Abstract
Background
There are only limited data on the failure patterns after surgical resection for duodenal cancer, and the role of adjuvant chemoradiotherapy (CRT) also remains controversial. In this study, the treatment outcomes of surgery alone were compared to those of surgery plus adjuvant CRT for duodenal cancer.
Methods
Between January 1991 and February 2013, a total of 47 patients with duodenal cancer had pancreaticoduodenectomy, and their age ranged from 31 to 80 (median 62). Twenty-five patients (53%) underwent surgery alone, while 22 (47%) underwent surgery plus adjuvant CRT. Postoperative radiotherapy with concomitant 5-fluorouracil was given to tumor bed and regional lymph nodes up to 40–55.4 Gy. Median duration of follow-up was 31 months (range 6–286) for all patients and 90 months (range 14–286) for survivors.
Results
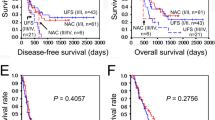
CRT (+) group included more patients with advanced nodal stage and overall stage group (p = 0.003 and 0.002, respectively). The 5-year overall survival rates were not different between CRT (−) and CRT (+) groups (50.1 vs. 46.7%, p = 0.794). CRT (+) group achieved a superior 5-year loco-regional relapse-free survival rate compared with CRT (−) group, but the difference did not reach a statistical significance (80.1 vs. 68.4%, p = 0.267). On multivariate analysis, however, the addition of CRT was the only favorable prognosticator predicting loco-regional relapse-free survival (p = 0.046). Two patients experienced grade 3 neutropenia during CRT.
Conclusions
Adjuvant CRT after pancreaticoduodenectomy was correlated with an improved loco-regional control in duodenal cancer. Considering the high loco-regional recurrence in surgery alone group, CRT may be considered as adjuvant treatment.


Similar content being viewed by others
References
Siegel RL, Miller KD, Jemal A (2016) Cancer statistics, 2016. CA Cancer J Clin 66:7–30
Hatzaras I, Palesty JA, Abir F et al (2007) Small-bowel tumors: epidemiologic and clinical characteristics of 1260 cases from the connecticut tumor registry. Arch Surg 142:229–235
Bilimoria KY, Bentrem DJ, Wayne JD et al (2009) Small bowel cancer in the United States: changes in epidemiology, treatment, and survival over the last 20 years. Ann Surg 249:63–71
Halfdanarson TR, McWilliams RR, Donohue JH et al (2010) A single-institution experience with 491 cases of small bowel adenocarcinoma. Am J Surg 199:797–803
Singhal N, Singhal D (2007) Adjuvant chemotherapy for small intestine adenocarcinoma. Cochrane Libr
Young JI, Mongoue-Tchokote S, Wieghard N et al (2016) Treatment and survival of small-bowel adenocarcinoma in the United States: a comparison with colon cancer. Dis Colon Rectum 59:306–315
Bakaeen FG, Murr MM, Sarr MG et al (2000) What prognostic factors are important in duodenal adenocarcinoma? Arch Surg 135:635–642
Kelsey CR, Nelson JW, Willett CG et al (2007) Duodenal adenocarcinoma: patterns of failure after resection and the role of chemoradiotherapy. Int J Radiat Oncol Biol Phys 69:1436–1441
Poultsides GA, Huang LC, Cameron JL et al (2012) Duodenal adenocarcinoma: clinicopathologic analysis and implications for treatment. Ann Surg Oncol 19:1928–1935
Kim K, Chie EK, Jang J-Y et al (2012) Role of adjuvant chemoradiotherapy for duodenal cancer: a single center experience. Am J Clin Oncol 35:533–536
Edge SB, Byrd DR, Compton CC et al (eds) (2010) AJCC cancer staging manual, 7th edn. Springer, Berlin
Moon YW, Rha SY, Shin SJ et al (2010) Adenocarcinoma of the small bowel at a single Korean institute: management and prognosticators. J Cancer Res Clin Oncol 136:387–394
Dabaja BS, Suki D, Pro B et al (2004) Adenocarcinoma of the small bowel. Cancer 101:518–526
Struck A, Howard T, Chiorean EG et al (2009) Non-ampullary duodenal adenocarcinoma: factors important for relapse and survival. J Surg Oncol 100:144–148
Swartz MJ, Hughes MA, Frassica DA et al (2007) Adjuvant concurrent chemoradiation for node-positive adenocarcinoma of the duodenum. Arch Surg 142:285–288
Manfredi S, Thiebot T, Henno S et al (2009) Complete response of an initially non-surgical adenocarcinoma of the duodenum to chemotherapy with the FOLFOX 4 regimen. J Gastrointest Surg 13:2309
Catania C, Pelosi G, Fazio N et al (2010) A FOLFIRI-induced complete tumor response in a patient with FOLFOX-refractory metastatic duodenal adenocarcinoma. Acta Oncol 49:123–124
Ecker BL, McMillan MT, Datta J et al (2017) Adjuvant chemotherapy versus chemoradiotherapy in the management of patients with surgically resected duodenal adenocarcinoma: a propensity score-matched analysis of a nationwide clinical oncology database. Cancer 123:967–976
Hammel P, Huguet F, van Laethem J-L et al (2016) Effect of chemoradiotherapy vs chemotherapy on survival in patients with locally advanced pancreatic cancer controlled after 4 months of gemcitabine with or without erlotinib: the LAP07 randomized clinical trial. JAMA 315:1844–1853
Fuchs CS, Niedzwiecki D, Mamon HJ et al (2017) Adjuvant chemoradiotherapy with epirubicin, cisplatin, and fluorouracil compared with adjuvant chemoradiotherapy with fluorouracil and leucovorin after curative resection of gastric cancer: results from CALGB 80101 (alliance). J Clin Oncol 35:3671–3677
National Comprehensive Cancer Network (2018). Gastric cancer (version 5.2017). https://www.nccn.org/professionals/physician_gls/pdf/gastric.pdf. Assessed 1 Jan 2018
Vittinghoff E, McCulloch CE (2007) Relaxing the rule of ten events per variable in logistic and Cox regression. Am J Epidemiol 165:710–718
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All the authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Jang, BS., Park, H.J., Kim, K. et al. Role of Adjuvant Chemoradiotherapy for Duodenal Cancer: An Updated Analysis of Long-Term Follow-Up from Single Institution. World J Surg 42, 3294–3301 (2018). https://doi.org/10.1007/s00268-018-4692-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-018-4692-9