Abstract
Purpose
The aim of this study was to evaluate the effects of a high initial-dose (60 mg/kg) intravenous tranexamic acid (IV-TXA) on fibrinolysis and inflammation after total knee arthroplasty (TKA).
Methods
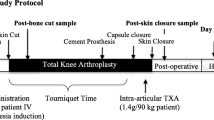
A total of 132 patients were categorized into two groups based on different TXA regimens: 20 mg/kg before incision (A) or 60 mg/kg before incision (B). All patients received five doses of 1 g TXA at three, six, 12, 18, and 24 hours after the first dose. The primary outcomes were peri-operative blood loss and transfusion rate. Other outcome measurements such as, haemoglobin level, fibrinolysis parameters [fibrin(−ogen) degradation products (FDP), D-dimer], inflammatory factors [C-reactive protein (CRP), interleukin-6 (IL-6)], visual analog scale (VAS) score, consumption of analgesic rescue, coagulation parameters [activated partial thromboplastin time (APTT), prothrombin time (PT), platelet count, thrombelastography (TEG), and anti-factor Xa activity (AFXa)] and complications, were also compared.
Results
There was a favourable effect in reducing peri-operative blood loss and transfusion rate for patients in Group B, compared with patients in Group A. In addition, the levels of FDP, D-dimer, CRP, IL-6, and dynamic pain in Group B were significantly lower than those in Group A on post-operative days one, two and three. There were no statistically significant differences in postoperative coagulation parameters and complications between the two groups.
Conclusion
A high initial-dose (60 mg/kg) IV-TXA before surgery followed by five doses can further reduce blood loss, provide additional fibrinolysis and inflammation control, and ameliorate post-operative pain following TKA, without increasing the risk of treatment-related complications.







Similar content being viewed by others
References
Xie J, Ma J, Yao H, Yue C, Pei F (2016) Multiple boluses of intravenous Tranexamic acid to reduce hidden blood loss after primary Total knee Arthroplasty without tourniquet: a randomized clinical trial. J Arthroplast. https://doi.org/10.1016/j.arth.2016.04.034
Lei Y, Xie J, Xu B, Xie X, Huang Q, Pei F (2017) The efficacy and safety of multiple-dose intravenous tranexamic acid on blood loss following total knee arthroplasty: a randomized controlled trial. Int Orthop 41(10):2053–2059. https://doi.org/10.1007/s00264-017-3519-x
Liu KL, Chen IH, Wen SH (2016) Low dose tranexamic acid reduces blood transfusion rate after total knee arthroplasty: a population-based study in Taiwan. J Formos Med Assoc. https://doi.org/10.1016/j.jfma.2015.12.012
Rineau E, Chaudet A, Chassier C, Bizot P, Lasocki S (2016) Implementing a blood management protocol during the entire perioperative period allows a reduction in transfusion rate in major orthopedic surgery: a before-after study. Transfusion 56(3):673–681. https://doi.org/10.1111/trf.13468
Xie J, Hu Q, Ma J, Huang Q, Pei F (2017) Multiple boluses of intravenous tranexamic acid to reduce hidden blood loss and the inflammatory response following enhanced-recovery primary total hip arthroplasty: a randomised clinical trial. Bone Joint J 99-b(11):1442–1449. https://doi.org/10.1302/0301-620x.99b11.bjj-2017-0488.r1
Morrison RJM, Tsang B, Fishley W, Harper I, Joseph JC, Reed MR (2017) Dose optimisation of intravenous tranexamic acid for elective hip and knee arthroplasty: the effectiveness of a single pre-operative dose. Bone Joint Res 6(8):499–505. https://doi.org/10.1302/2046-3758.68.bjr-2017-0005.r1
Maniar RN, Kumar G, Singhi T, Nayak RM, Maniar PR (2012) Most effective regimen of tranexamic acid in knee arthroplasty: a prospective randomized controlled study in 240 patients. Clin Orthop Relat Res 470(9):2605–2612. https://doi.org/10.1007/s11999-012-2310-y
Xie J, Hu Q, Huang Q, Ma J, Lei Y, Pei F (2017) Comparison of intravenous versus topical tranexamic acid in primary total hip and knee arthroplasty: an updated meta-analysis. Thromb Res 153:28–36. https://doi.org/10.1016/j.thromres.2017.03.009
Akgul T, Buget M, Salduz A, Edipoglu IS, Ekinci M, Kucukay S, Sen C (2016) Efficacy of preoperative administration of single high dose intravenous tranexamic acid in reducing blood loss in total knee arthroplasty: a prospective clinical study. Acta Orthop Traumatol Turc 50(4):429–431. https://doi.org/10.1016/j.aott.2016.06.007
Whiting DR, Sierra RJ (2015) Efficacy of combined use of intraarticular and intravenous tranexamic acid in total knee arthroplasty. Ann Transl Med 3(Suppl 1):S39
Zhang S, Xie J, Cao G, Lei Y, Huang Q, Pei F (2019) Six-dose intravenous Tranexamic acid regimen further inhibits postoperative fibrinolysis and reduces hidden blood loss following Total knee Arthroplasty. J Knee Surg. https://doi.org/10.1055/s-0039-1694768
Blanie A, Bellamy L, Rhayem Y, Flaujac C, Samama CM, Fontenay M, Rosencher N (2013) Duration of postoperative fibrinolysis after total hip or knee replacement: a laboratory follow-up study. Thromb Res 131(1):e6–e11. https://doi.org/10.1016/j.thromres.2012.11.006
Huang Z, Xie X, Li L, Huang Q, Ma J, Shen B, Kraus VB, Pei F (2017) Intravenous and topical Tranexamic acid alone are superior to tourniquet use for primary Total knee Arthroplasty: a prospective, randomized controlled trial. J Bone Joint Surg Am 99(24):2053–2061. https://doi.org/10.2106/jbjs.16.01525
Nadler SB, Hidalgo JH, Bloch T (1962) Prediction of blood volume in normal human adults. Surgery 51(2):224–232
Gross JB (1983) Estimating allowable blood loss: corrected for dilution. Anesthesiology 58(3):277–280
Kathariya R (2013) Intra-operative hemorrhage: a review of literature. J Med Diagn Methods 02(6):146. https://doi.org/10.4172/2168-9784.1000146
Sehat KR, Evans RL, Newman JH (2004) Hidden blood loss following hip and knee arthroplasty. Correct management of blood loss should take hidden loss into account. J Bone Joint Surg-British 86(4):561–565
Wei MY, Ward SM (2015) The anti-factor Xa range for low molecular weight heparin Thromboprophylaxis. Hematol Rep 7(4):5844. https://doi.org/10.4081/hr.2015.5844
Kundu R, Das A, Basunia SR, Bhattacharyya T, Chattopadhyay S, Mukherjee A (2015) Does a single loading dose of tranexamic acid reduce perioperative blood loss and transfusion requirements after total knee replacement surgery? A randomized, controlled trial. J Nat Sci Biol Med 6(1):94–99. https://doi.org/10.4103/0976-9668.149099
Holt JB, Miller BJ, Callaghan JJ, Clark CR, Willenborg MD, Noiseux NO (2016) Minimizing blood transfusion in Total hip and knee Arthroplasty through a multimodal approach. J Arthroplast 31(2):378–382. https://doi.org/10.1016/j.arth.2015.08.025
Brito SA, Rankin EA, McNear M (2016) Acute blood loss Anemia in the octogenarian Total knee Arthroplasty, estimated blood loss and transfusions rates. J Natl Med Assoc 108(1):86–89. https://doi.org/10.1016/j.jnma.2015.12.011
Iwai T, Tsuji S, Tomita T, Sugamoto K, Hideki Y, Hamada M (2013) Repeat-dose intravenous tranexamic acid further decreases blood loss in total knee arthroplasty. Int Orthop 37(3):441–445. https://doi.org/10.1007/s00264-013-1787-7
Huang GP, Jia XF, Xiang Z, Ji Y, Wu GY, Tang Y, Li J, Zhang J (2016) Tranexamic acid reduces hidden blood loss in patients undergoing Total knee Arthroplasty: a comparative study and meta-analysis. Med Sci Monit 22:797–802
Lee SY, Chong S, Balasubramanian D, Na YG, Kim TK (2017) What is the ideal route of Administration of Tranexamic Acid in TKA? A randomized controlled trial. Clin Orthop Relat Res 475(8):1987–1996. https://doi.org/10.1007/s11999-017-5311-z
Bidolegui F, Arce G, Lugones A, Pereira S, Vindver G (2014) Tranexamic acid reduces blood loss and transfusion in patients undergoing Total knee Arthroplasty without tourniquet: a prospective randomized controlled trial. Open Orthop J 8(1):250
Jain NP, Nisthane PP, Shah NA (2016) Combined Administration of Systemic and Topical Tranexamic Acid for Total knee Arthroplasty: can it be a better regimen and yet safe? A randomized controlled trial. J Arthroplast 31(2):542–547
Hogan CA, Golightly LK, Phong S, Dayton MR, Lyda C, Barber GR (2016) Perioperative blood loss in total hip and knee arthroplasty: Outcomes associated with intravenous tranexamic acid use in an academic medical center. SAGE Open Med 4:4/0/2050312116637024
Hourlier H, Reina N, Fennema P (2015) Single dose intravenous tranexamic acid as effective as continuous infusion in primary total knee arthroplasty: a randomised clinical trial. Arch Orthop Trauma Surg 135(4):465–471. https://doi.org/10.1007/s00402-015-2168-z
Jain NP, Nisthane PP, Shah NA (2016) Combined Administration of Systemic and Topical Tranexamic Acid for Total knee Arthroplasty: can it be a better regimen and yet safe? A randomized controlled trial. J Arthroplast 31(2):542–547. https://doi.org/10.1016/j.arth.2015.09.029
Rajesparan K, Biant LC, Ahmad M, Field RE (2009) The effect of an intravenous bolus of tranexamic acid on blood loss in total hip replacement. J Bone Joint Surg (Br) 91(6):776–783. https://doi.org/10.1302/0301-620x.91b6.22393
Claeys MA, Vermeersch N, Haentjens P (2007) Reduction of blood loss with tranexamic acid in primary total hip replacement surgery. Acta Chir Belg 107(4):397–401
Dowd NP, Karski JM, Cheng DC, Carroll JA, Lin Y, James RL, Butterworth J (2002) Pharmacokinetics of tranexamic acid during cardiopulmonary bypass. Anesthesiology 97(2):390–399
Jimenez JJ, Iribarren JL, Brouard M, Hernandez D, Palmero S, Jimenez A, Lorente L, Machado P, Borreguero JM, Raya JM, Martin B, Perez R, Martinez R, Mora ML (2011) Safety and effectiveness of two treatment regimes with tranexamic acid to minimize inflammatory response in elective cardiopulmonary bypass patients: a randomized double-blind, dose-dependent, phase IV clinical trial. J Cardiothorac Surg 6:138. https://doi.org/10.1186/1749-8090-6-138
Bhat A, Bhowmik DM, Vibha D, Dogra M, Agarwal SK (2014) Tranexamic acid overdosage-induced generalized seizure in renal failure. Saudi J Kidney Dis Transpl 25(1):130–132
Murkin JM, Florian F, Jeff G, Bryan Y, Christiana B, Michael C (2010) High-dose tranexamic acid is associated with nonischemic clinical seizures in cardiac surgical patients. Anesth Analg 110(2):350
Johnson DJ, Johnson CC, Goobie SM, Nami N, Wetzler JA, Sponseller PD, Frank SM (2017) High-dose versus low-dose Tranexamic acid to reduce transfusion requirements in pediatric scoliosis surgery. J Pediatr Orthop 37(8):e552–e557. https://doi.org/10.1097/bpo.0000000000000820
Jules-Elysee KM, Lipnitsky JY, Patel N, Anastasian G, Wilfred SE, Urban MK, Sculco TP (2011) Use of low-dose steroids in decreasing cytokine release during bilateral total knee replacement. Reg Anesth Pain Med 36(1):36–40. https://doi.org/10.1097/AAP.0b013e31820306c5
Bautmans I, Njemini R, De Backer J, De Waele E, Mets T (2010) Surgery-induced inflammation in relation to age, muscle endurance, and self-perceived fatigue. J Gerontol A Biol Sci Med Sci 65(3):266–273. https://doi.org/10.1093/gerona/glp145
Jimenez JJ, Iribarren JL, Lorente L, Rodriguez JM, Hernandez D, Nassar I, Perez R, Brouard M, Milena A, Martinez R, Mora ML (2007) Tranexamic acid attenuates inflammatory response in cardiopulmonary bypass surgery through blockade of fibrinolysis: a case control study followed by a randomized double-blind controlled trial. Crit Care 11(6):R117. https://doi.org/10.1186/cc6173
Robson SC, Shephard EG, Kirsch RE (1994) Fibrin degradation product D-dimer induces the synthesis and release of biologically active IL-1 beta, IL-6 and plasminogen activator inhibitors from monocytes in vitro. Br J Haematol 86(2):322–326
Robertshaw HJ (2008) An anti-inflammatory role for tranexamic acid in cardiac surgery? Crit Care 12(1):105. https://doi.org/10.1186/cc6210
Later AF, Sitniakowsky LS, van Hilten JA, van de Watering L, Brand A, Smit NP, Klautz RJ (2013) Antifibrinolytics attenuate inflammatory gene expression after cardiac surgery. J Thorac Cardiovasc Surg 145(6):1611–1616, 1616.e1611-1614. https://doi.org/10.1016/j.jtcvs.2012.11.042
Wasko MK, Bobecka-Wesolowska K, Tomasiuk R, Kowalczewski J (2015) Measurement of the inflammatory response in the early postoperative period after hip and knee arthroplasty. Clin Chem Lab Med 53(11):1785–1792. https://doi.org/10.1515/cclm-2014-1055
De Oliveira GS Jr, Almeida MD, Benzon HT, McCarthy RJ (2011) Perioperative single dose systemic dexamethasone for postoperative pain: a meta-analysis of randomized controlled trials. Anesthesiology 115(3):575–588. https://doi.org/10.1097/ALN.0b013e31822a24c2
Fowler SJ, Symons J, Sabato S, Myles PS (2008) Epidural analgesia compared with peripheral nerve blockade after major knee surgery: a systematic review and meta-analysis of randomized trials. Br J Anaesth 100(2):154–164. https://doi.org/10.1093/bja/aem373
Si HB, Yang TM, Zeng Y, Zhou ZK, Pei FX, Lu YR, Cheng JQ, Shen B (2017) Correlations between inflammatory cytokines, muscle damage markers and acute postoperative pain following primary total knee arthroplasty. BMC Musculoskelet Disord 18(1):265. https://doi.org/10.1186/s12891-017-1597-y
Gillette BP, DeSimone LJ, Trousdale RT, Pagnano MW, Sierra RJ (2013) Low risk of thromboembolic complications with tranexamic acid after primary total hip and knee arthroplasty. Clin Orthop Relat Res 471(1):150–154. https://doi.org/10.1007/s11999-012-2488-z
Kim YH, Park JW, Kim JS, Seo DH (2018) Does tranexamic acid increase the risk of thromboembolism after bilateral simultaneous total knee arthroplasties in Asian population? Arch Orthop Trauma Surg 138(1):83–89. https://doi.org/10.1007/s00402-017-2791-y
Xie J, Ma J, Kang P, Zhou Z, Shen B, Yang J, Pei F (2015) Does tranexamic acid alter the risk of thromboembolism following primary total knee arthroplasty with sequential earlier anticoagulation? A large, single center, prospective cohort study of consecutive cases. Thromb Res 136(2):234–238. https://doi.org/10.1016/j.thromres.2015.05.014
Toshio F, Yuko H, Chikako K, Yoshiyuki M, Toshiro S (2012) Reversal of anticoagulant effects of edoxaban, an oral, direct factor Xa inhibitor, with haemostatic agents. Thromb Haemost 107(02):253–259
Hamim Z, Brown KS, Vandell AG, Madhuri D, Jen-Fue M, Victor D, Barbara L, Annette F, Wenqin F, Ling H (2015) Edoxaban effects on bleeding following punch biopsy and reversal by a 4-factor prothrombin complex concentrate. Circulation 131(1):82–90
Eva H, Franz K, Wilfried K, Baerbel D, Jochen MC, Ingo P, Yoshiyuki M, Gerhard D (2015) Effective reversal of edoxaban-associated bleeding with four-factor prothrombin complex concentrate in a rabbit model of acute hemorrhage. Anesthesiology 122(2):387–398
Honda Y, Furugohri T, Morishima Y (2018) Tranexamic acid failed to reverse the anticoagulant effect and bleeding by an Oral direct factor Xa inhibitor edoxaban. Pharmacology 101(1–2):92–95. https://doi.org/10.1159/000484172
Levy JH, Moore KT, Neal MD, Schneider D, Marcsisin VS, Ariyawansa J, Weitz JI (2018) Rivaroxaban reversal with prothrombin complex concentrate or tranexamic acid in healthy volunteers. J Thromb Haemost 16(1):54–64. https://doi.org/10.1111/jth.13894
Chen BY, Zhaoqing W, Charles F, Andrew S (2010) Clinical laboratory measurement of direct factor Xa inhibitors: anti-Xa assay is preferable to prothrombin time assay. Thromb Haemost 104(6):1263–1271
Guervil DJ, Rosenberg AF, Winterstein AG, Harris NS, Johns TE, Zumberg MS (2011) Activated partial thromboplastin time versus antifactor Xa heparin assay in monitoring unfractionated heparin by continuous intravenous infusion. Ann Pharmacother 45(7–8):861
Helviz Y, Dzigivker I, Raveh-Brawer D, Hersch M, Zevin S, Einav S (2016) Anti-factor Xa activity of prophylactic enoxaparin regimens in critically ill patients. Isr Med Assoc J 18(2):108–113
Ali Algadiem E, Aleisa AA, Alsubaie HI, Buhlaiqah NR, Algadeeb JB, Alsneini HA (2016) Blood loss estimation using gauze visual analogue. Trauma Mon 21(2):e34131. https://doi.org/10.5812/traumamon.34131
Acknowledgments
We acknowledge the nursing staffs from Department of Orthopedics Surgery, West China Hospital and the patients for their support during the study period. We thank Prof. Dan Deng (Department of Health Statistics and Information Management, College of Public Health and Management, Chongqing Medical University), for statistical counseling. Yi-Ting Lei wants to thank, in particular, the patience, care and support from Dan-Li Cui over the past few years.
Funding
This study was funded by the National Health and Family Planning Commission of the People’s Republic of China (CN) program (201302007).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical approval
This trial was approved by the institutional review board (2017–128) and registered the International Clinical Trial Registry (ChiCTR1800016640).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Level of evidence: I
Rights and permissions
About this article
Cite this article
Lei, YT., Xie, JW., Huang, Q. et al. The antifibrinolytic and anti-inflammatory effects of a high initial-dose tranexamic acid in total knee arthroplasty: a randomized controlled trial. International Orthopaedics (SICOT) 44, 477–486 (2020). https://doi.org/10.1007/s00264-019-04469-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-019-04469-w