Abstract
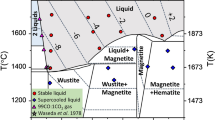
Thermodynamic models which incorporate the effects of long-range magnetic order are developed for mineral endmembers and their solid solutions. The temperature dependence of the magnetic entropy of solution is obtained from a statistical mechanical description of spin configurations. The non-configurational magnetic Gibbs Free energy is modeled as a Taylor expansion of low order and even degree. Taylor coefficients are calibrated from determinations of magnetic transition temperatures and from heat capacity measurements. The technique is used to derived model equations for the thermodynamic properties of magnetite (Fe3O4), hematite (Fe2O3) and the solid solution series magnetite-ulvöspinel (Fe2TiO4) and hematite-ilmenite (FeTiO3). These models are utilized to calculate phase diagrams, and the effect of the interplay between “magnetic” and “chemical” ordering is explored. A petrologic application showing the influence of magnetic ordering on temperatures derived from the Fe−Ti oxide geothermometer demonstrates that the effect is significant (a correction on the order of 100 °C) at derived temperatures below the Néel point for the cubic phase.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 9 October 1996 / Revised, accepted: 2 April 1997
Rights and permissions
About this article
Cite this article
Ghiorso, M. Thermodynamic analysis of the effect of magnetic ordering on miscibility gaps in the Fe−Ti cubic and rhombohedral oxide minerals and the Fe−Ti oxide geothermometer. Phys Chem Min 25, 28–38 (1997). https://doi.org/10.1007/s002690050083
Issue Date:
DOI: https://doi.org/10.1007/s002690050083