaging with Radioembolization or Chemotherapy for Initially Unresectable Intrahepatic Cholangiocarcinoma. Ann Surg Oncol. 2020;27(10):3729–37.
Hyder O, Hatzaras I, Sotiropoulos GC, et al. Recurrence after operative management of intrahepatic cholangiocarcinoma. Surgery. 2013;153(6):811–8.
Kohler M, Harders F, Lohofer F, et al. (2019) Prognostic Factors for Overall Survival in Advanced Intrahepatic Cholangiocarcinoma Treated with Yttrium-90 Radioembolization. J Clin Med, 9(1)
Mosconi C, Gramenzi A, Ascanio S, et al. Yttrium-90 radioembolization for unresectable/recurrent intrahepatic cholangiocarcinoma: a survival, efficacy and safety study. Br J Cancer. 2016;115(3):297–302.
Gangi A, Shah J, Hatfield N, et al. Intrahepatic Cholangiocarcinoma Treated with Transarterial Yttrium-90 Glass Microsphere Radioembolization: Results of a Single Institution Retrospective Study. J Vasc Interv Radiol. 2018;29(8):1101–8.
Shroff RT, Javle MM, Xiao L, et al. Gemcitabine, Cisplatin, and nab-Paclitaxel for the Treatment of Advanced Biliary Tract Cancers: A Phase 2 Clinical Trial. JAMA Oncol. 2019;5(6):824–30.
Acknowledgements
The authors thank Vanessa L. Gates, Karen Grace, Krystina Salzig, Melissa Williams, and Carlene del Castillo for their compassionate care of patients and dedication to clinical research.
Funding
This study was not supported by any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A.C.G. consults for ABK Biomedical Inc. A.D.Y. receives support from the National Heart, Lung, and Blood Institute award K08HL145139. A.R. consults for Boston Scientific. L.K. is a speaker for Gilead Sciences and Eisai and an advisor for Roche, Genentech, and Exelixis. A.B.B. is a consultant/advisor for Merck Sharp & Dohme, Array BioPharma, Bristol-Myers Squibb, Samsung Bioepis, Pfizer, HalioDx, Abbvie, Janssen Oncology, Natera, Apexigen, Artemida, Xencor, and Therabionic and receives research funding from Infinity Pharmaceuticals, Merck Sharp & Dohme, Taiho Pharmaceutical, Bristol-Myers Squibb, Celgene, Rafael Pharmaceuticals, MedImmune, Xencor, Astellas Pharma, Amgen, Syncore, Elevar Therapeutics, Tyme Inc, ST Pharm, and ITM Solucin GmbH. R.S. consults for Boston Scientific, Astrazeneca, Genentech, Sirtex, Cook, Eisai, Bard, and QED Therapeutics. R.J.L. is an adviser and on speaker bureau for Boston Scientific, consultant for Bard, Varian, ABK Medical, Alhambra Medical, and receives research support from National Institutes of Health grant R01CA233878-01. All remaining authors have declared no conflicts of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
Consent for publication was obtained for every individual person’s data included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gupta, A.N., Gordon, A.C., Gabr, A. et al. Yttrium-90 Radioembolization of Unresectable Intrahepatic Cholangiocarcinoma: Long-Term Follow-up for a 136-Patient Cohort. Cardiovasc Intervent Radiol 45, 1117–1128 (2022). https://doi.org/10.1007/s00270-022-03183-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-022-03183-2