Abstract
Introduction
The Masquelet procedure proved its efficiency in treating infected nonunion filling bony gaps up to 25 cm. Yet the use of local antibiotics is still questionable in the daily practice with lack of evidence regarding its usefulness in controlling infection. An experimental rat model is put in place to study the antibacterial properties of the induced membrane produced during the first stage of Masquelet.
Method
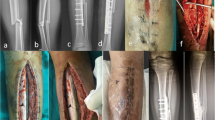
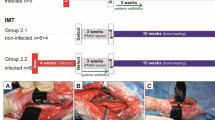
Twenty-three-month-old wistar male rats are inoculated with a 0.5 mL solution of 10^8 CFU/mL MRSA over a critical fracture done on the right femur. Six weeks later, remaining 11 rats exhibiting signs of a chronic infection with a sinus tract and oozing pus along with radiological nonunion are used for a first stage Masquelet procedure. They are randomly divided into two groups with six rats having no local antibiotic in the cement mixture and five rats having 3 g of vancomycin mixed with gentamycin loaded cement. Six weeks later (twelve weeks from baseline), all eleven rats are euthanized and blood samples for C-reactive protein are withdrawn. The induced membrane is identified and resected along with bone fragments and sent for cultures and pathology.
Results
MRSA is isolated in the cultures of all six rats in the first group where no local antibiotic was added. Altered polymorphonuclears with abscess and pus are noted on four of six pathology samples. However in the second group where local antibiotics were added, three out of five rats exhibited eradication of MRSA (p = 0.034) and all samples did not exhibit clear infection signs on pathology. A pyo-epithelioid over a foreign body reaction is seen predominantly in this group demonstrating a regenerative process.
Discussion
The induced membrane does not have antimicrobial properties capable of overcoming an infected nonunion on its own. When local antibiotics were added during the first stage of the Masquelet procedure, new bone formation occurred indicating the need to control an infection in order for bone union to occur.
Conclusion
Local antibiotics use in adjunction to extensive debridement is advisable during the first stage of a Masquelet procedure for an infected nonunion.






Similar content being viewed by others
References
Cook GE, Markel DC, Ren W et al (2015) Infection in orthopaedics. J Orthop Trauma 29(Suppl 1):S19–S23. https://doi.org/10.1097/BOT.0000000000000461
Perry KI, Hanssen AD (2017) Orthopaedic infection: prevention and diagnosis. J Am Acad Orthop Surg 25:S4–S6. https://doi.org/10.5435/JAAOS-D-16-00634
Rodriguez-Merchan EC, Forriol F (2004) Nonunion: general principles and experimental data. Clin Orthop Relat Res:4–12. https://doi.org/10.1097/01.blo.0000118182.32042.1a
Bose D, Stubbs D, Mcnally M Management of infected nonunion of the long bones by a multidisciplinary team. Bone Joint J:814–817. https://doi.org/10.1302/0301-620X.97B6.33276
Patzakis MJ, Zalavras CG (2005) Chronic posttraumatic osteomyelitis and infected nonunion of the tibia: current management concepts. J Am Acad Orthop Surg 13:417–427
Struijs PAA, Poolman RW, Bhandari M (2007) Infected nonunion of the long bones. J Orthop Trauma 21:507–511. https://doi.org/10.1097/BOT.0b013e31812e5578
Rose REC, Palmer WSO (2007) The Illizarov method in infected non-union of long bones. West Indian Med J 56:246–251. https://doi.org/10.1097/BOT.0b013e31812e5578
Motsitsi NS (2008) Management of infected nonunion of long bones: the last decade (1996-2006). Injury 39:155–160. https://doi.org/10.1016/j.injury.2007.08.032
Pelissier P, Masquelet AC, Bareille R et al (2004) Induced membranes secrete growth factors including vascular and osteoinductive factors and could stimulate bone regeneration. J Orthop Res 22:73–79. https://doi.org/10.1016/S0736-0266(03)00165-7
Han W, Shen J, Wu H et al (2017) Induced membrane technique: advances in the management of bone defects. Int J Surg 42:110–116. https://doi.org/10.1016/j.ijsu.2017.04.064
Masquelet AC, Begue T (2010) The concept of induced membrane for reconstruction of long bone defects. Orthop Clin North Am 41:27–37. https://doi.org/10.1016/j.ocl.2009.07.011
Taylor BC, French BG, Ty Fowler TT et al (2012) Induced membrane technique for reconstruction to manage bone loss. J Am Acad Orthop Surg 20:142–150
El-Alfy BS, Ali AM (2015) Management of segmental skeletal defects by the induced membrane technique. Indian J Orthop 49:643. https://doi.org/10.4103/0019-5413.168757
Lovati AB, Romanò CL, Bottagisio M et al (2016) Modeling staphylococcus epidermidis-induced non-unions: subclinical and clinical evidence in rats. PLoS One 11:1–19. https://doi.org/10.1371/journal.pone.0147447
Lovati AB, Drago L, Bottagisio M et al (2016, 2016) Systemic and local administration of antimicrobial and cell therapies to prevent methicillin-resistant Staphylococcus epidermidis-induced femoral nonunions in a rat model. Mediators Inflamm. https://doi.org/10.1155/2016/9595706
Chen X, Tsukayama DT, Kidder LS et al (2005) Characterization of a chronic infection in an internally-stabilized segmental defect in the rat femur. J Orthop Res 23:816–823. https://doi.org/10.1016/j.orthres.2005.01.009
Nana A, Nelson SB, Mclaren A, Chen AF (2016) What’s new in musculoskeletal infection : update on biofilms. J Bone Joint Surg Am 98:1226–1234
Mills L, Tsang J, Hopper G et al (2016) The multifactorial aetiology of fracture nonunion and the importance of searching for latent infection. Bone Jt Res 5:512–519. https://doi.org/10.1302/2046-3758.510.BJR-2016-0138
Stucken C, Olszewski DC, Creevy WR et al (2013) Preoperative diagnosis of infection in patients with nonunions. J Bone Jt Surgery-American Vol 95:1409–1412. https://doi.org/10.2106/JBJS.L.01034
Rand BCC, Penn-Barwell JG, Wenke JC (2015) Combined local and systemic antibiotic delivery improves eradication of wound contamination: an animal experimental model of contaminated. Bone Jt J 97-B:1423–1427. https://doi.org/10.1302/0301-620X.97B10.35651
Masquelet A (2008) Le concept de membrane induite pour la reconstruction des pertes de substance des os longs. The reconstruction of wide diaphyseal bone defect by foreign body induced membrane and bone graft. E-memoires l’Academie Natl Chir 7:34–38
Karger C, Kishi T, Schneider L et al (2012) Treatment of posttraumatic bone defects by the induced membrane technique. Orthop Traumatol Surg Res 98:97–102. https://doi.org/10.1016/j.otsr.2011.11.001
Xie Z, Cui X, Zhao C et al (2013) Gentamicin-loaded borate bioactive glass eradicates osteomyelitis due to Escherichia coli in a rabbit model. Antimicrob Agents Chemother 57:3293–3298. https://doi.org/10.1128/AAC.00284-13
Branstetter JG, Jackson SR, Haggard WO et al (2009) Locally-administered antibiotics in wounds in a limb. J Bone Jt Surg - Br Vol 91-B:1106–1109. https://doi.org/10.1302/0301-620X.91B8.22216
Bhatia C, Tiwari AK, Sharma SB et al (2017) Role of antibiotic cement coated nailing in infected nonunion of tibia. Malaysian Orthop J 11:6–11. https://doi.org/10.5704/MOJ.1703.019
McNally MA, Ferguson JY, Lau ACK et al (2016) Single-stage treatment of chronic osteomyelitis with a new absorbable, gentamicin-loaded, calcium sulphate/hydroxyapatite biocomposite: a prospective series of 100 cases. Bone Jt J 98-B:1289–1296. https://doi.org/10.1302/0301-620X.98B9.38057
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Level of Evidence
A randomized controlled experimental study
Rights and permissions
About this article
Cite this article
Roukoz, S., El Khoury, G., Saghbini, E. et al. Does the induced membrane have antibacterial properties? An experimental rat model of a chronic infected nonunion. International Orthopaedics (SICOT) 44, 391–398 (2020). https://doi.org/10.1007/s00264-019-04453-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-019-04453-4