Abstract
Introduction:
The rising popularity of facial filler injections has corresponded with an increase in reported complications. While a filler emergency kit was previously introduced, advancements in the field have highlighted certain limitations, prompting the development of the updated filler emergency kit (UFEK).
Methods:
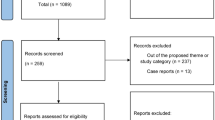
The authors conducted literature research up to February 2023, focusing on PubMed and open web searches for articles referred to filler emergent complications: vascular occlusion, blindness and anaphylaxis. Approximately 1200 articles were obtained from PubMed and other sources, and 45 articles were reviewed.
Results:
The developed UFEK protocol delineates specific interventions meticulously tailored to address diverse emergent scenarios linked to soft tissue fillers complications. This protocol emphasizes the urgent requirement for timely and personalized interventions.
Conclusion:
The UFEK offers a standardized, comprehensive and effective approach. This work contributes to the responsible and informed progression of the field of aesthetic medicine, providing more value and safety, both for clinicians and patients.
Level of Evidence IV This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.

Similar content being viewed by others
References
Sito G, Manzoni V, Sommariva R (2019) Vascular complications after facial filler injection. J Clin Aesthet Dermatol 12(6):E65–E72. https://doi.org/10.1111/ijcp.14675
Databank N (2022) Aesthetic plastic surgery national databank statistics 2020–2021. Aesthet Surg J 42(1):1–18. https://doi.org/10.1093/asj/sjac116
Bellew S, Del Rosso JQ (2011) The spectrum of adverse reactions after treatment with injectable fillers in the glabellar region: results from the injectable filler safety study. Dermatol Dermatol Surg 2011:451–452. https://doi.org/10.1016/j.yder.2010.12.019
Schelke L, Decates T, Kadouch J, Velthuis P (2020) Incidence of vascular obstruction after filler injections. Aesthet Surg J 40(8):NP457–NP460. https://doi.org/10.1093/asj/sjaa086
Lemperle G, Duffy DM (2006) Treatment options for dermal filler complications. Aesthet Surg J 26(3):356–364. https://doi.org/10.1016/j.asj.2006.04.002
Carruthers A, Carruthers J (2007) Non-animal-based hyaluronic acid fillers: scientific and technical considerations. Plast Reconstr Surg 120(6S):33–40. https://doi.org/10.1097/01.prs.0000248808.75700.5f
Sahin I, Isik S (2012) Blindness following cosmetic injections of the face. Plast Reconstr Surg 130(5):2012. https://doi.org/10.1097/PRS.0b013e318267d740
Beleznay K, Carruthers JDA, Humphrey S, Jones D (2015) Avoiding and treating blindness from fillers: a review of the world literature. Dermatol Surg 41(10):1097–1117. https://doi.org/10.1097/DSS.0000000000000486
Lemperle G, Rullan PP, Gauthier-Hazan N (2006) Avoiding and treating dermal filler complications. Plast Reconstr Surg 118(3 SUPPL.):92–107. https://doi.org/10.1097/01.prs.0000234672.69287.77
Fakih-Gomez N, Orte-Aldea MC, Poonja K, Khanna D (2019) Hyaluronic acid filler emergency kit. Am J Cosmet Surg 36(4):183–190. https://doi.org/10.1177/0748806818825214
Rohrich RJ, Nguyen AT, Kenkel JM (2009) Lexicon for soft tissue implants. Dermatol Surg 35(SUPPL. 2):1605–1611. https://doi.org/10.1111/j.1524-4725.2009.01337.x
Kreidstein ML, Pang CY, Levine RH, Knowlton RJ (1991) The isolated perfused human skin flap: design, perfusion technique, metabolism, and vascular reactivity. Plast Reconstr Surg 87(4):741–749
Delorenzi C (2014) Complications of injectable fillers, part 2: vascular complications. Aesthet Surg J 34(4):584–600. https://doi.org/10.1177/1090820X14525035
Rzany B, DeLorenzi C (2015) Understanding, avoiding, and managing severe filler complications. Plast Reconstr Surg 136(5):196S-203S. https://doi.org/10.1097/PRS.0000000000001760
Cavallini M, Gazzola R, Metalla M, Vaienti L (2013) The role of hyaluronidase in the treatment of complications from hyaluronic acid dermal fillers. Aesthet Surg J 33(8):1167–1174. https://doi.org/10.1177/1090820X13511970
Dayan SH, Arkins JP, Brindise R (2011) Soft tissue fillers and biofilms. Facial Plast Surg 27(1):23–28. https://doi.org/10.1055/s-0030-1270415
DeLorenzi C (2017) New high dose pulsed hyaluronidase protocol for hyaluronic acid filler vascular adverse events. Aesthet Surg J 37(7):814–825. https://doi.org/10.1093/asj/sjw251
Schelke LW, Velthuis P, Kadouch J, Swift A (2023) Early ultrasound for diagnosis and treatment of vascular adverse events with hyaluronic acid fillers. J Am Acad Dermatol 88(1):79–85. https://doi.org/10.1016/j.jaad.2019.07.032
Schelke LW, Velthuis PJ, Decates T et al (2023) Ultrasound-guided targeted vs regional flooding: a comparative study for improving the clinical outcome in soft tissue filler vascular adverse event management. Aesthet Surg J 43(1):86–96
Carruthers J, Fagien S, Dolman P (2015) Retro or peribulbar injection techniques to reverse visual loss after filler injections. Dermatol Surg 41:S354–S357. https://doi.org/10.1097/DSS.0000000000000558
Carruthers JDA, Fagien S, Rohrich RJ, Weinkle S, Carruthers A (2014) Blindness caused by cosmetic filler injection: a review of cause and therapy. Plast Reconstr Surg 134(6):1197–1201. https://doi.org/10.1097/PRS.0000000000000754
Chesnut C (2018) Restoration of visual loss with retrobulbar hyaluronidase injection after hyaluronic acid filler. Dermatol Surg 44(3):435–437. https://doi.org/10.1097/DSS.0000000000001237
Fagien S, Carruthers J (2018) Commentary on restoration of visual loss with retrobulbar hyaluronidase injection after hyaluronic acid filler. Dermatol Surg 44(3):437–443. https://doi.org/10.1097/DSS.0000000000001372
Zhu GZ, Sun ZS, Liao WX et al (2017) Efficacy of retrobulbar hyaluronidase injection for vision loss resulting from hyaluronic acid filler embolization. Aesthet Surg J 38(1):12–22. https://doi.org/10.1093/asj/sjw216
Hwang CJ, Mustak H, Gupta AA, Ramos RM, Goldberg RA, Duckwiler GR (2019) Role of retrobulbar hyaluronidase in filler-associated blindness: evaluation of fundus perfusion and electroretinogram readings in an animal model. Ophthal Plast Reconstr Surg 35(1):33–37. https://doi.org/10.1097/IOP.0000000000001132
Tansatit T, Apinuntrum P, Phetudom T (2015) A cadaveric feasibility study of the intraorbital cannula injections of hyaluronidase for initial salvation of the ophthalmic artery occlusion. Aesthet Plast Surg 39(2):252–261. https://doi.org/10.1007/s00266-015-0456-1
Ramesh S, Fiaschetti D, Goldberg RA (2018) Orbital and ocular ischemic syndrome with blindness after facial filler injection. Ophthal Plast Reconstr Surg 34(4):E108–E110. https://doi.org/10.1097/IOP.0000000000001104
Jajoria H, Venkataram A, Mysore V (2020) Importance of choke vessels in injectable fillers. J Cutan Aesthet Surg 13(3):185–190
Ashton MW, Taylor GI, Corlett RJ (2018) The role of anastomotic vessels in controlling tissue viability and defining tissue necrosis with special reference to complications following injection of hyaluronic acid fillers. Plast Reconstr Surg 141:818e–830e. https://doi.org/10.1097/PRS.0000000000004287
Saint-Cyr M, Wong C, Schaverien M, Mojallal A, Rohrich RJ (2009) The perforasome theory: vascular anatomy and clinical implications. Plast Reconstr Surg 124(5):1529–1544. https://doi.org/10.1097/PRS.0b013e3181b98a6c
Taylor GI, Corlett RJ, Ashton MW (2017) The functional angiosome: clinical implications of the anatomical concept. Plast Reconstr Surg 140(4):721–733. https://doi.org/10.1097/PRS.0000000000003694
Pafitanis G, Hung-Chi C (2019) The pinprick test: key considerations in execution of skin flap perfusion testing. Plast Reconstr Surg Glob Open 7(9):e2370
Mishra S (2011) A simple method for predicting survival of pedicled skin flaps before completely raising them. Indian J Plast Surg 44(3):453–457. https://doi.org/10.4103/0970-0358.90821
Galiè N (2009) Phosphodiesterase type 5 inhibitors. Pulm Hypertens 5(August):350–354. https://doi.org/10.2165/00063030-200013020-00002
Samlaska CP, Winfield EA (1994) Pentoxifylline. J Am Acad Dermatol 30(4):603–621. https://doi.org/10.1016/S0190-9622(94)70069-9
Shi L, Carson D (2009) Collagenase santyl ointment: a selective agent for wound debridement. J Wound Ostomy Cont Nurs 36(SUPPL. 6):3–7. https://doi.org/10.1097/WON.0b013e3181bfdd1a
Carle MV, Roe R, Novack R, Boyer DS (2014) Cosmetic facial fillers and severe vision loss. JAMA Ophthalmol 132(5):637–639. https://doi.org/10.1001/jamaophthalmol.2014.498
Shin H, Lemke BN, Stevens T, Lim MJ (1988) Posterior ciliary-artery occlusion after subcutaneous silicone-oil injection. Ann Ophthalmol 20(9):342–344
Lazzeri D, Agostini T, Figus M, Nardi M, Pantaloni M, Lazzeri S (2012) Blindness following cosmetic injections of the face. Plast Reconstr Surg 129(4):995–1012. https://doi.org/10.1097/PRS.0b013e3182442363
Bandyk DF (2018) The diabetic foot: Pathophysiology, evaluation, and treatment. Semin Vasc Surg 31(2–4):43–48. https://doi.org/10.1053/j.semvascsurg.2019.02.001
Smith J, Jones L, Garcia M (2022) Antibiotic selection for the empiric treatment of Staphylococcus aureus infections: considerations for methicillin-resistant strains. J Infect Dis 215(3):432–439
Ramamoorthy S, Cidlowski JA (2016) Corticosteroids: mechanisms of action in health and disease. Rheum Dis Clin North Am 42(1):15–31. https://doi.org/10.1016/j.rdc.2015.08.002
Alam M, Dover JS (2007) Management of complications and sequelae with temporary injectable fillers. Plast Reconstr Surg 120(6S):98–105. https://doi.org/10.1097/01.prs.0000248859.14788.60
Wyns H, Meyer E, Watteyn A et al (2013) Pharmacokinetics of dexamethasone after intravenous and intramuscular administration in pigs. Vet J 198(1):286–288. https://doi.org/10.1016/j.tvjl.2013.06.015
Tupper J, Visser S, Em C (2010) Emergency files a review and update. Can Fam Phys 56:1009–1011
Acknowledgements
N.F.G., C.A.P.P., C.M.G., and J.K. were involved in the hypothesis/clinical observation. N.F.G., C.A.P.P., A.V.G., C.M.G., and J.K contributed to the study design/methodology. N.F.G., C.A.P.P., A.V.G., and C.M.G. contributed to the research/data assessment. N.F.G., C.A.P.P., A.V.G., and C.M.G. analyzed the data. N.F.G., C.A.P.P., A.V.G., C.M.G., and J.K. wrote the paper.
Funding
None of the authors received any funding or financial support for the content of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors N.F. and J.K. are both consultants for Merz Aesthetics (Frankfurt, Germany).
Human or Animal Rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
For this type of study, formal consent is not needed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fakih-Gomez, N., Porcar Plana, C.A., Verano-Garcia, A. et al. Updated Filler Emergency Kit: Next-Generation Emergency Solution. Aesth Plast Surg 48, 1174–1180 (2024). https://doi.org/10.1007/s00266-023-03722-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-023-03722-3