Abstract
Background
Despite significant improvements in perioperative mortality as well as response rates to multimodality treatment, results after surgical resection of pancreatic adenocarcinoma with respect to long-term outcomes remain disappointing. Patient recruitment for prospective international trials on adjuvant and neoadjuvant regimens is challenging for various reasons. We set out to assess the preconditions and potential to perform perioperative trials for pancreatic cancer within a well-established Austrian nationwide network of surgical and medical oncologists (Austrian Breast & Colorectal Cancer Study Group).
Methods
From 2005 to 2010 five high-volume centers and one medium-volume center completed standardized data entry forms with 33 parameters (history and patient related data, preoperative clinical staging and work-up, surgical details and intraoperative findings, postoperative complications, reinterventions, reoperations, 30-day mortality, histology, and timing of multimodality treatment). Outside of the study group, in Austria pancreatic resections are performed in three “high-volume” centers (>10 pancreatic resections per year), three “medium-volume” centers (5–10 pancreatic resections per year), and the rest in various low-volume centers (<5 pancreatic resections per year) in Austria. Nationwide data for prevalence of and surgical resections for pancreatic adenocarcinoma were contributed by the National Cancer Registry of Statistics of Austria and the Austrian Health Institute.
Results
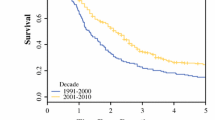
In total, 492 consecutive patients underwent pancreatic resection for ductal adenocarcinoma. All postoperative complications leading to hospital readmission were treated at the primary surgical department and documented in the database. Overall morbidity and pancreatic fistula rate were 45.5 % and 10.1 %, respectively. Within the entire cohort there were 9.8 % radiological reinterventions and 10.4 % reoperations. Length of stay was 16 days in median (0–209); 12 of 492 patients died within 30 days after operation, resulting in a 30-day mortality rate of 2.4 %. Seven of the total 19 deaths (36.8 %) occurred after 30 days, during hospitalization at the surgical department, resulting in a hospital mortality rate of 3.9 % (19/492). With a standardized histopathological protocol, there were 70 % (21/30) R0 resections, 30 % (9/30) R1 resections, and no R2 resections in Vienna and 62.7 % (32/51) R0 resections, 35.3 % (18/51) R1 resections, and 2 % (1/51) R2 resections in Salzburg. Resection margin status with nonstandardized protocols was classified as R0 in 82 % (339/411), R1 in 16 % (16/411), and R2 in 1.2 % (5/411). Perioperative chemotherapy was administered in 81.1 % of patients (8.3 % neoadjuvant; 68.5 % adjuvant; 4.3 % palliative); chemoradiotherapy (1.6 % neoadjuvant; 3 % adjuvant; 0.2 % palliative), in 4.9 % of patients. The six centers that contributed to this registry initiative provided surgical treatment to 40 % of all Austrian patients, resulting in a median annual recruitment of 85 (51–104) patients for the entire ABCSG-group and a median of 11.8 (0–38) surgeries for each individual department.
Conclusions
Surgical quality data of the ABCSG core pancreatic group are in line with international standards. With continuing centralization the essential potential to perform prospective clinical trials for pancreatic adenocarcinoma is given in Austria. Several protocol proposals aiming at surgical and multimodality research questions are currently being discussed.
Similar content being viewed by others
References
Winter JM, Cameron JL, Campbell KA et al (2006) 1423 pancreaticoduodenectomies for pancreatic cancer: a single-institution experience. J Gastrointest Surg 10:1199–1210 discussion 1210–1191
McPhee JT, Hill JS, Whalen GF et al (2007) Perioperative mortality for pancreatectomy: a national perspective. Ann Surg 246:246–253
Lemmens VE, Bosscha K, van der Schelling G et al (2011) Improving outcome for patients with pancreatic cancer through centralization. Br J Surg 98:1455–1462
Birkmeyer JD, Siewers AE, Finlayson EV et al (2002) Hospital volume and surgical mortality in the United States. N Engl J Med 346:1128–1137
Allareddy V, Ward MM, Konety BR (2010) Effect of meeting Leapfrog volume thresholds on complication rates following complex surgical procedures. Ann Surg 251:377–383
de Wilde RF, Besselink MG, van der Tweel I et al (2012) Impact of nationwide centralization of pancreaticoduodenectomy on hospital mortality. Br J Surg 99:404–410
Neoptolemos JP, Stocken DD, Friess H et al (2004) A randomized trial of chemoradiotherapy and chemotherapy after resection of pancreatic cancer. N Engl J Med 350:1200–1210
Neoptolemos JP, Moore MJ, Cox TF et al (2012) Effect of adjuvant chemotherapy with fluorouracil plus folinic acid or gemcitabine vs observation on survival in patients with resected periampullary adenocarcinoma: the ESPAC-3 periampullary cancer randomized trial. JAMA 308:147–156
Suc B, Msika S, Piccinini M et al (2004) Octreotide in the prevention of intra-abdominal complications following elective pancreatic resection: a prospective, multicenter randomized controlled trial. Arch Surg 139:288–294 discussion 295
Jakesz R, Greil R, Gnant M et al (2007) Extended adjuvant therapy with anastrozole among postmenopausal breast cancer patients: results from the randomized Austrian Breast and Colorectal Cancer Study Group Trial 6a. J Natl Cancer Inst 99:1845–1853
Gnant M, Mlineritsch B, Schippinger W et al (2009) Endocrine therapy plus zoledronic acid in premenopausal breast cancer. N Engl J Med 360:679–691
Tausch C, Taucher S, Dubsky P et al (2012) Prognostic value of number of removed lymph nodes, number of involved lymph nodes, and lymph node ratio in 7502 breast cancer patients enrolled onto trials of the Austrian Breast and Colorectal Cancer Study Group (ABCSG). Ann Surg Oncol 19:1808–1817
van Heek NT, Kuhlmann KF, Scholten RJ et al (2005) Hospital volume and mortality after pancreatic resection: a systematic review and an evaluation of intervention in the Netherlands. Ann Surg 242:781–788 discussion 788–790
Gangl O, Froschl U, Hofer W et al (2011) Unplanned reoperation and reintervention after pancreatic resections: an analysis of risk factors. World J Surg 35:2306–2314. doi:10.1007/s00268-011-1213-5
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
DeOliveira ML, Winter JM, Schafer M et al (2006) Assessment of complications after pancreatic surgery: a novel grading system applied to 633 patients undergoing pancreaticoduodenectomy. Ann Surg 244:931–937 discussion 937–939
Bassi C, Dervenis C, Butturini G et al (2005) Postoperative pancreatic fistula: an international study group (ISGPF) definition. Surgery 138:8–13
Sarr MG (2003) The potent somatostatin analogue vapreotide does not decrease pancreas-specific complications after elective pancreatectomy: a prospective, multicenter, double-blinded, randomized, placebo-controlled trial. J Am Coll Surg 196:556–564 discussion 564–555; author reply 565
Wente MN, Veit JA, Bassi C et al (2007) Postpancreatectomy hemorrhage (PPH): an International Study Group of Pancreatic Surgery (ISGPS) definition. Surgery 142:20–25
Verbeke CS, Leitch D, Menon KV et al (2006) Redefining the R1 resection in pancreatic cancer. Br J Surg 93:1232–1237
Esposito I, Kleeff J, Bergmann F et al (2008) Most pancreatic cancer resections are R1 resections. Ann Surg Oncol 15:1651–1660
Fernandez-del Castillo C, Morales-Oyarvide V, McGrath D et al (2012) Evolution of the Whipple procedure at the Massachusetts General Hospital. Surgery 152:S56–S63
Riall TS, Nealon WH, Goodwin JS et al (2008) Outcomes following pancreatic resection: variability among high-volume providers. Surgery 144:133–140
Gillen S, Schuster T, Meyer Zum Buschenfelde C et al (2010) Preoperative/neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of response and resection percentages. PLoS Med 2010(7):e1000267
Sahora K, Kuehrer I, Schindl M et al (2011) NeoGemTax: gemcitabine and docetaxel as neoadjuvant treatment for locally advanced nonmetastasized pancreatic cancer. World J Surg 35:1580–1589. doi:10.1007/s00268-011-1113-8
Brown KM, Siripurapu V, Davidson M et al (2008) Chemoradiation followed by chemotherapy before resection for borderline pancreatic adenocarcinoma. Am J Surg 195:318–321
Strobel O, Berens V, Hinz U et al (2012) Resection after neoadjuvant therapy for locally advanced, “unresectable” pancreatic cancer. Surgery 152:S33–S42
Stokes JB, Nolan NJ, Stelow EB et al (2011) Preoperative capecitabine and concurrent radiation for borderline resectable pancreatic cancer. Ann Surg Oncol 18:619–627
Sahora K, Kuehrer I, Eisenhut A et al (2011) NeoGemOx: gemcitabine and oxaliplatin as neoadjuvant treatment for locally advanced, nonmetastasized pancreatic cancer. Surgery 149:311–320
Walters DM, Lapar DJ, de Lange EE et al (2011) Pancreas-protocol imaging at a high-volume center leads to improved preoperative staging of pancreatic ductal adenocarcinoma. Ann Surg Oncol 18:2764–2771
Glant JA, Waters JA, House MG et al (2011) Does the interval from imaging to operation affect the rate of unanticipated metastasis encountered during operation for pancreatic adenocarcinoma? Surgery 150:607–616
Acknowledgements
The authors thank Gerhard Fülöp (Austrian Health Institute) for contributing absolute numbers of annual pancreatic resections in Austria.
Conflict of interest
The authors declare no conflict of interest
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gangl, O., Sahora, K., Kornprat, P. et al. Preparing for Prospective Clinical Trials: A National Initiative of an Excellence Registry for Consecutive Pancreatic Cancer Resections. World J Surg 38, 456–462 (2014). https://doi.org/10.1007/s00268-013-2283-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-013-2283-3