Abstract
Purpose
Targeting of anti-programmed cell death protein-1 (PD-1) and anti-programmed death-ligand 1 (PD-L1) is a standard therapeutic strategy for various cancers. The aim of the present study was to investigate the prognostic effect of pretreatment PD-L1 expression levels in peripheral blood mononuclear cell (PBMC) subsets for patients with several cancer types receiving anti-PD-1 blockade therapies.
Patients and methods
Thirty-two patients undergoing anti-PD-L1 blockade therapy, including 15 with non-small cell lung cancer, 14 with gastric cancer, 1 with melanoma, 1 with parotid cancer, and 1 with bladder cancer, were recruited for the present study. PD-L1 expression levels in CD3+, CD4+, CD8+, CD45RA+ and CCR7+ T cells; CD20+ B cells; CD14+ and CD16+ monocytes were measured via flow cytometry before treatment. The percentages of PD-L1+ cells in respective PBMC subsets were compared with respect to different clinicopathological conditions and the association with overall survival (OS) was assessed.
Results
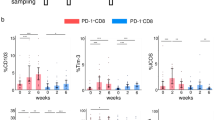
The percentages of PD-L1+ with CD3+, CD4+ and CD8+ T cells including naïve and memory T cell subsets, or CD20+ B cells during pretreatment were not markedly correlated with the OS of patients (p > 0.05); however, the percentage of the PD-L1+ CD14+ monocyte subset was significantly correlated with OS (p = 0.0426).
Conclusion
Increase in pretreatment expression levels of PD-L1 on CD14+ monocytes is associated with the OS of patients treated with immune checkpoint inhibitors. Further evaluation of large sample size and each specific cancer type might clarify the predictive role of PBMC in patients.





Similar content being viewed by others
References
Freeman GJ, Long AJ, Iwai Y, Bourque K, Chernova T, Nishimura H, Fitz LJ, Malenkovich N, Okazaki T, Byrne MC, Horton HF, Fouser L, Carter L, Ling V, Bowman MR, Carreno BM, Collins M, Wood CR, Honjo T (2000) Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med 192(7):1027–1034
Carreno BM, Collins M (2002) The B7 family of ligands and its receptors: new pathways for costimulation and inhibition of immune responses. Annu Rev Immunol 20:29–53. https://doi.org/10.1146/annurev.immunol.20.091101.091806
Barber DL, Wherry EJ, Masopust D, Zhu B, Allison JP, Sharpe AH, Freeman GJ, Ahmed R (2006) Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 439(7077):682–687. https://doi.org/10.1038/nature04444
Zou W, Chen L (2008) Inhibitory B7-family molecules in the tumour microenvironment. Nat Rev Immunol 8(6):467–477. https://doi.org/10.1038/nri2326
Zou W, Wolchok JD, Chen L (2016) PD-L1 (B7–H1) and PD-1 pathway blockade for cancer therapy: Mechanisms, response biomarkers, and combinations. Sci Transl Med 8(328):328. https://doi.org/10.1126/scitranslmed.aad7118
Hamanishi J, Mandai M, Matsumura N, Abiko K, Baba T, Konishi I (2016) PD-1/PD-L1 blockade in cancer treatment: perspectives and issues. Int J Clin Oncol 21(3):462–473. https://doi.org/10.1007/s10147-016-0959-z
Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF, Powderly JD, Carvajal RD, Sosman JA, Atkins MB, Leming PD, Spigel DR, Antonia SJ, Horn L, Drake CG, Pardoll DM, Chen L, Sharfman WH, Anders RA, Taube JM, McMiller TL, Xu H, Korman AJ, Jure-Kunkel M, Agrawal S, McDonald D, Kollia GD, Gupta A, Wigginton JM, Sznol M (2012) Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med 366(26):2443–2454. https://doi.org/10.1056/NEJMoa1200690
Ohigashi Y, Sho M, Yamada Y, Tsurui Y, Hamada K, Ikeda N, Mizuno T, Yoriki R, Kashizuka H, Yane K, Tsushima F, Otsuki N, Yagita H, Azuma M, Nakajima Y (2005) Clinical significance of programmed death-1 ligand-1 and programmed death-1 ligand-2 expression in human esophageal cancer. Clin Cancer Res 11(8):2947–2953. https://doi.org/10.1158/1078-0432.CCR-04-1469
Thompson RH, Kuntz SM, Leibovich BC, Dong H, Lohse CM, Webster WS, Sengupta S, Frank I, Parker AS, Zincke H, Blute ML, Sebo TJ, Cheville JC, Kwon ED (2006) Tumor B7–H1 is associated with poor prognosis in renal cell carcinoma patients with long-term follow-up. Can Res 66(7):3381–3385. https://doi.org/10.1158/0008-5472.CAN-05-4303
Wang Q, Liu F, Liu L (2017) Prognostic significance of PD-L1 in solid tumor: An updated meta-analysis. Medicine 96(18):e6369. https://doi.org/10.1097/MD.0000000000006369
Kulangara K, Zhang N, Corigliano E, Guerrero L, Waldroup S, Jaiswal D, Ms MJ, Shah S, Hanks D, Wang J, Lunceford J, Savage MJ, Juco J, Emancipator K (2019) Clinical utility of the combined positive score for programmed death ligand-1 expression and the approval of pembrolizumab for treatment of gastric cancer. Arch Pathol Lab Med 143(3):330–337. https://doi.org/10.5858/arpa.2018-0043-OA
Teng MW, Ngiow SF, Ribas A, Smyth MJ (2015) Classifying cancers based on T-cell Infiltration and PD-L1. Can Res 75(11):2139–2145. https://doi.org/10.1158/0008-5472.CAN-15-0255
Meng X, Huang Z, Teng F, Xing L, Yu J (2015) Predictive biomarkers in PD-1/PD-L1 checkpoint blockade immunotherapy. Cancer Treat Rev 41(10):868–876. https://doi.org/10.1016/j.ctrv.2015.11.001
Fuchs CS, Doi T, Jang RW, Muro K, Satoh T, Machado M, Sun W, Jalal SI, Shah MA, Metges JP, Garrido M, Golan T, Mandala M, Wainberg ZA, Catenacci DV, Ohtsu A, Shitara K, Geva R, Bleeker J, Ko AH, Ku G, Philip P, Enzinger PC, Bang YJ, Levitan D, Wang J, Rosales M, Dalal RP, Yoon HH (2018) Safety and efficacy of pembrolizumab monotherapy in patients with previously treated advanced gastric and gastroesophageal junction cancer: phase 2 clinical KEYNOTE-059 Trial. JAMA oncology 4(5):e180013. https://doi.org/10.1001/jamaoncol.2018.0013
Brown JM, Recht L, Strober S (2017) The promise of targeting macrophages in cancer therapy. Clin Cancer Res 23(13):3241–3250. https://doi.org/10.1158/1078-0432.CCR-16-3122
Tanaka A, Sakaguchi S (2019) Targeting Treg cells in cancer immunotherapy. Eur J Immunol 49(8):1140–1146. https://doi.org/10.1002/eji.201847659
Ando K, Hamada K, Watanabe M, Ohkuma R, Shida M, Onoue R, Kubota Y, Matsui H, Ishiguro T, Hirasawa Y, Ariizumi H, Tsurutani J, Yoshimura K, Tsunoda T, Kobayashi S, Wada S (2019) Plasma levels of soluble PD-L1 correlate with tumor regression in patients with lung and gastric cancer treated with immune checkpoint inhibitors. Anticancer Res 39(9):5195–5201. https://doi.org/10.21873/anticanres.13716
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, Kaplan R, Lacombe D, Verweij J (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 11). Eur J Cancer 45(2):228–247. https://doi.org/10.1016/j.ejca.2008.10.026
Seymour L, Bogaerts J, Perrone A, Ford R, Schwartz LH, Mandrekar S, Lin NU, Litiere S, Dancey J, Chen A, Hodi FS, Therasse P, Hoekstra OS, Shankar LK, Wolchok JD, Ballinger M, Caramella C, de Vries EGE, group Rw (2017) iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics. Lancet Oncol 18(3):e143–e152. https://doi.org/10.1016/S1470-2045(17)30074-8
Sallusto F, Geginat J, Lanzavecchia A (2004) Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu Rev Immunol 22:745–763. https://doi.org/10.1146/annurev.immunol.22.012703.104702
Sallusto F, Lenig D, Forster R, Lipp M, Lanzavecchia A (1999) Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401(6754):708–712. https://doi.org/10.1038/44385
Ziegler-Heitbrock L, Ancuta P, Crowe S, Dalod M, Grau V, Hart DN, Leenen PJ, Liu YJ, MacPherson G, Randolph GJ, Scherberich J, Schmitz J, Shortman K, Sozzani S, Strobl H, Zembala M, Austyn JM, Lutz MB (2010) Nomenclature of monocytes and dendritic cells in blood. Blood 116(16):e74–80. https://doi.org/10.1182/blood-2010-02-258558
Lee HW, Choi HJ, Ha SJ, Lee KT (1835) Kwon YG (2013) Recruitment of monocytes/macrophages in different tumor microenvironments. Biochem Biophys Acta 2:170–179. https://doi.org/10.1016/j.bbcan.2012.12.007
Farhood B, Najafi M, Mortezaee K (2019) CD8(+) cytotoxic T lymphocytes in cancer immunotherapy: A review. J Cell Physiol 234(6):8509–8521. https://doi.org/10.1002/jcp.27782
Kato T, Noma K, Ohara T, Kashima H, Katsura Y, Sato H, Komoto S, Katsube R, Ninomiya T, Tazawa H, Shirakawa Y, Fujiwara T (2018) Cancer-associated fibroblasts affect intratumoral CD8(+) and FoxP3(+) T cells Via IL6 in the tumor microenvironment. Clin Cancer Res 24(19):4820–4833. https://doi.org/10.1158/1078-0432.CCR-18-0205
Topalian SL, Taube JM, Anders RA, Pardoll DM (2016) Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat Rev Cancer 16(5):275–287. https://doi.org/10.1038/nrc.2016.36
Zarif JC, Hernandez JR, Verdone JE, Campbell SP, Drake CG, Pienta KJ (2016) A phased strategy to differentiate human CD14+monocytes into classically and alternatively activated macrophages and dendritic cells. Biotechniques 61(1):33–41. https://doi.org/10.2144/000114435
Qian BZ, Pollard JW (2010) Macrophage diversity enhances tumor progression and metastasis. Cell 141(1):39–51. https://doi.org/10.1016/j.cell.2010.03.014
Zhang QW, Liu L, Gong CY, Shi HS, Zeng YH, Wang XZ, Zhao YW, Wei YQ (2012) Prognostic significance of tumor-associated macrophages in solid tumor: a meta-analysis of the literature. PLoS ONE 7(12):e50946. https://doi.org/10.1371/journal.pone.0050946
Laviron M, Boissonnas A (2019) Ontogeny of tumor-associated macrophages. Front Immunol 10:1799. https://doi.org/10.3389/fimmu.2019.01799
Zhang Y, Zhu W, Zhang X, Qu Q, Zhang L (2017) Expression and clinical significance of programmed death-1 on lymphocytes and programmed death ligand-1 on monocytes in the peripheral blood of patients with cervical cancer. Oncol Lett 14(6):7225–7231. https://doi.org/10.3892/ol.2017.7105
Wang D, Quiros J, Mahuron K, Pai CC, Ranzani V, Young A, Silveria S, Harwin T, Abnousian A, Pagani M, Rosenblum MD, Van Gool F, Fong L, Bluestone JA, DuPage M (2018) Targeting EZH2 reprograms intratumoral regulatory T cells to enhance cancer immunity. Cell Rep 23(11):3262–3274. https://doi.org/10.1016/j.celrep.2018.05.050
Colombo MP, Piconese S (2007) Regulatory-T-cell inhibition versus depletion: the right choice in cancer immunotherapy. Nat Rev Cancer 7(11):880–887. https://doi.org/10.1038/nrc2250
Shang B, Liu Y, Jiang SJ, Liu Y (2015) Prognostic value of tumor-infiltrating FoxP3+ regulatory T cells in cancers: a systematic review and meta-analysis. Sci Rep 5:15179. https://doi.org/10.1038/srep15179
Burel JG, Pomaznoy M, Lindestam Arlehamn CS, Weiskopf D, da Silva AR, Jung Y, Babor M, Schulten V, Seumois G, Greenbaum JA, Premawansa S, Premawansa G, Wijewickrama A, Vidanagama D, Gunasena B, Tippalagama R, deSilva AD, Gilman RH, Saito M, Taplitz R, Ley K, Vijayanand P, Sette A, Peters B (2019) Circulating T cell-monocyte complexes are markers of immune perturbations. eLife. https://doi.org/10.7554/eLife.46045
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing.
Funding
No sponsor was involved.
Author information
Authors and Affiliations
Contributions
KH and SW conceived and designed this study. KH, RO, YK, AH, HM, TI, YH, HA, TA, and SW collected samples and recorded the general data and patient indications. MS, MW, and RO prepared the samples. MS and RO performed all flow cytometry analyses. KA analyzed the data and wrote the first draft of the manuscript. JT, KY, TT, SK, and SW critically reviewed and revised the manuscript. All Authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None of the Authors declares any conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The “Showa University Ethics Committee” approved the study with number 2165 and 2253.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ando, K., Hamada, K., Shida, M. et al. A high number of PD-L1+ CD14+ monocytes in peripheral blood is correlated with shorter survival in patients receiving immune checkpoint inhibitors. Cancer Immunol Immunother 70, 337–348 (2021). https://doi.org/10.1007/s00262-020-02686-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-020-02686-6