Abstract
Background
Wound contractures can cause severe deformities and disabilities. Recent studies have suggested that leukotriene receptor antagonists have an inhibitory effect on the healing contraction process. This study aimed to evaluate the influence of the leukotriene inhibitor montelukast on the cutaneous healing process and the wound contraction phenomenon in rats.
Methods
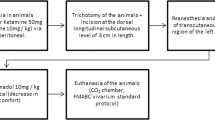
For this study, 60 male rats were randomly divided into four groups (MK-7d, SF-7d, MK-14d, and SF-14d) according to the drug given through a rigid orogastric tube (MK group: montelukast 10 mg/kg/day; SF group: normal saline solution) and the day the animals were killed (7d: postoperative day 7; 14d: postoperative day 14). An excisional wound (2 × 2 cm) was created on the dorsum of each rat. The wounds were left open to heal spontaneously and documented by standard digital photographs on different postoperative days. Wound contraction rates were calculated with specific software, and specimens were histologically evaluated using picrosirius red stain. Results were analyzed using the Aspin-Welch, Mann–Whitney, and t tests, assuming a significance level of 5%.
Results
The wound contraction rates were similar between the control and study groups (p > 0.05). On postoperative day 7, the wounds showed a marginally significant reduction in collagen maturation in the study group (40.1% ± 6.88% vs 61.2% ± 8.02%; p = 0.0607). On postoperative day 14, this reduction was statistically significant in the MK group (26% ± 5.66% vs 68.3% ± 7.76%; p = 0.0001).
Conclusions
Montelukast does not alter the contraction rate of excisional wounds in rats but has a significant and progressive inhibitory effect on collagen maturation.





Similar content being viewed by others
References
Bastos EM, Neto MS, Alves MT, Garcia EB, Santos RA, Heink T, Pereira JB, Ferreira LM (2007) Histologic analysis of zafirlukast’s effect on capsule formation around silicone implants. Aesth Plast Surg 31:559–565
Brissett AE, Sherris DA (2001) Scar contractures, hypertrophic scars, and keloids. Facial Plast Surg 17:263–272
Chen S, Jin G, Shan J, Zhang M, Men Q, Xu L (2003) Cardioprotective effect and influence on NOS expression of Montelukast sodium in rats. Yao Xue Xue Bao 38:821–825
da Rocha FA, Teixeira MM, Rocha JC, Girao VC, Bezerra MM, Ribeiro RA, Cunha FQ (2004) Blockade of leukotriene B4 prevents articular incapacitation in rat zymosan-induced arthritis. Eur J Pharmacol 497:81–86
Davidson JM (2001) Experimental animal wound models. Wounds 13:9–23
Dorsett-Martin WA (2004) Rat models of skin wound healing: a review. Wound Repair Regen 12:591–599
Gryskiewicz JM (2003) Investigation of Accolate and Singulair for treatment of capsular contracture yields safety concerns. Aesth Surg J 23:98–102
Holma R, Salmenpera P, Riutta A, Virtanen I, Korpela R, Vapaatalo H (2001) Acute effects of the cys-leukotriene–1 receptor antagonist, montelukast, on experimental colitis in rats. Eur J Pharmacol 429:309–318
Kabasakal L, Sener G, Cetinel S, Contuk G, Gedik N, Yegen BC (2005) Burn-induced oxidative injury of the gut is ameliorated by the leukotriene receptor blocker montelukast. Prostaglandins Leukot Essent Fatty Acids 72:431–440
Leick-Maldonado EA, Kay FU, Leonhardt MC, Kasahara DI, Prado CM, Fernandes FT, Martins MA, Tiberio IF (2004) Comparison of glucocorticoid and cysteinyl leukotriene receptor antagonist treatments in an experimental model of chronic airway inflammation in guinea-pigs. Clin Exp Allergy 34:145–152
Lipworth BJ (1999) Leukotriene-receptor antagonists. Lancet 353:57–62
Markham A, Faulds D (1998) Montelukast. Drugs 2:251–256
Mogford JE, Mustoe TA (2001) Experimental models of wound healing. In: Falanga V (ed) Cutaneous Wound Healing. Martin Dunitz, London, pp 109–122
Montandon D, D’anoiran G, Gabbiani G (1977) The mechanism of wound contraction and epithelialization: clinical and experimental studies. Clin Plast Surg 4:325–346
Nayak A (2004) A review of montelukast in the treatment of asthma and allergic rhinitis. Expert Opin Pharmacother 5:679–686
Reid RR, Greve SD, Casas LA (2005) The effect of zafirlukast (Accolate) on early capsular contracture in the primary augmentation patient: a pilot study. Aesth Surg J 25:26–30
Saito H, Morikawa H, Howie K, Crawford L, Baatjes AJ, Denburg E, Cyr MM, Denburg JA (2004) Effects of a cysteinyl leukotriene receptor antagonist on eosinophil recruitment in experimental allergic rhinitis. Immunology 113:246–252
Schlesinger SL, Ellenbogen R, Desvigne MN, Svehlak S, Heck R (2002) Zafirlukast (Accolate): a new treatment for capsular contracture. Aesth Surg J 22:329–336
Schneider JC, Holavanahalli R, Helm P, Goldstein R, Kowalske K (2006) Contractures in burn injury: defining the problem. J Burn Care Res 27:508–514
Scuderi N, Mazzocchi M, Fioramonti P, Bistoni G (2006) The effects of zafirlukast on capsular contracture: preliminary report. Aesth Plast Surg 30:513–520
Sener G, Kabasakal L, Cetinel S, Contuk G, Gedik N, Yegen BC (2005) Leukotriene receptor blocker montelukast protects against burn-induced oxidative injury of the skin and remote organs. Burns 31:587–596
Sener G, Kapucu C, Cetinel S, Cikler E, Ayanoglu-Dulger G (2005) Gastroprotective effect of leukotriene receptor blocker montelukast in alendronat-induced lesions of the rat gastric mucosa. Prostaglandins Leukot Essent Fatty Acids 72:1–11
Sener G, Sehirli O, Cetinel S, Ercan F, Yuksel M, Gedik N, Yegen BC (2005) Amelioration of sepsis-induced hepatic and ileal injury in rats by the leukotriene receptor blocker montelukast. Prostaglandins Leukot Essent Fatty Acids 73:453–462
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was developed by the Post-Graduate Program in Surgery and Plastic and Reconstructive Surgery Unit of Federal University of Paraná, Curitiba, Brazil.
Rights and permissions
About this article
Cite this article
Tolazzi, A.R.D., Tolazzi, K.D., Garcia, M. et al. Influence of Leukotriene Inhibitor Montelukast on Wound Contraction and Cutaneous Healing Process in Rats. Aesth Plast Surg 33, 84–89 (2009). https://doi.org/10.1007/s00266-008-9241-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-008-9241-8