Abstract
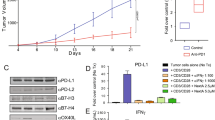
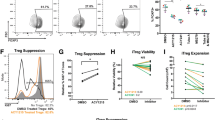
Histone deacetylase (HDAC) inhibitors impair tumor cell proliferation and alter gene expression. However, the impact of these changes on anti-tumor immunity is poorly understood. Here, we showed that the class I HDAC inhibitor, entinostat (ENT), promoted the expression of immune-modulatory molecules, including MHCII, costimulatory ligands, and chemokines on murine breast tumor cells in vitro and in vivo. ENT also impaired tumor growth in vivo—an effect that was dependent on both CD8+ T cells and IFNγ. Moreover, ENT promoted intratumoral T-cell clonal expansion and enhanced their functional activity. Importantly, ENT sensitized normally unresponsive tumors to the effects of PD1 blockade, predominantly through increases in T-cell proliferation. Our findings suggest that class I HDAC inhibitors impair tumor growth by enhancing the proliferative and functional capacity of CD8+ T cells and by sensitizing tumor cells to T-cell recognition.






Similar content being viewed by others
Abbreviations
- ATCC:
-
American-Type Culture Collection
- CIITA:
-
Class II transcriptional activator
- ENT:
-
Entinostat
- GZB:
-
Granzyme B
- HAT:
-
Histone acetyltransferase
- HDAC:
-
Histone deacetylase
- MHCII:
-
MHC class II
- PAN:
-
Panobinostat
- TIL:
-
Tumor-infiltrating lymphocyte
- TNBC:
-
Triple negative breast cancer
References
Siegel RL, Miller KD, Jemal A (2018) Cancer statistics, 2018. CA Cancer J Clin 68:7–30
Gu G, Dustin D, Fuqua SAW (2016) Targeted therapy for breast cancer and molecular mechanisms of resistance to treatment. Curr Opin Pharmacol 31:97–103
Forero A, Li Y, Chen D, Grizzle WE, Updike KL, Merz ND, Downs-Kelly E, Burwell TC, Vaklavas C, Buchsbaum DJ, Myers RM, LoBuglio AF, Varley KE (2016) Expression of the MHC class II pathway in triple-negative breast cancer tumor cells is associated with a good prognosis and infiltrating lymphocytes. Cancer Immunol Res 4:390–399
McCaw TR, Li M, Starenki D, Cooper SJ, Liu M, Meza-Perez S, Arend RC, Buchsbaum DJ, Forero A, Randall TD (2018) The expression of MHC class II molecules on murine breast tumors delays T cell exhaustion, expands the T cell repertoire, and slows tumor growth. Cancer Immunol Immunother 68:175–188
Meazza R, Comes A, Orengo AM, Ferrini S, Accolla RS (2003) Tumor rejection by gene transfer of the MHC class II transactivator in murine mammary adenocarcinoma cells. Eur J Immunol 33:1183–1192
Mortara L, Castellani P, Meazza R, Tosi G, De Lerma Barbaro A, Procopio FA, Comes A, Zardi L, Ferrini S, Accolla RS (2006) CIITA-induced MHC class II expression in mammary adenocarcinoma leads to a Th1 polarization of the tumor microenvironment, tumor rejection, and specific antitumor memory. Clin Cancer Res 12:3435–3443
Berdasco M, Esteller M (2010) Aberrant epigenetic landscape in cancer: how cellular identity goes awry. Dev Cell 19:698–711
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674
Fraga MF, Ballestar E, Villar-Garea A, Boix-Chornet M, Espada J, Schotta G, Bonaldi T, Haydon C, Ropero S, Petrie K, Iyer NG, Perez-Rosado A, Calvo E, Lopez JA, Cano A, Calasanz MJ, Colomer D, Piris MA, Ahn N, Imhof A, Caldas C, Jenuwein T, Esteller M (2005) Loss of acetylation at Lys16 and trimethylation at Lys20 of histone H4 is a common hallmark of human cancer. Nat Genet 37:391–400
Bowman GD, Poirier MG (2015) Post-translational modifications of histones that influence nucleosome dynamics. Chem Rev 115:2274–2295
de Ruijter AJM, van Gennip AH, Caron HN, Kemp S, van Kuilenburg ABP (2003) Histone deacetylases (HDACs): characterization of the classical HDAC family. Biochem J 370:737–749
Johnstone RW (2002) Histone-deacetylase inhibitors: novel drugs for the treatment of cancer. Nat Rev Drug Discov 1:287
Nakagawa M, Oda Y, Eguchi T, Aishima S, Yao T, Hosoi F, Basaki Y, Ono M, Kuwano M, Tanaka M, Tsuneyoshi M (2007) Expression profile of class I histone deacetylases in human cancer. Oncol Rep 18:769–774
Suzuki J, Chen YY, Scott GK, Devries S, Chin K, Benz CC, Waldman FM, Hwang ES (2009) Protein acetylation and histone deacetylase expression associated with malignant breast cancer progression. Clin Cancer Res 15:3163–3171
McCaw TR, Randall TD, Forero A, Buchsbaum DJ (2017) Modulation of antitumor immunity with histone deacetylase inhibitors. Immunotherapy 9:1359–1372
Munster PN, Troso-Sandoval T, Rosen N, Rifkind R, Marks PA, Richon VM (2001) The histone deacetylase inhibitor suberoylanilide hydroxamic acid induces differentiation of human breast cancer cells. Can Res 61:8492–8497
Vigushin DM, Ali S, Pace PE, Mirsaidi N, Ito K, Adcock I, Coombes RC (2001) Trichostatin A is a histone deacetylase inhibitor with potent antitumor activity against breast cancer in vivo. Clin Cancer Res 7:971–976
Khan AN, Magner WJ, Tomasi TB (2007) An epigenetic vaccine model active in the prevention and treatment of melanoma. J Transl Med 5:64
Woods DM, Woan K, Cheng F, Wang H, Perez-Villarroel P, Lee C, Lienlaf M, Atadja P, Seto E, Weber J, Sotomayor EM, Villagra A (2013) The antimelanoma activity of the histone deacetylase inhibitor panobinostat (LBH589) is mediated by direct tumor cytotoxicity and increased tumor immunogenicity. Melanoma Res 23:341–348
Woods DM, Sodre AL, Villagra A, Sarnaik A, Sotomayor EM, Weber J (2015) HDAC inhibition upregulates PD-1 ligands in melanoma and augments immunotherapy with PD-1 blockade. Cancer Immunol Res 3:1375–1385
Zheng H, Zhao W, Yan C, Watson CC, Massengill M, Xie M, Massengill C, Noyes DR, Martinez GV, Afzal R, Chen Z, Ren X, Antonia SJ, Haura EB, Ruffell B, Beg AA (2016) HDAC inhibitors enhance T Cell chemokine expression and augment response to PD-1 immunotherapy in lung adenocarcinoma. Clin Cancer Res 22:4119–4132
Kim K, Skora AD, Li Z, Liu Q, Tam AJ, Blosser RL, Diaz LA, Papadopoulos N, Kinzler KW, Vogelstein B, Zhou S (2014) Eradication of metastatic mouse cancers resistant to immune checkpoint blockade by suppression of myeloid-derived cells. Proc Natl Acad Sci 111:11774–11779
Orillion A, Hashimoto A, Damayanti N, Shen L, Adelaiye-Ogala R, Arisa S, Chintala S, Ordentlich P, Kao C, Elzey B, Gabrilovich D, Pili R (2017) Entinostat neutralizes myeloid-derived suppressor cells and enhances the antitumor effect of PD-1 inhibition in murine models of lung and renal cell carcinoma. Clin Cancer Res 23:5187–5201
Pallandre J-R, Borg C, Rognan D, Boibessot T, Luzet V, Yesylevskyy S, Ramseyer C, Pudlo M (2015) Novel aminotetrazole derivatives as selective STAT3 non-peptide inhibitors. Eur J Med Chem 103:163–174
Shen L, Pili R (2012) Class I histone deacetylase inhibition is a novel mechanism to target regulatory T cells in immunotherapy. Oncoimmunology 1:948–950
Terranova-Barberio M, Thomas S, Munster PN (2016) Epigenetic modifiers in immunotherapy: a focus on checkpoint inhibitors. Immunotherapy 8:705–719
Nazarov VI, Pogorelyy MV, Komech EA, Zvyagin IV, Bolotin DA, Shugay M, Chudakov DM, Lebedev YB, Mamedov IZ (2015) tcR: an R package for T cell receptor repertoire advanced data analysis. BMC Bioinform 16:175
Venturi V, Kedzierska K, Turner SJ, Doherty PC, Davenport MP (2007) Methods for comparing the diversity of samples of the T cell receptor repertoire. J Immunol Methods 321:182–195
Magner WJ, Kazim AL, Stewart C, Romano MA, Catalano G, Grande C, Keiser N, Santaniello F, Tomasi TB (2000) Activation of MHC class I, II, and CD40 gene expression by histone deacetylase inhibitors. J Immunol 165:7017–7024
Zika E, Greer SF, Zhu XS, Ting JPY (2003) Histone deacetylase 1/mSin3A disrupts gamma interferon-induced CIITA function and major histocompatibility complex class II enhanceosome formation. Mol Cell Biol 23:3091–3102
Wright KL, Ting JP (2006) Epigenetic regulation of MHC-II and CIITA genes. Trends Immunol 27:405–412
West AC, Mattarollo SR, Shortt J, Cluse LA, Christiansen AJ, Smyth MJ, Johnstone RW (2013) An intact immune system is required for the anticancer activities of histone deacetylase inhibitors. Can Res 73:7265–7276
Ayers M, Lunceford J, Nebozhyn M, Murphy E, Loboda A, Kaufman DR, Albright A, Cheng JD, Kang SP, Shankaran V, Piha-Paul SA, Yearley J, Seiwert TY, Ribas A, McClanahan TK (2017) IFN-γ–related mRNA profile predicts clinical response to PD-1 blockade. J Clin Investig 127:2930–2940
Callahan MJ, Nagymanyoki Z, Bonome T, Johnson ME, Litkouhi B, Sullivan EH, Hirsch MS, Matulonis UA, Liu J, Birrer MJ, Berkowitz RS, Mok SC (2008) Increased HLA-DMB expression in the tumor epithelium is associated with increased CTL infiltration and improved prognosis in advanced-stage serous ovarian cancer. Clin Cancer Res 14:7667–7673
Johnson DB, Estrada MV, Salgado R, Sanchez V, Doxie DB, Opalenik SR, Vilgelm AE, Feld E, Johnson AS, Greenplate AR, Sanders ME, Lovly CM, Frederick DT, Kelley MC, Richmond A, Irish JM, Shyr Y, Sullivan RJ, Puzanov I, Sosman JA, Balko JM (2016) Melanoma-specific MHC-II expression represents a tumour-autonomous phenotype and predicts response to anti-PD-1/PD-L1 therapy. Nat Commun 7:10582
Kohm AP, McMahon JS, Podojil JR, Begolka WS, DeGutes M, Kasprowicz DJ, Ziegler SF, Miller SD (2006) Cutting edge: anti-CD25 monoclonal antibody injection results in the functional inactivation, not depletion, of CD4 + CD25 + T regulatory cells. J Immunol 176:3301–3305
Brogdon JL, Xu Y, Szabo SJ, An S, Buxton F, Cohen D, Huang Q (2007) Histone deacetylase activities are required for innate immune cell control of Th1 but not Th2 effector cell function. Blood 109:1123–1130
Long J, Chang L, Shen Y, Gao WH, Wu YN, Dou HB, Huang MM, Wang Y, Fang WY, Shan JH, Wang YY, Zhu J, Chen Z, Hu J (2015) Valproic acid ameliorates graft-versus-host disease by downregulating Th1 and Th17 Cells. J Immunol 195:1849–1857
Moreira JMA, Scheipers P, Sørensen P (2003) The histone deacetylase inhibitor trichostatin A modulates CD4 + T cell responses. BMC Cancer 3:1–18
Skov S, Rieneck K, Bovin LF, Skak K, Tomra S, Michelsen BK, Ødum N (2003) Histone deacetylase inhibitors: a new class of immunosuppressors targeting a novel signal pathway essential for CD154 expression. Immunobiology 101:1430–1438
Cao K, Wang G, Li W, Zhang L, Wang R, Huang Y, Du L, Jiang J, Wu C, He X, Roberts AI, Li F, Rabson AB, Wang Y, Shi Y (2015) Histone deacetylase inhibitors prevent activation-induced cell death and promote anti-tumor immunity. Oncogene 34:5960–5970
Philip M, Fairchild L, Sun L, Horste EL, Camara S, Shakiba M, Scott AC, Viale A, Lauer P, Merghoub T, Hellmann MD, Wolchok JD, Leslie CS, Schietinger A (2017) Chromatin states define tumour-specific T cell dysfunction and reprogramming. Nature 545:452
Taube JM, Anders RA, Young GD, Xu H, Sharma R, McMiller TL, Chen S, Klein AP, Pardoll DM, Topalian SL, Chen L (2012) Colocalization of inflammatory response with B7-H1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Sci Transl Med 4:127
Acknowledgements
The authors would like to thank Uma Mudunuru and Scott Simpler for animal husbandry, and Eddy Yang and Debbie Della Manna of the NanoString Laboratory and Sagar Hanamanthu of the Comprehensive Flow Cytometry Core for lending respective expertise.
Funding
This work was supported by the University of Alabama at Birmingham Comprehensive Cancer Center (P30 CA013148), National Institutes of Health grant CA216234, and by the Breast Cancer Research Foundation of Alabama.
Author information
Authors and Affiliations
Contributions
TRM designed, performed, and interpreted experiments and wrote the manuscript. ML designed, performed, and interpreted experiments. DS performed TCR repertoire sequencing and analysis. SJC performed TCR repertoire sequencing and analysis. ML designed, performed, and interpreted experiments. RCA designed and interpreted experiments and edited the manuscript. AF designed and interpreted experiments and edited the manuscript. DJB designed and interpreted experiments and edited the manuscript. TDR designed and interpreted experiments and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human participants and/or animals
All procedures involving animals were performed in accordance with the guidelines of the National Research Council (United States) Committee for the Update of the Guide for the Care and Use of Laboratory Animals and were approved by the University of Alabama at Birmingham Institutional Animal Care and Use Committee (IACUC) in protocol 09854.
Animal source
BALB/c mice were purchased from Charles River Laboratories International, Inc. BALB/c.scid mice (CBySmn.CB17-PrkdcscidIJ) were purchased from The Jackson Laboratory.
Cell line authentication
TS/A murine mammary adenocarcinoma cells were provided by Dr. Roberto S. Accolla, Department of Clinical and Biological Sciences, University of Insubria, Italy. TS/A cells were obtained at passage 22 and passaged two times prior to freezing archival samples. 4T1 cells were purchased from the American Type Culture Collection (ATCC). Samples of 4T1 and TS/A cells were authenticated by IDEXX BioAnalytics: case #23272-2019. All cell lines tested negative for mycoplasma (and 13 other mouse pathogens) via PCR performed by Charles River Research Animal Diagnostic Services.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
McCaw, T.R., Li, M., Starenki, D. et al. Histone deacetylase inhibition promotes intratumoral CD8+ T-cell responses, sensitizing murine breast tumors to anti-PD1. Cancer Immunol Immunother 68, 2081–2094 (2019). https://doi.org/10.1007/s00262-019-02430-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-019-02430-9