Abstract
Objectives
The purpose of this study was to identify important prognostic factors related to the status of a pancreatic tumor, its treatment, and the patient’s general condition.
Methods
Between April 1992 and December 2006, 140 patients underwent a pancreatic resection for invasive ductal carcinoma. Prognostic factors were defined by univariate and multivariate analyses.
Results
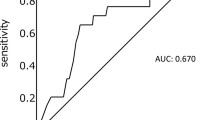
The study included 103 tumors in the head of the pancreas and 37 tumors in the body or tail. The median survival time and the actuarial 5-year survival rate for all patients were 14.5 months and 12.3%, respectively. Using the significant prognostic factors identified by univariate analysis, multivariate analysis revealed that a preoperative serum CA19-9 concentration >100 U/ml (HR = 1.84, p = 0.0074), a tumor size >3 cm (HR = 1.74, p = 0.0235), venous involvement (HR = 2.39, p = 0.0006), a transfusion requirement of ≥1000 ml (HR = 2.23, p = 0.0006), and a serum albumin concentration on 1 postoperative month (1POM) < 3 g/dl (HR = 2.40, p = 0.0009) were significant adverse prognostic factors. The presence of hypoalbuminemia on 1POM significantly correlated with a longer surgical procedure (p = 0.0041), extended nerve plexus resection around the superior mesenteric artery (p = 0.0456), and a longer postoperative hospital stay (p = 0.0063).
Conclusion
To improve long-term survival, preserving the patient’s general condition by performing a curative resection with a short operation time and minimal blood loss should be the most important principle in the surgical treatment of pancreatic cancer.

Similar content being viewed by others
References
Sohn TA, Yeo CJ, Cameron JL et al (2000) Resected adenocarcinoma of the pancreas—616 patients: results, outcomes, and prognostic indicators. J Gastrointest Surg 4:567–579
Cleary SP, Gryfe R, Guindi M et al (2004) Prognostic factors in resected pancreatic adenocarcinoma: analysis of actual 5-year survivors. J Am Coll Surg 198:722–731
Han SS, Jang JY, Kim SW et al (2006) Analysis of long-term survivors after surgical resection for pancreatic cancer. Pancreas 32:271–275
Moon HJ, An JY, Heo JS et al (2006) Predicting survival after surgical resection for pancreatic ductal adenocarcinoma. Pancreas 32:37–43
Matsuno S, Egawa S, Fukuyama S et al (2004) Panreatic cancer registry in Japan: 20 years of experience. Pancreas 28:219–230
Riall TS, Cameron JL, Lillemoe KD et al (2005) Pancreaticoduodenectomy with or without distal gastrectomy and extended retroperitoneal lymphadenectomy for periampullary adenocarcinoma–Part 3: update on 5-year survival. J Gastrointest Surg 9:1191–1206
Pedrazzoli S, DiCarlo V, Dionigi R et al (1998) Standard versus extended lymphadenectomy associated with pancreatoduodenectomy in the surgical treatment of adenocarcinoma of the head of the pancreas. A multicenter, prospective, randomized study. Ann Surg 228:508–517
Farnell MB, Pearson RK, Sarr MG et al (2005) A prospective randomized trial comparing standard pancreatoduodenectomy with pancreatoduodenectomy with extended lymphadenectomy in respectable pancreatic head adenocarcinoma. Surgery 138:618–630
Shimada K, Sano T, Sakamoto Y et al (2006) Clinical implications of combined portal vein resection as a palliative procedure in patients undergoing pancreaticoduodenectomy for pancreatic head carcinoma. Ann Surg Oncol 13:1569–1578
Nagakawa T, Kayahara M, Ueno K et al (1992) A clinicopathologic study on neural invasion in cancer of the pancreatic head. Cancer 69:930–935
Nakao A, Harada A, Nonami T et al (1996) Clinical significance of carcinoma invasion of the extrapancreatic nerve plexus in pancreatic cancer. Pancreas 12:357–361
Neoptolemos JP, Stocken DD, Friess H et al (2004) A randomized trial of chemoradiotherapy and chemotherapy after resection of pancreatic cancer. N Engl J Med 350:1200–1210
Oettle H, Post S, Neuhaus P et al (2007) Adjuvant chemotherapy with gemcitabine vs observation in patients undergoing curative-intent resection of pancreatic cancer: a randomized controlled trial. JAMA 297:267–277
Howard TJ, Krug JE, Yu J et al (2006) A margin-negative R0 resection accomplished with minimal postoperative complications is the surgeon’s contribution to long-term survival in pancreatic cancer. J Gastrointest Surg 10:1338–1346
Rey-Ferro M, Castaño R, Orozco O et al (1997) Nutritional and immunologic evaluation of patients with gastric cancer before and after surgery. Nutrition 13:878–881
Marrelli D, Roviello F, De Stefano A et al (2000) Surgical treatment of gastrointestinal carcinomas in octogenarians: risk factors for complications and long-term outcome. Eur J Surg Oncol 26:371–376
Marrelli D, Pedrazzani C, Neri A et al (2007) Complications after extended (D2) and superextended (D3) lymphadenectomy for gastric cancer: analysis of potential risk factors. Ann Surg Oncol 14:25–33
Gibbs J, Cull W, Henderson W et al (1999) Preoperative serum albumin level as a predictor of operative mortality and morbidity: results from the National VA Surgical Risk Study. Arch Surg 134:36–42
Palma S, Cosano A, Mariscal M et al (2007) Cholesterol and serum albumin as risk factors for death in patients undergoing general surgery. Br J Surg 94:369–375
Japan Pancreas Society (2003) Classification of pancreatic carcinoma, 2nd English edn. Kanehara, Tokyo
Misuta K, Shimada H, Miura Y et al (2005) The role of splenomesenteric vein anastomosis after division of the splenic vein in pancreatoduodenectomy. J Gastrointest Surg 9:245–253
Miura Y, Ueda M, Kunihiro O et al (2004) Hepatic arterial infusion therapy for pancreatic cancer. Gan To Kagaku Ryoho 31:195–198
Wakefield CH, Carey PD, Foulds S et al (1993) Changes in major histocompatibility complex class II expression in monocytes and T cells of patients developing infection after surgery. Br J Surg 80:205–209
Spolarics Z, Siddiqi M, Siegel JH et al (2003) Depressed interleukin-12-producing activity by monocytes correlates with adverse clinical course and a shift toward Th2-type lymphocyte pattern in severely injured male trauma patients. Crit Care Med 31:1722–1729
Hensler T, Hecker H, Heeg K et al (1997) Distinct mechanisms of immunosuppression as a consequence of major surgery. Infect Immun 65:2283–2291
Edna TH, Bjerkeset T (1998) Perioperative blood transfusions reduce long-term survival following surgery for colorectal cancer. Dis Colon Rectum 41:451–459
Amato AC, Pescatori M (1998) Effect of perioperative blood transfusions on recurrence of colorectal cancer: meta-analysis stratified on risk factors. Dis Colon Rectum 41:570–585
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ueda, M., Endo, I., Nakashima, M. et al. Prognostic Factors After Resection of Pancreatic Cancer. World J Surg 33, 104–110 (2009). https://doi.org/10.1007/s00268-008-9807-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-008-9807-2