Abstract
Purpose
The lysyl oxidase (LOX) family plays a crucial role in the formation and stabilisation of extracellular matrix (ECM) by catalysing the cross-linking of collagen and elastin, implicating its important fundamental roles in injury healing. A high level of transforming growth factor-β1 (TGF-β1) accompanies the inflammatory phase of an injury of the knee joint. Our purpose was to detect the expressions of the LOX family in anterior cruciate ligament (ACL) and medial collateral ligament (MCL) response to TGF-β1.
Methods
This study used reversed transcript PCR, real time quantitative PCR and Western blot for analyses.
Results
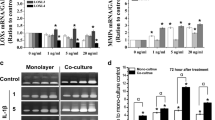
The results showed significant increases in mRNA levels of LOX family members. At 5 ng/ml concentration of TGF-β1, the gene profiles of LOXs showed most active, and LOX and LOXL-3 showed increasing peaks at 12 hours after TGF-β1 treatment (LOX: 7.2, 8.8-fold and LOXL-3: 3.8, 5.4-fold compared with normal controls in ACL and MCL, respectively); LOXL-1, LOXL-2 and LOXL-4 reached their highest amounts at six hours (LOXL-1: 1.9, 2.4-fold; LOXL-2: 14.8, 16.2-fold; LOXL-4: 2.5, 4.4-fold in ACL and MCL, respectively). Protein assays revealed that LOXs in ACL cells had relatively lower response to TGF-β1 compared with those in MCL cells.
Conclusions
The differential expression and activities of LOXs might help to explain the intrinsic difference between ACL and MCL, and LOXs could imply a potential capability for ACL healing.





Similar content being viewed by others
References
Nebelung W, Wuschech H (2005) Thirty-five years of follow-up of anterior cruciate ligament-deficient knees in high-level athletes. Arthroscopy 21(6):696–702
Legnani C, Ventura A, Terzaghi C, et al (2010) Anterior cruciate ligament reconstruction with synthetic grafts. A review of literature. Int Orthop 34(4):465–471
Furumatsu T, Hachioji M, Saiga K, et al. (2010) Anterior cruciate ligament-derived cells have high chondrogenic potential. Biochem Biophys Res Commun 391(1):1142–1147
Tang Z, Yang L, Wang Y, et al (2008) Contributions of different intraarticular tissues to the acute phase elevation of synovial fluid MMP-2 following rat ACL rupture. J Orthop Res 27(2):243–248
Inoue M, Kratz G, Haegerstrand A, Stahle-Backdahl M (1995) Collagenase expression is rapidly induced in wound edge keratinocytes after acute injury in human skin, persists during healing, and stops at re-epithelialization. J Invest Derm 104(4):479–483
Tang Z, Yang L, Xue R, et al (2009) Differential expression of matrix metalloproteinases and tissue inhibitors of metalloproteinases in anterior cruciate ligament and medial collateral ligament fibroblasts after a mechanical injury: Involvement of the p65 subunit of NF-kappa B. Wound Repair Regen 17(5):709–716
Meaney Murray MM, Rice K, Wright RJ, Spector M (2003) The effect of selected growth factors on human anterior cruciate ligament cell interactions with a three-dimensional collagen-GAG scaffold. J Orthop Res 21(2):238–244
Zhang Y, Cheng X, Wang J, Wang Y, Shi B, Huang C et al (2006) Novel chitosan/collagen scaffold containing transforming growth factor-beta1 DNA for periodontal tissue engineering. Biochem Biophys Res Commun 344(1):362–369
Shao H-J, Lee YT, Chen CS, et al (2010) Modulation of gene expression and collagen production of anterior cruciate ligament cells through cell shape changes on polycaprolactone/chitosan blends. Biomaterials 31(17):4695–4705
Kim Y, Boyd CD, Csiszar K (1995) A new gene with sequence and structural similarity to the gene encoding human lysyl oxidase. J Biol Chem 270(13):7176–7182
Saito H, Papaconstantinou J, Sato H, Goldstein S (1997) Regulation of a novel gene encoding a lysyl oxidase-related protein in cellular adhesion and senescence. J Biol Chem 272(13):8157–8160
Jang W, Hua A, Spilson SV, Miller W, Roe BA, Meisler MH (1999) Comparative sequence of human and mouse BAC clones from the mnd2 region of chromosome 2p13. Genome Res 9(1):53–61
Maki JM, Tikkanen H, Kivirikko KI (2001) Cloning and characterization of a fifth human lysyl oxidase isoenzyme: the third member of the lysyl oxidase-related subfamily with four scavenger receptor cysteine-rich domains. Matrix Biol 20(7):493–496
Kagan HM, Li W (2003) Lysyl oxidase: properties, specificity, and biological roles inside and outside of the cell. J Cell Biochem 88(4):660–672
Csiszar K (2001) Lysyl oxidases: A novel multifunctional amine oxidase family. Prog Nucleic Acid Res Mol Biol 70(70):1–32
Lucero HA, Kagan HM (2006) Lysyl oxidase: an oxidative enzyme and effector of cell function. Cell Mol Life Sci 63(19–20):2304–2316
Goto Y, Uchio-Yamada K, Anan S, et al (2005) Transforming growth factor-beta1 mediated up-regulation of lysyl oxidase in the kidneys of hereditary nephrotic mouse with chronic renal fibrosis. Virchows Arch 447(5):859–868
Roy R, Polgar P, Wang Y, et al (1996) Regulation of lysyl oxidase and cyclooxygenase expression in human lung fibroblasts: interactions among TGF-beta, IL-1 beta, and prostaglandin E. J Cell Biochem 62(3):411–417
Yoshida M, Fujii K (1999) Differences in cellular properties and responses to growth factors between human ACL and MCL cells. J Orthop Sci 4(4):293–298
Barlow Y, Willoughby (1992) Pathophysiology of soft tissue repair. Brit Med Bull 48(3):698–711
Marshak DR, Lukas TJ, Watterson DM (1985) Drug–protein interactions: Binding of chlorpromazine to calmodulin, calmodulin fragments and related calcium binding proteins. Biochemistry 24(1):144–150
Abi Ezzi SS, Foulk RA, Harwood FL et al (1997) Decrease in fibronectin occurs coincident with the increased expression of its integrin receptor alpha5beta1 in stress-deprived ligaments. Iowa Orthop J 17:102–109
Arnoczky SP (1991) Physiological principles of ligament injuries and healing. In: Scott WN (ed) Ligament and extensor mechanism injuries of the knee: diagnosis and treatment. CV Mosby, St. Louis, pp 67–81
Lee J, Harwood FL, Akeson WH, Amiel D (1998) Growth factor expression in healing rabbit medial collateral and anterior cruciate ligaments. Iowa Orthop J 18:19–25
Okuizumi T, Tohyama H, Kondo E, Yasuda K (2004) The effect of cell-based therapy with autologous synovial fibroblasts activated by exogenous TGF-beta1 on the in situ frozen-thawed anterior cruciate ligament. J Orthop Sci 9(5):488–494
Falah M, Nierenberg G, Soudry M, et al (2010) Treatment of articular cartilage lesions of the knee. Int Orthop 34(5):621–630
Shanley CJ, Gharaee-Kermani M, Sarkar R, et al (1997) Transforming growth factor-beta 1 increases lysyl oxidase enzyme activity and mRNA in rat aortic smooth muscle cells. J Vasc Surg 25(3):446–452
Kim Y-M, Kim E-C, Kim Y (2011) The human lysyl oxidase-like 2 protein functions as an amine oxidase toward collagen and elastin. Mol Biol Rep 38(1):145–149
Yu Q, Horak K, Larson DF (2006) Role of T lymphocytes in hypertension-induced cardiac extracellular matrix remodeling. Hypertension 48(1):98–104
Kim DJ, Lee DC, Yang SJ, et al (2008) Lysyl oxidase like 4, a novel target gene of TGF-beta1 signaling, can negatively regulate TGF-beta1-induced cell motility in PLC/PRF/5 hepatoma cells. Biochem Biophys Res Commun 373(4):521–527
Acknowledgements
This study was supported by Project 111 (B06023, China), an OREF Award (USA) and by NIH AR45635 (USA).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xie, J., Jiang, J., Zhang, Y. et al. Up-regulation expressions of lysyl oxidase family in Anterior Cruciate Ligament and Medial Collateral Ligament fibroblasts induced by Transforming Growth Factor-Beta 1. International Orthopaedics (SICOT) 36, 207–213 (2012). https://doi.org/10.1007/s00264-011-1261-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-011-1261-3