Abstract
Purpose
To investigate the potential role for CD44+ and CD90+ hepatocellular carcinoma (HCC) cellular subpopulations in biological response to thermal ablation-induced heat stress.
Methods
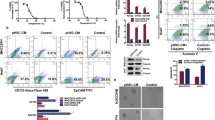
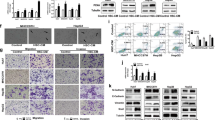
This study was approved by the institutional animal care committee. The N1S1 rat HCC cell line was subjected to sublethal heat stress (45 °C) or control (37 °C) for 10 min, costained with fluorescent-labeled antibodies against CD44, CD90, and 7-AAD after a 48-h recovery and analyzed by flow cytometry to assess the percentage of live CD44+ and CD90+ HCC cells (n = 4). Experiments were repeated with pretreatment of N1S1 cells with a dose titration of the dual PI3K-mTOR inhibitor BEZ235 or vehicle control (n = 3). Rats bearing orthotopic N1S1 tumors were subjected to ultrasound-guided partial laser ablation (n = 5) or sham ablation (n = 3), euthanized 24 h after ablation, and liver/tumor analyzed for immunohistochemical staining of CD44 and CD90. Differences between groups were compared with an unpaired t test.
Results
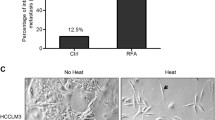
Sublethal heat stress induced a significant increase in the relative proportion of live CD44+ and CD90+ HCC cells compared to the control group: CD44+CD90− (5.3-fold; p = 0.0001), CD44−CD90+ (2.4-fold; p = 0.003), and CD44+CD90+ (22.0-fold; p < 0.03). Inhibition of PI3K-mTOR prevented heat stress-induced enrichment of the population of live CD44+ HCC cells (p < 0.01), but not CD90+ cells (p > 0.10). Immunohistochemical analysis demonstrated preferential localization of clusters of CD44+ cells at both the tumor margin and ablation margin.
Conclusion
These studies provide experimental evidence supporting a role for HCC cells expressing the putative stem cell marker CD44 in HCC response to heat stress.





Similar content being viewed by others
References
Jordan CT, Guzman ML, Noble M (2006) Cancer stem cells. N Engl J Med 355:1253–1261
Hamburger AW, Salmon SE (1977) Primary bioassay of human tumor stem cells. Science 197(4302):461–463
Reya T, Morrison SJ, Clarke MF, Weissman IL (2001) Stem cells, cancer, and cancer stem cells. Nature 414(6859):105–111
Visvader JE, Lindeman GJ (2008) Cancer stem cells in solid tumours: accumulating evidence and unresolved questions. Nat Rev Cancer 8:755–768
Magee JA, Piskounova E, Morrison SJ (2012) Cancer stem cells: impact, heterogeneity, and uncertainty. Cancer Cell 21:283–296
Brabletz T (2012) EMT and MET in metastasis: where are the cancer stem cells? Cancer Cell 22:699–701
Park CY, Tseng D, Weissman IL (2009) Cancer stem cell-directed therapies: recent data from the laboratory and clinic. Mol Ther 17:219–230
Rosen JM, Jordan CT (2009) The increasing complexity of the cancer stem cell paradigm. Science 324(5935):1670–1673
Tong CM, Ma S, Guan XY (2011) Biology of hepatic cancer stem cells. J Gastroenterol Hepatol 26:1229–1237
Haraguchi N, Ishii H, Mimori K et al (2010) CD13 is a therapeutic target in human liver cancer stem cells. J Clin Investig 120:3326–3339
Henry JC, Park JK, Jiang J et al (2010) miR-199a-3p targets CD44 and reduces proliferation of CD44 positive hepatocellular carcinoma cell lines. Biochem Biophys Res Commun 403:120–125
Yang ZF, Ho DW, Ng MN et al (2008) Significance of CD90+ cancer stem cells in human liver cancer. Cancer Cell 13:153–166
Ma S, Lee TK, Zheng BJ et al (2008) CD133+ HCC cancer stem cells confer chemoresistance by preferential expression of the Akt/PKB survival pathway. Oncogene 27:1749–1758
Ma S, Chan KW, Lee TK et al (2008) Aldehyde dehydrogenase discriminates the CD133 liver cancer stem cell populations. Mol Cancer Res 6:1146–1153
Yamashita T, Forgues M, Wang W et al (2008) EpCAM and alpha-fetoprotein expression defines novel prognostic subtypes of hepatocellular carcinoma. Cancer Res 68:1451–1461
Terris B, Cavard C, Perret C (2010) EpCAM, a new marker for cancer stem cells in hepatocellular carcinoma. J Hepatol 52:280–281
Yoon SK (2012) The biology of cancer stem cells and its clinical implication in hepatocellular carcinoma. Gut Liver 6:29–40
Ji J, Wang XW (2012) Clinical implications of cancer stem cell biology in hepatocellular carcinoma. Semin Oncol 39:461–472
Rountree CB, Mishra L, Willenbring H (2012) Stem cells in liver diseases and cancer: recent advances on the path to new therapies. Hepatology 55:298–306
Colombo F, Baldan F, Mazzucchelli S et al (2011) Evidence of distinct tumour-propagating cell populations with different properties in primary human hepatocellular carcinoma. PLoS One 6:e21369
Marquardt JU, Raggi C, Andersen JB et al (2011) Human hepatic cancer stem cells are characterized by common stemness traits and diverse oncogenic pathways. Hepatology 54:1031–1042
Song K, Wu J, Jiang C (2013) Dysregulation of signaling pathways and putative biomarkers in liver cancer stem cells [review]. Oncol Rep 29:3–12
Chen X, Lingala S, Khoobyari S et al (2011) Epithelial mesenchymal transition and hedgehog signaling activation are associated with chemoresistance and invasion of hepatoma subpopulations. J Hepatol 55:838–845
Lee TK, Castilho A, Ma S, Ng IO (2009) Liver cancer stem cells: implications for a new therapeutic target. Liver Int 29:955–965
Martelli AM, Evangelisti C, Follo MY et al (2011) Targeting the phosphatidylinositol 3-kinase/Akt/mammalian target of rapamycin signaling network in cancer stem cells. Curr Med Chem 18:2715–2726
Dubrovska A, Kim S, Salamone RJ et al (2009) The role of PTEN/Akt/PI3K signaling in the maintenance and viability of prostate cancer stem-like cell populations. Proc Natl Acad Sci USA 106:268–273
Sunayama J, Matsuda K, Sato A et al (2010) Crosstalk between the PI3K/mTOR and MEK/ERK pathways involved in the maintenance of self-renewal and tumorigenicity of glioblastoma stem-like cells. Stem Cells 28:1930–1939
Xu Q, Simpson SE, Scialla TJ et al (2003) Survival of acute myeloid leukemia cells requires PI3 kinase activation. Blood 102:972–980
Xu Q, Thompson JE, Carroll M (2005) mTOR regulates cell survival after etoposide treatment in primary AML cells. Blood 106:4261–4268
Hsu HS, Lin JH, Huang WC et al (2011) Chemoresistance of lung cancer stemlike cells depends on activation of Hsp27. Cancer 117:1516–1528
Prinsloo E, Setati MM, Longshaw VM, Blatch GL (2009) Chaperoning stem cells: a role for heat shock proteins in the modulation of stem cell self-renewal and differentiation? Bioessays 31:370–377
Wright MH, Calcagno AM, Salcido CD et al (2008) Brca1 breast tumors contain distinct CD44+/CD24−; and CD133+ cells with cancer stem cell characteristics. Breast Cancer Res 10:R10
Lencioni R, Crocetti L (2012) Local-regional treatment of hepatocellular carcinoma. Radiology 262:43–58
Tiong L, Maddern GJ (2011) Systematic review and meta-analysis of survival and disease recurrence after radiofrequency ablation for hepatocellular carcinoma. Br J Surg 98:1210–1224
Wang JH, Wang CC, Hung CH et al (2012) Survival comparison between surgical resection and radiofrequency ablation for patients in BCLC very early/early stage hepatocellular carcinoma. J Hepatol 56:412–418
Cho YK, Kim JK, Kim MY et al (2009) Systematic review of randomized trials for hepatocellular carcinoma treated with percutaneous ablation therapies. Hepatology 49:453–459
Yin XY, Xie XY, Lu MD et al (2009) Percutaneous thermal ablation of medium and large hepatocellular carcinoma: long-term outcome and prognostic factors. Cancer 115:1914–1923
Kao WY, Chiou YY, Hung HH et al (2011) Risk factors for long-term prognosis in hepatocellular carcinoma after radiofrequency ablation therapy: the clinical implication of aspartate aminotransferase–platelet ratio index. Eur J Gastroenterol Hepatol 23:528–536
Ng KK, Poon RT, Lo CM et al (2008) Analysis of recurrence pattern and its influence on survival outcome after radiofrequency ablation of hepatocellular carcinoma. J Gastrointest Surg 12:183–191
Liu Y, Zheng Y, Li S et al (2013) Percutaneous microwave ablation of larger hepatocellular carcinoma. Clin Radiol 68:21–26
Thompson SM, Callstrom MR, Knudsen B et al (2012) Development and preliminary testing of a translational model of hepatocellular carcinoma for MR imaging and interventional oncologic investigations. J Vasc Interv Radiol 23:385–395
Thompson SM, Callstrom MR, Knudsen B et al (2013) AS30D model of hepatocellular carcinoma: tumorigenicity and preliminary characterization by imaging, histopathology, and immunohistochemistry. Cardiovasc Intervent Radiol 36:198–203
Yamashita T, Honda M, Nio K et al (2010) Oncostatin m renders epithelial cell adhesion molecule-positive liver cancer stem cells sensitive to 5-fluorouracil by inducing hepatocytic differentiation. Cancer Res 70:4687–4697
Wang XQ, Ongkeko WM, Chen L et al (2010) Octamer 4 (Oct4) mediates chemotherapeutic drug resistance in liver cancer cells through a potential Oct4-AKT-ATP-binding cassette G2 pathway. Hepatology 52:528–539
Cheung ST, Cheung PF, Cheng CK et al (2011) Granulin-epithelin precursor and ATP-dependent binding cassette (ABC)B5 regulate liver cancer cell chemoresistance. Gastroenterology 140:344–355
Lingala S, Cui YY, Chen X et al (2010) Immunohistochemical staining of cancer stem cell markers in hepatocellular carcinoma. Exp Mol Pathol 89:27–35
Hermansen SK, Christensen KG, Jensen SS, Kristensen BW (2011) Inconsistent immunohistochemical expression patterns of four different CD133 antibody clones in glioblastoma. J Histochem Cytochem 59:391–407
Mather JP (2012) In vitro models. Stem Cells 30:95–99
Acknowledgments
This publication was supported by CTSA Grant TL1 TR000137 from the National Center for Advancing Translational Science (NCATS). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH. Additional research support provided in part by a SIR Foundation allied scientist training Grant. Infrastructure support was provided by NIH construction grant NIH C06 RR018898.
Conflict of interest
Mr. Scott Thompson received a research grant from the SIR Foundation—Allied Scientist Training Grant. Dr. Lewis Roberts received research grants from Bristol Myers-Squibb, Merck, Nordion, and Bayer. Dr. Matthew Callstrom, Ms. Kim Butters, Ms. Shari Sutor, Mr. Bruce Knudsen, Dr. Joseph Grande, and Dr. David Woodrum declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thompson, S.M., Callstrom, M.R., Butters, K.A. et al. Role for Putative Hepatocellular Carcinoma Stem Cell Subpopulations in Biological Response to Incomplete Thermal Ablation: In Vitro and In Vivo Pilot Study. Cardiovasc Intervent Radiol 37, 1343–1351 (2014). https://doi.org/10.1007/s00270-013-0828-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-013-0828-3