Abstract
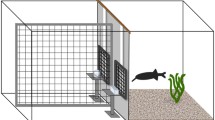
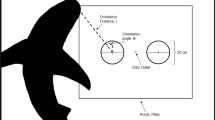
Pollimyrus adspersus discriminates the individually variable waveforms of Electric Organ Discharges (EODs) of conspecifics of only 150–250 μs duration. We examined: (1) the discrimination threshold for artificially generated EODs of similar waveform, (2) the mechanism of signal analysis (spectral vs temporal) present, by determining the discrimination between different waveforms of identical amplitude spectra, and (3) the threshold field intensity and reach of discrimination. The triphasic P. adspersus EOD waveform was artificially generated by superimposing two Gaussians, one wide, the second narrow, inverted, and of threefold amplitude. The natural variability among individual EOD waveforms was simulated by phase-shifting one Gaussian relative to the other. The symmetrical waveform where the peaks of the two Gaussians coincided was used as a reference (phase shift=0, rewarded stimulus S+). Results were: (1) in food-rewarded conditioning experiments, trained fish (N=7) detected a phase-shift in artificial EOD stimuli as low as 2 μs (N=2 fish), 6 μs (N=1) and 10 μs (N=1). (2) All fish tested (N=3) discriminated between artificial EODs of identical amplitude spectra but different waveforms (hence, different phase spectra), demonstrating a temporal mechanism of signal analysis. (3) The maximum reach of waveform discrimination was 130 cm at 4.9 μVp-p/cm and 100 μS/cm water conductivity (test signal generated at natural amplitude), that is, similar to the reach of EOD detection. Therefore, among the three kinds of electroreceptor organ present in mormyrids, we consider Knollenorgane the relevant sensory organs for EOD waveform discrimination.







Similar content being viewed by others
References
Bell CC (1989) Sensory coding and corollary discharge effects in mormyrid electric fish. J Exp Biol 146:229–253
Bell CC, Grant K (1992) Corollary discharge effects and sensory processing in the mormyromast regions of the mormyrid electrosensory lobe. II. Cell types and corollary discharge plasticity. J Neurophysiol 68:859–875
Bell CC, Russell CJ (1978) Effect of electric organ discharge on ampullary receptors in a mormyrid. Brain Res 145:85–96
Bell CC, Grant K, Serrier J (1992) Corollary discharge effects and sensory processing in the mormyromast regions of the mormyrid electrosensory lobe. I. Field intensities and cellular activity in associated structures. J Neurophysiol 68:843–858
Bell CC, Dunn K, Hall C, Caputi A (1995) Electric organ corollary discharge pathways in mormyrid fish. I. The mesencephalic command associated nucleus. J Comp Physiol A 177:449–462
Bennett MVL (1965) Electroreceptors in mormyrids. Cold Spring Harbor Symp Quant Biol 30:245–262
Bennett MVL (1967) Mechanisms of electroreception. In: Cahn P (ed) Lateral line detectors. Indiana University Press, Bloomington, pp 313–393
Bennett MVL (1971) Electroreception. In: Hoar WS, Randall DJ (ed) Fish physiology. Academic, London, pp 493–574
Bigorne R (1990) Révision systématique du genre Pollimyrus (Teleostei, Mormyridae) en Afrique de l’Ouest. Rev Hydrobiol Trop 23:313–327
Bratton BO, Kramer B (1988) Intraspecific variability of the pulse-type discharges of the African electric fishes, Pollimyrus isidori and Petrocephalus bovei (Mormyridae, Teleostei), and their dependence on water conductivity. Exp Biol 47:227–238
Bratton BO, Kramer B (1989) Patterns of the electric organ discharge during courtship and spawning in the mormyrid fish, Pollimyrus isidori. Behav Ecol Sociobiol 24:349–368
Carr CE, Maler L, Taylor B (1986) A time comparison circuit in the electric fish midbrain. II. Functional morphology. J Neurosci 6:1372–1383
Cochran WG, Cox GM (1968) Experimental designs. Wiley, New York
Crawford JD (1991) Sex recognition by electric cues in a sound-producing mormyrid fish, Pollimyrus isidori. Brain Behav Evol 38:20–38
Crawford JD (1992) Individual and sex specificity in the electric organ discharges of breeding mormyrid fish (Pollimyrus isidori). J Exp Biol 164:79–102
Crawford JD, Jacob P, Bénech V (1997) Sound production and reproductive ecology of strongly acoustic fish in Africa: Pollimyrus isidori, Mormyridae. Behaviour 134:677–725
Denizot J, Kirschbaum F, Schugardt C, Bensouilah M (1998) Larval electroreceptors indicate a larval electric system in mormyrids. Neurosci Lett 241:103–106
Graff C, Kramer B (1992) Trained weakly-electric fishes Pollimyrus isidori and Gnathonemus petersii (Mormyridae,Teleostei) discriminate between waveforms of electric pulse discharges. Ethology 90:279–292
Hopkins CD, Bass AH (1981) Temporal coding of species recognition signals in an electric fish. Science 212:85–87
Kaunzinger I, Kramer B (1995) Electrosensory stimulus-intensity thresholds in the weakly electric knifefish Eigenmannia: reduced sensitivity at harmonics of its own organ discharge. J Exp Biol 198:2365–2372
Kaunzinger I, Kramer B (1996) Electrosensory phase sensitivity in the weakly electric fish Eigenmannia in the detection of signals similar to its own. J Comp Physiol A 179:323–330
Kawasaki M, Guo YX (1996) Neural circuitry for comparison of timing in the electrosensory Lateral Line Lobe of the African wave-type electric fish Gymnarchus niloticus. J Neurosci 16:380–391
Kirschbaum F (1987) Reproduction and development of the weakly electric fish, Pollimyrus isidori (Mormyridae, Teleostei) in captivity. Environ Biol Fish 20:11–31
Knudsen EI (1975) Spatial aspects of the electric fields generated by weakly electric fish. J Comp Physiol A 99:103–118
Kramer B (1996) Electroreception and communication in fishes (Progress in zoology, vol 42). Fischer, Stuttgart
Kramer B (1999) Waveform discrimination, phase sensitivity and jamming avoidance in a wave-type electric fish. J Exp Biol 202:1387–1398
Kramer B, Kuhn B (1993) Electric signalling and impedance matching in a variable environment: the electric organ of a mormyrid fish actively adapts to changes in water conductivity. Naturwissenschaften 80:43–46
Kramer B, Westby GWM (1985) No sex difference in the waveform of the pulse type electric fish, Gnathonemus petersii (Mormyridae). Experientia 41:1530–1531
Kramer B, Weymann D (1987) A microprocessor system for the digital synthesis of pulsed and continuous discharges of electric fish (or animal vocalizations). Behav Brain Res 23:167–174
Krebs J, Ashcroft R, Webber M (1978) Song repertoires and territory defence in the great tit. Nature 271:539–542
Lücker, H, Kramer B (1981) Development of a sex difference in the preferred latency response in the weakly electric fish, Pollimyrus isidori (Cuvier et Valenciennes) (Mormyridae, Teleostei). Behav Ecol Sociobiol 9:103–109
McGregor PK, Westby GWM (1992) Discrimination of individually characteristic electric organ discharges by a weakly electric fish. Anim Behav 43:977–986
Moiseff A, Konishi M (1981) Neuronal and behavioral sensitivity to binaural time difference in the owl. J Neurosci 1:40–48
Moller P (1995) Electric fishes. History and behavior. Chapman & Hall, London
Moller P, Serrier J, Belbenoit P, Push S (1979) Notes on ethology and ecology of the Swashi River mormyrids (Lake Kainji, Nigeria). Behav Ecol Sociobiol 4:357–368
Moller P, Serrier J, Bowling D (1989) Electric organ discharge displays during social encounter in the weakly electric fish Brienomyrus niger L. (Mormyridae). Ethology 82:177–191
Postner M, Kramer B (1995) Electrosensory thresholds in larvae of the weakly electric fish Pollimyrus isidori (Mormyridae, Teleostei) during ontogeny. J Exp Biol 198:783–791
Rose G, Heiligenberg W (1985) Temporal hyperacuity in the electric sense of fish. Nature 318:178–180
Russell CJ, Bell CC (1978) Neuronal responses to electrosensory input in mormyrid Valvula Cerebelli. J Neurophysiol 41:1495–1510
Scheffel A, Kramer B (1997) Electrocommunication and social behaviour in Marcusenius senegalensis (Mormyridae, Teleostei). Ethology 103:404–420
Scheffel A, Kramer B (2000) Electric signals in the social behavior of sympatric elephant fish (Mormyridae, Teleostei) from the Upper Zambezi River. Naturwissenschaften 87:142–147
Squire A, Moller P (1982) Effects of water conductivity on electrocommunication in the weak-electric fish Brienomyrus niger (Mormyriformes). Anim Behav 30:375–382
Szabo T, Fessard A (1974) Physiology of electroreceptors. In: Fessard A (ed) Handbook of sensory physiology, vol III/3. Springer, Berlin Heidelberg New York, pp 59–124
Takahashi T, Moiseff A, Konishi M (1984) Time and intensity cues are processed independently in the auditory system of the owl. J Neurosci 4:1781–1786
Westby GWM (1984) Simple computer model accounts for observed individual and sex differences in electric fish signals. Anim Behav 32:1254–1256
Westby GWM, Kirschbaum F (1982) Sex differences in the waveform of the pulse-type electric fish, Pollimyrus isidori (Mormyridae). J Comp Physiol A 145:399–403
Zakon HH (1986) The electroreceptive periphery. In: Bullock TH, Heiligenberg W (eds) Electroreception. Wiley, New York, pp 103–156
Zipser B, Bennett MVL (1976a) Responses of cells of posterior lateral line lobe to activation of electroreceptors in a mormyrid fish. J Neurophysiol 39:693–712
Zipser B, Bennett MVL (1976b) Interaction of electrosensory and electromotor signals in lateral line lobe of a mormyrid fish. J Neurophysiol 39:713–721
Acknowledgements
G.W.M. Westby’s computer program for generating artificial Pollimyrus EODs written for our MINC computer (DEC) was rewritten for IBM-compatible computers by S.P. and Helge Knüttel; we gratefully acknowledge G.W.M.W’s and H.K’s expertise and kind help. H.K. also assisted in developing additional set-up orientated computer programs. We wish to thank Dieter Weymann, electronics workshop of the Faculty of biology, for expert electronic assistance, including the construction of the DAM stimulator (see Methods). S.P. would like to thank J.M. Burzler for advice on continuous Fourier Transformations, and B. Laggerbauer for helpful comments on an earlier version of the manuscript. Susanne Hanika and Birgit Steib read earlier versions of the study. Lars Schmidt-Eisenlohr determined the EOD waveform discrimination limen in one fish during his practical in our laboratory. Our experiments comply with the “Principles of animal care”, publication no. 86–23, revised 1985 of the National Institute of Health, and also the current laws for experimentation in Germany. Support was given by the Deutsche Forschungsgemeinschaft (Kr 446/10).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J. Krause
Rights and permissions
About this article
Cite this article
Paintner, S., Kramer, B. Electrosensory basis for individual recognition in a weakly electric, mormyrid fish, Pollimyrus adspersus (Günther, 1866). Behav Ecol Sociobiol 55, 197–208 (2003). https://doi.org/10.1007/s00265-003-0690-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-003-0690-4