Abstract
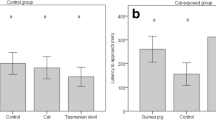
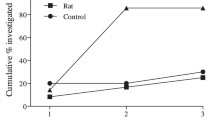
Inability to recognise and/or express effective anti-predator behaviour against novel predators as a result of ontogenetic and/or evolutionary isolation is known as ‘prey naiveté’. Natural selection favours prey species that are able to successfully detect, identify and appropriately respond to predators prior to their attack, increasing their probability of escape and/or avoidance of a predator. However, for many prey species, learning and experience are necessary to develop and perform appropriate anti-predator behaviours. Here, we investigate how a remnant population of bilbies (Macrotis lagotis) in south-west Queensland responded to the scents of two predators, native dingoes (Canis familiaris) and introduced feral cats (Felis catus); a procedural control (rabbits; Oryctolagus cuniculus); and an experimental control (no scent). Bilbies in Queensland have shared more than 8000 years of co-evolutionary history with dingoes and less than 140 years with feral cats and less than 130 years with rabbits. Bilbies spent the greatest proportion of time investigating and the least amount of time digging when cat and dingo/dog faeces were present. Our results show that wild-living bilbies displayed anti-predator responses towards the olfactory cues of both a long-term predator (dingoes) and an evolutionary novel predator (cats). Our findings suggest that native species can develop anti-predator responses towards introduced predators, providing support for the idea that predator naiveté can be overcome through learning and natural selection as a result of exposure to introduced predators.
Significance statement
Not so naïve—As a result of lifetime and/or evolutionary isolation from predators, some prey species appear to be naïve towards introduced predators. This is particularly the case in Australia, where native mammalian species appear to be naïve towards recently introduced predators such as the feral cat and European red fox. In a study of wild-living bilbies, we found that 150 years of co-evolutionary experience is enough to develop predator recognition.



Similar content being viewed by others
Data availability
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Abbott I (2002) Origin and spread of the cat, Felis catus, on mainland Australia, with a discussion of the magnitude of its early impact on native fauna. Wildl Res 29:51–74. https://doi.org/10.1071/WR01011
Anton A, Geraldi N, Ricciardi A, Dick J (2020) Global determinantsof prey naiveté to exotic predators. Proc R Soc B 287:20192978. https://doi.org/10.1098/rspb.2019.2978
Banks PB, Carthey AJR, Bytheway JP (2018) Australian native mammals recognize and respond to alien predators: a metaanalysis. Proc R Soc B 285:20180857
Barrio IC, Bueno CG, Banks PB, Tortosa FS (2010) Prey naiveté in an introduced prey species: the wild rabbit in Australia. Behav Ecol 21:986–991. https://doi.org/10.1093/beheco/arq103
Berejikian BA, Tezak E, LaRae AL (2003) Innate and enhanced predator recognition in hatchery-reared Chinook salmon. Environ Biol Fish 67:241–251. https://doi.org/10.1023/A:1025887015436
Blanchard CD, Blanchard RJ (2004) Antipredator defense. In: Whishaw IQ, Kolb B (eds) The behavior of the laboratory rat: a handbook with tests. Oxford University Press, Oxford, p 520
Blumstein DT, Daniel JC (2007) Quantifying behavior the JWatcher way. Sinauer Associates Incorporated, Sunderland, MA
Blumstein DT, Mari M, Daniel JC, Ardron JG, Griffin AS, Evans CS (2002) Olfactory predator recognition: wallabies may have to learn to be wary. Anim Conserv 5:87–93. https://doi.org/10.1017/s1367943002002123
Blumstein DT, Barrow L, Luterra M (2008) Olfactory predator discrimination in yellow-bellied marmots. Ethology 114:1135–1143. https://doi.org/10.1111/j.1439-0310.2008.01563.x
Bouskila A, Blumstein DT (1992) Rules of thumb for predation hazard assessment: predictions from a dynamic model. Am Nat 139:161–176. https://doi.org/10.1086/285318
Brown GE, Chivers DP (2005) Learning as an adaptive response to predation. Ecology of predator–prey interactions. Oxford University Press, Oxford, England, pp 34–54
Burbidge AA, Woinarski J (2016) Macrotis lagotis. The IUCN Red List of Threatened Species 2016, https://doi.org/10.2305/IUCN.UK.2016-2.RLTS.T12650A21967189.en
Carthey AJ, Banks PB (2014) Naïveté in novel ecological interactions: lessons from theory and experimental evidence. Biol Rev 89:932–949. https://doi.org/10.1111/brv.12087
Carthey AJR, Banks PB (2018) Naïve, bold, or just hungry? An invasive exotic prey species recognises but does not respond to its predators. Biol Invasions 20:3417–3429
Carthey AJR, Blumstein DT (2018) Predicting predator recognition in a changing world. Trends Ecol Evol 33:106–115. https://doi.org/10.1016/j.tree.2017.10.009
Carthey AJ, Bucknall MP, Wierucka K, Banks PB (2017) Novel predators emit novel cues: a mechanism for prey naivety towards alien predators. Sci Rep 7:16377
Chivers DP, Smith RJF (1995) Free-living fathead minnows rapidly learn to recognize pike as predators. J Fish Biol 46:949–954
Chivers DP, Brown GE, Smith RJF (1995) Acquired recognition of chemical stimuli from pike, Esox lucius, by brook sticklebacks, Culaea inconstans (Osteichthyes, Gasterosteidae). Ethology 99:234–242. https://doi.org/10.1111/j.1439-0310.1995.tb00897.x
Cox JG, Lima SL (2006) Naiveté and an aquatic–terrestrial dichotomy in the effects of introduced predators. Trends Ecol Evol 21:674–680. https://doi.org/10.1016/j.tree.2006.07.011
Cremona T, Crowther MS, Webb JK (2014) Variation of prey responses to cues from a mesopredator and an apex predator. Austral Ecol 39:749–754. https://doi.org/10.1111/aec.12138
Dickman CR, Doncaster CP (1984) Responses of small mammals to red fox (Vulpes vulpes) odour. J Zool 204:521–531. https://doi.org/10.1111/j.1469-7998.1984.tb02384.x
Fendt M (2006) Exposure to urine of canids and felids, but not of herbivores, induces defensive behavior in laboratory rats. J Chem Ecol 32:2617–2627. https://doi.org/10.1007/s10886-006-9186-9
Ferrari MCO (2014) Short-term environmental variation in predation risk leads to differential performance in predation-related cognitive function. Anim Behav 95:9–14. https://doi.org/10.1016/j.anbehav.2014.06.001
Ferrari MCO, Crane AL, Brown GE, Chivers DP (2015) Getting ready for invasions: can background level of risk predict the ability of naïve prey to survive novel predators? Sci Rep 5:8309. https://doi.org/10.1038/srep08309
Ferrero DM, Lemon JK, Fluegge D, Pashkovski SL, Korzan WJ, Datta SR, Spehr M, Fendt M, Liberles SD (2011) Detection and avoidance of a carnivore odor by prey. Proc Natl Acad Sci U S A 108:11235–11240. https://doi.org/10.1073/pnas.1103317108
Fortin M-J, Dale MR (2005) Spatial analysis: a guide for ecologists. Cambridge University Press, Cambridge
Gibson LA (2001) Seasonal changes in the diet, food availability and food preference of the greater bilby (Macrotis lagotis) in south-western Queensland. Wildl Res 28:121–134. https://doi.org/10.1071/WR00003
Goldthwaite RO, Coss RG, Owings DH (1990) Evolutionary dissipation of an antisnake system: differential behavior by California and Arctic ground squirrels in above- and below-ground contexts. Behaviour 112:246–269. https://doi.org/10.1163/156853990X00220
Gorman ML, Trowbridge BJ (1989) The role of odor in the social lives of carnivores. In: Gittleman JL (ed) Carnivore behaviour, ecology, and evolution. Springer, USA, pp 57–88
Göth A (2001) Innate predator-recognition in Australian brush-turkey (Alectura lathami, Megapodiidae) hatchlings. Behaviour 138:117–136
Griffin AS, Blumstein DT, Evans CS (2000) Training captive-bred or translocated animals to avoid predators. Conserv Biol 14:1317–1326
Hayes RA, Nahrung HF, Wilson JC (2006) The response of native Australian rodents to predator odours varies seasonally: a by-product of life history variation? Anim Behav 71:1307–1314. https://doi.org/10.1016/j.anbehav.2005.08.017
Hedges SB, Dudley J, Kumar S (2006) TimeTree: a public knowledge-base of divergence times among organisms. Bioinformatics 22:2971–2972. https://doi.org/10.1093/bioinformatics/btl505
Johnson C, Johnson K (1983) Behaviour of the bilby, Macrotis lagotis (Reid), (Marsupialia: Thylacomyidae) in captivity. Wildl Res 10:77–87. https://doi.org/10.1071/WR9830077
Jolly CJ, Phillips BL (2020) Effects of rapid evolution due to predator-free conservation on endangered species recovery. Conserv Biol published online. https://doi.org/10.1111/cobi.13521
Jolly CJ, Webb JK, Phillips BL (2018) The perils of paradise: an endangered species conserved on an island loses antipredator behaviours within 13 generations. Biol Lett 14:20180222. https://doi.org/10.1098/rsbl.2018.0222
Kovacs E, Crowther M, Webb J, Dickman C (2012) Population and behavioural responses of native prey to alien predation. Oecologia 168:947–957. https://doi.org/10.1007/s00442-011-2168-9
Lavery HJ, Kirkpatrick TH (1997) Field management of the bilby Macrotis lagotis in an area of south-western Queensland. Biol Conserv 79:271–281. https://doi.org/10.1016/S0006-3207(96)00085-7
Legge S, Woinarski JCZ, Burbidge AA et al (2018) Havens for threatened Australian mammals: the contributions of fenced areas and offshore islands to protecting mammal species that are susceptible to introduced predators. Wildl Res 45:627–644
Li C, Yang X, Ding Y, Zhang L, Fang H, Tang S, Jiang Z (2011) Do Père David’s deer lose memories of their ancestral predators? PLoS One 6:e23623. https://doi.org/10.1371/journal.pone
Lima SL, Bednekoff PA (1999) Temporal variation in danger drives antipredator behavior: the predation risk allocation hypothesis. Am Nat 153:649–659
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68:619–640. https://doi.org/10.1139/z90-092
Lollback GW, Mebberson R, Evans N, Shuker JD, Hero J-M (2015) Estimating the abundance of the bilby (Macrotis lagotis): a vulnerable, uncommon, nocturnal marsupial. Austr Mamm 37:75–85
Maloney RF, McLean IG (1995) Historical and experimental learned predator recognition in free-living New-Zealand robins. Anim Behav 50:1193–1201
Masini CV, Sauer S, Campeau S (2005) Ferret odor as a processive stress model in rats. Behav Neurosci 119:280–292. https://doi.org/10.1037/0735-7044.119.1.280
McEvoy J, Sinn DL, Wapstra E (2008) Know thy enemy: behavioural response of a native mammal (Rattus lutreolus velutinus) to predators of different coexistence histories. Austr Ecol 33:922–931. https://doi.org/10.1111/j.1442-9993.2008.01863.x
Monclús R, Rödel HG, von Holst D, De Miguel J (2005) Behavioural and physiological responses of naïve European rabbits to predator odour. Anim Behav 70:753–761. https://doi.org/10.1016/j.anbehav.2004.12.019
Moseby KE, Blumstein DT, Letnic M (2016) Harnessing natural selection to tackle the problem of prey naïveté. Evol Appl 9:334–343. https://doi.org/10.1111/eva.12332
Nolte DL, Mason JR, Epple G, Aronov E, Campbell DL (1994) Why are predator urines aversive to prey? J Chem Ecol 20:1505–1516. https://doi.org/10.1007/bf02059876
Owings DH, Owings SC (1979) Snake-directed behavior by black-tailed prairie dogs (Cynomys ludovicianus). Z Tierpsychol 49:35–54. https://doi.org/10.1111/j.1439-0310.1979.tb00272.x
Peckarsky BL, Penton MA (1988) Why do Ephemerella nymphs scorpion posture: a “ghost of predation past”? Oikos 53:185–193. https://doi.org/10.2307/3566061
Powell F, Banks PB (2004) Do house mice modify their foraging behaviour in response to predator odours and habitat? Anim Behav 67:753–759. https://doi.org/10.1016/j.anbehav.2003.08.016
Quinn GP, Keough MJ (2002) Experimental design and data analysis for biologists. Cambridge university press, Cambridge
Ross AK, Letnic M, Blumstein DT, Moseby KE (2019) Reversing the effects of evolutionary prey naiveté through controlled predator exposure. J Appl Ecol 56:1761–1769
Saul WC, Jeschke JM (2015) Eco-evolutionary experience in novel species interactions. Ecol Lett 18:236–245. https://doi.org/10.1111/ele.12408
Schlaepfer MA, Sherman PW, Blossey B, Runge MC (2005) Introduced species as evolutionary traps. Ecol Lett 8:241–246. https://doi.org/10.1111/j.1461-0248.2005.00730.x
Southgate R, Christie P, Bellchambers K (2000) Breeding biology of captive, reintroduced and wild greater bilbies, Macrotis lagotis (Marsupialia: Peramelidae). Wildl Res 26:621–628. https://doi.org/10.1071/WR99104
Steindler LA, Blumstein DT, West R, Moseby KE, Letnic M (2018) Discrimination of introduced predators by ontogenetically naïve prey scales with duration of shared evolutionary history. Anim Behav 137:133–139. https://doi.org/10.1016/j.anbehav.2018.01.013
Turner AM, Turner SE, Lappi HM (2006) Learning, memory and predator avoidance by freshwater snails: effects of experience on predator recognition and defensive strategy. Anim Behav 72:1443–1450
Zhang S-J, Wang G-D, Ma P, Zhang L-L, Yin T-T, Liu Y-H, Otecko NO, Wang M, Ma Y-P, Wang L, Mao B, Savolainen P, Zhang Y-P (2020) Genomic regions under selection in the feralization of the dingoes. Nat Commun 11:671. https://doi.org/10.1038/s41467-020-14515-6
Zenger KR, Richardson BJ, Vachot-Griffin AM (2003) A rapid population expansion retains genetic diversity within European rabbits in Australia. Mol Ecol 12:789–794. https://doi.org/10.1046/j.1365-294X.2003.01759.x
Acknowledgements
We thank private pet owners (Edwina Ring), Calabash Kennels & Cattery (Iris Bleach) and the Sydney Cat Protection Society for supply of odours and Sam Fischer and Chris Mitchell (Queensland National Park staff) for their assistance with the study. We would like to thank the reviewers for the comments and feedback, as they have greatly improved the structure of the paper.
Funding
This research was funded by the Holsworth Wildlife Research Endowment (to LS and ML) and Australian Research Council grant LP130100173 (ML).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors delare that they have no conflicts of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This research was conducted according to University of New South Wales Animal Ethics approval 15/19A.
Informed consent
Written informed consent was obtained from the owners of dogs, cats and rabbits whose faeces were used in this experiment.
Additional information
Communicated by A. I Schulte-Hostedde
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 16 kb).
Rights and permissions
About this article
Cite this article
Steindler, L., Letnic, M. Not so naïve: endangered mammal responds to olfactory cues of an introduced predator after less than 150 years of coexistence. Behav Ecol Sociobiol 75, 8 (2021). https://doi.org/10.1007/s00265-020-02952-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-020-02952-8