Abstract
Background
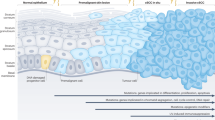
T-lymphocytes are involved in tumor progression and regression. Actinic keratoses (AK) are atypical proliferations of keratinocytes of the skin. Some AK progress into invasive cutaneous squamous cell carcinomas (cSCC). Keratoacanthomas (KA) are either classified as a cSCC subtype or a benign tumor with histologic resemblance to well-differentiated cSCC as it is supposed to regress spontaneously. In contrast, cSCC represent malignant tumors that may metastasize.
Objectives
To compare the T-lymphocyte profiles of AK, KA and cSCC in relation to PD-L1 expression.
Methods
Tissue micro-arrays of 103 cases of AK, 43 cases of KA and 106 cases of cSCC were stained by immunohistochemistry for E-cadherin, CD3, CD4, CD8, FOXp3, and the receptor–ligand pair PD-1/PD-L1. Immunohistological scores were computationally determined to assess PD-L1 expression as well as the expression profiles of T-lymphocytes.
Results
AK had lower numbers of CD3+ and PD-1+ cells compared to KA and lower numbers of CD3+, CD8+ and PD-1+ cells in comparison with cSCC. KA showed significantly higher numbers of CD4+ and FOXp3+ cells as well as lower numbers of CD8+ cells in comparison with invasive cSCC. cSCC expressed significantly more PD-L1 in comparison with AK and KA. Among cSCC PD-L1 expression was higher in moderately and poorly-differentiated cSCC than in well-differentiated cSCC. Increased PD-L1 expression also correlated with increased numbers of CD4+, CD8+ and FOXp3+ cells in cSCC.
Conclusions
Tumor-associated T-lymphocyte infiltrates showed significant differences between AK, KA and invasive cSCC. PD-L1 expression correlated with invasion of T-cell infiltrates in invasive cSCC.





Similar content being viewed by others
Abbreviations
- AK:
-
Actinic keratosis
- cSCC:
-
Cutaneous squamous cell carcinoma
- KA:
-
Keratoacanthoma
- NCT:
-
National Center of Tumor Diseases
- OTR:
-
Organ transplant recipients
- ROI:
-
Region of interest
- TGF-β:
-
Transforming growth factor beta
- TMA:
-
Tissue micro-array
References
Schmults CD, Karia PS, Carter JB, Han J, Qureshi AA (2013) Factors predictive of recurrence and death from cutaneous squamous cell carcinoma: a 10-year, single-institution cohort study. JAMA Dermatol 149:541–547. https://doi.org/10.1001/jamadermatol.2013.2139
Karia PS, Han J, Schmults CD (2013) Cutaneous squamous cell carcinoma: estimated incidence of disease, nodal metastasis, and deaths from disease in the United States, 2012. J Am Acad Dermatol 68:957–966. https://doi.org/10.1016/j.jaad.2012.11.037
Lamberg AL, Solvsten H, Lei U et al (2016) The Danish nonmelanoma skin cancer dermatology database. Clin Epidemiol 8:633–636. https://doi.org/10.2147/CLEP.S99464
Rogers HW, Weinstock MA, Feldman SR, Coldiron BM (2015) Incidence estimate of nonmelanoma skin cancer (Keratinocyte Carcinomas) in the US population, 2012. JAMA Dermatol 151:1081–1086. https://doi.org/10.1001/jamadermatol.2015.1187
Lobeck A, Weiss C, Orouji A et al. (2017) Single center analysis of the dermatosurgical patient cohort of a tumor center in Germany. Hautarzt 68:377–384. https://doi.org/10.1007/s00105-017-3951-2
Thompson AK, Kelley BF, Prokop LJ, Murad MH, Baum CL (2016) Risk factors for cutaneous squamous cell carcinoma recurrence, metastasis, and disease-specific death: a systematic review and meta-analysis. JAMA Dermatol 152:419–428. https://doi.org/10.1001/jamadermatol.2015.4994
Burnworth B, Popp S, Stark HJ, Steinkraus V, Brocker EB, Hartschuh W, Birek C, Boukamp P (2006) Gain of 11q/cyclin D1 overexpression is an essential early step in skin cancer development and causes abnormal tissue organization and differentiation. Oncogene 25:4399–4412. https://doi.org/10.1038/sj.onc.1209474
Gleich T, Chiticariu E, Huber M, Hohl D (2016) Keratoacanthoma: a distinct entity? Exp Dermatol 25:85–91. https://doi.org/10.1111/exd.12880
Kwiek B, Schwartz RA (2016) Keratoacanthoma (KA): an update and review. J Am Acad Dermatol 74:1220–1233. https://doi.org/10.1016/j.jaad.2015.11.033
Selmer J, Skov T, Spelman L, Weedon D (2016) Squamous cell carcinoma and keratoacanthomas are biologically distinct and can be diagnosed by light microscopy: a review. Histopathology 69:535–541. https://doi.org/10.1111/his.13018
Stockfleth E (2017) The importance of treating the field in actinic keratosis. J Eur Acad Dermatol Venereol 31(Suppl 2):8–11. https://doi.org/10.1111/jdv.14092
Fernandez Figueras MT (2017) From actinic keratosis to squamous cell carcinoma: pathophysiology revisited. J Eur Acad Dermatol Venereol 31(Suppl 2):5–7. https://doi.org/10.1111/jdv.14151
Cassarino DS, Derienzo DP, Barr RJ (2006) Cutaneous squamous cell carcinoma: a comprehensive clinicopathologic classification—part two. J Cutan Pathol 33:261–279. https://doi.org/10.1111/j.0303-6987.2006.00516.x
Ra SH, Li X, Binder S (2011) Molecular discrimination of cutaneous squamous cell carcinoma from actinic keratosis and normal skin. Mod Pathol 24:963–973. https://doi.org/10.1038/modpathol.2011.39
Martincorena I, Roshan A, Gerstung M et al (2015) Tumor evolution. High burden and pervasive positive selection of somatic mutations in normal human skin. Science 348:880–886. https://doi.org/10.1126/science.aaa6806
Siegel JA, Korgavkar K, Weinstock MA (2017) Current perspective on actinic keratosis: a review. Br J Dermatol 177:350–358. https://doi.org/10.1111/bjd.14852
Vesely MD, Kershaw MH, Schreiber RD, Smyth MJ (2011) Natural innate and adaptive immunity to cancer. Annu Rev Immunol 29:235–271. https://doi.org/10.1146/annurev-immunol-031210-101324
Yanofsky VR, Mitsui H, Felsen D, Carucci JA (2013) Understanding dendritic cells and their role in cutaneous carcinoma and cancer immunotherapy. Clin Dev Immunol. 2013:624123. https://doi.org/10.1155/2013/624123
Gajewski TF, Woo SR, Zha Y, Spaapen R, Zheng Y, Corrales L, Spranger S (2013) Cancer immunotherapy strategies based on overcoming barriers within the tumor microenvironment. Curr Opin Immunol 25:268–276. https://doi.org/10.1016/j.coi.2013.02.009
Szturz P, Vermorken JB (2017) Immunotherapy in head and neck cancer: aiming at EXTREME precision. BMC Med 15:110. https://doi.org/10.1186/s12916-017-0879-4
Bluth MJ, Zaba LC, Moussai D et al (2009) Myeloid dendritic cells from human cutaneous squamous cell carcinoma are poor stimulators of T-cell proliferation. J Invest Dermatol 129:2451–2462. https://doi.org/10.1038/jid.2009.96
Kambayashi Y, Fujimura T, Aiba S (2013) Comparison of immunosuppressive and immunomodulatory cells in keratoacanthoma and cutaneous squamous cell carcinoma. Acta Derm Venereol 93:663–668. https://doi.org/10.2340/00015555-1597
Lai C, August S, Behar R, Polak M, Ardern-Jones M, Theaker J, Al-Shamkhani A, Healy E (2015) Characteristics of immunosuppressive regulatory T cells in cutaneous squamous cell carcinomas and role in metastasis. Lancet 385 (Suppl 1):S59. https://doi.org/10.1016/S0140-6736(15)60374-9
Mishra AK, Kadoishi T, Wang X, Driver E, Chen Z, Wang XJ, Wang JH (2016) Squamous cell carcinomas escape immune surveillance via inducing chronic activation and exhaustion of CD8+ T Cells co-expressing PD-1 and LAG-3 inhibitory receptors. Oncotarget 7:81341–81356. https://doi.org/10.18632/oncotarget.13228
Gambichler T, Gnielka M, Ruddel I, Stockfleth E, Stucker M, Schmitz L (2017) Expression of PD-L1 in keratoacanthoma and different stages of progression in cutaneous squamous cell carcinoma. Cancer Immunol Immunother 66:1199–1204. https://doi.org/10.1007/s00262-017-2015-x
Ayers M, Lunceford J, Nebozhyn M et al (2017) IFN-gamma-related mRNA profile predicts clinical response to PD-1 blockade. J Clin Invest 127:2930–2940. https://doi.org/10.1172/JCI91190
Jiao Q, Liu C, Li W, Li W, Fang F, Qian Q, Zhang X (2017) Programmed death-1 ligands 1 and 2 expression in cutaneous squamous cell carcinoma and their relationship with tumour-infiltrating dendritic cells. Clin Exp Immunol 188:420–429. https://doi.org/10.1111/cei.12921
Stevenson ML, Wang CQ, Abikhair M, Roudiani N, Felsen D, Krueger JG, Pavlick AC, Carucci JA (2017) Expression of programmed cell death ligand in cutaneous squamous cell carcinoma and treatment of locally advanced disease with pembrolizumab. JAMA Dermatol 153:299–303. https://doi.org/10.1001/jamadermatol.2016.5118
Linedale R, Schmidt C, King BT, Ganko AG, Simpson F, Panizza BJ, Leggatt GR (2017) Elevated frequencies of CD8 T cells expressing PD-1, CTLA-4 and Tim-3 within tumour from perineural squamous cell carcinoma patients. PLoS One 12:e0175755. https://doi.org/10.1371/journal.pone.0175755
Mittal A, Colegio OR (2017) Skin cancers in organ transplant recipients. Am J Transplant 17:2509–2530. https://doi.org/10.1111/ajt.14382
Rosen RH, Gupta AK, Tyring SK (2012) Dual mechanism of action of ingenol mebutate gel for topical treatment of actinic keratoses: rapid lesion necrosis followed by lesion-specific immune response. J Am Acad Dermatol 66:486–493. https://doi.org/10.1016/j.jaad.2010.12.038
Walter A, Schafer M, Cecconi V et al (2013) Aldara activates TLR7-independent immune defence. Nat Commun 4:1560. https://doi.org/10.1038/ncomms2566
Speiser DE, Ho PC, Verdeil G (2016) Regulatory circuits of T cell function in cancer. Nat Rev Immunol 16:599–611. https://doi.org/10.1038/nri.2016.80
Spranger S, Spaapen RM, Zha Y, Williams J, Meng Y, Ha TT, Gajewski TF (2013) Up-regulation of PD-L1, IDO, and T(regs) in the melanoma tumor microenvironment is driven by CD8(+) T cells. Sci Transl Med 5:200ra116. https://doi.org/10.1126/scitranslmed.3006504
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12:252–264. https://doi.org/10.1038/nrc3239
Gajewski TF, Schreiber H, Fu YX (2013) Innate and adaptive immune cells in the tumor microenvironment. Nat Immunol 14:1014–1022. https://doi.org/10.1038/ni.2703
Okazaki T, Chikuma S, Iwai Y, Fagarasan S, Honjo T (2013) A rheostat for immune responses: the unique properties of PD-1 and their advantages for clinical application. Nat Immunol 14:1212–1218. https://doi.org/10.1038/ni.2762
Boussiotis VA, Chatterjee P, Li L (2014) Biochemical signaling of PD-1 on T cells and its functional implications. Cancer J 20:265–271. https://doi.org/10.1097/PPO.0000000000000059
Weedon D (2009) Skin tumors. Weedon’s skin pathology. 3rd edn. London: Churchill Livingstone, pp 702–708. ISBN 9780702047749
Felcht M, Heck M, Weiss C et al. (2012) Expression of the T-cell regulatory marker FOXP3 in primary cutaneous large B-cell lymphoma tumour cells. Br J Dermatol 167:348–358. https://doi.org/10.1111/j.1365-2133.2012.10987.x
Slater NA, Googe PB (2016) PD-L1 expression in cutaneous squamous cell carcinoma correlates with risk of metastasis. J Cutan Pathol 43:663–670. https://doi.org/10.1111/cup.12728
Garcia-Pedrero JM, Martinez-Camblor P, Diaz-Coto S, Munguia-Calzada P, Vallina-Alvarez A, Vazquez-Lopez F, Rodrigo JP, Santos-Juanes J (2017) Tumor programmed cell death ligand 1 expression correlates with nodal metastasis in patients with cutaneous squamous cell carcinoma of the head and neck. J Am Acad Dermatol 77:527–533. https://doi.org/10.1016/j.jaad.2017.05.047
Roper E, Lum T, Palme CE, Ashford B, Ch’ng S, Ranson M, Boyer M, Clark J, Gupta R (2017) PD-L1 expression predicts longer disease free survival in high risk head and neck cutaneous squamous cell carcinoma. Pathology 49:499–505. https://doi.org/10.1016/j.pathol.2017.04.004
Awad MM, Jones RE, Liu H et al (2016) Cytotoxic T cells in PD-L1-positive malignant pleural mesotheliomas are counterbalanced by distinct immunosuppressive factors. Cancer Immunol Res 4:1038–1048. https://doi.org/10.1158/2326-6066.CIR-16-0171
Li Z, Dong P, Ren M, Song Y, Qian X, Yang Y, Li S, Zhang X, Liu F (2016) PD-L1 expression is associated with tumor FOXP3(+) regulatory T-cell infiltration of breast cancer and poor prognosis of patient. J Cancer 7:784–793. https://doi.org/10.7150/jca.14549
Thompson ED, Taube JM, Asch-Kendrick RJ et al (2017) PD-L1 expression and the immune microenvironment in primary invasive lobular carcinomas of the breast. Mod Pathol 30:1551–1560. https://doi.org/10.1038/modpathol.2017.79
Stasikowska-Kanicka O, Wagrowska-Danilewicz M, Danilewicz M (2017) Immunohistochemical analysis of Foxp3+, CD4+, CD8 + cell infiltrates and PD-L1 in oral squamous cell carcinoma. Pathol Oncol Res doi. https://doi.org/10.1007/s12253-017-0270-y
Cunningham TJ, Tabacchi M, Eliane JP et al (2017) Randomized trial of calcipotriol combined with 5-fluorouracil for skin cancer precursor immunotherapy. J Clin Invest 127:106–116. https://doi.org/10.1172/JCI89820
Lai C, August S, Albibas A et al (2016) OX40 + regulatory T cells in cutaneous squamous cell carcinoma suppress effector T-cell responses and associate with metastatic potential. Clin Cancer Res 22:4236–4248. https://doi.org/10.1158/1078-0432.CCR-15-2614
Crespo E, Fernandez L, Lucia M et al (2017) Effector antitumor and regulatory T cell responses influence the development of nonmelanoma skin cancer in kidney transplant patients. Transplantation 101:2102–2110. https://doi.org/10.1097/TP.0000000000001759
Tanaka A, Sakaguchi S (2017) Regulatory T cells in cancer immunotherapy. Cell Res 27:109–118. https://doi.org/10.1038/cr.2016.151
Azzimonti B, Zavattaro E, Provasi M, Vidali M, Conca A, Catalano E, Rimondini L, Colombo E, Valente G (2015) Intense Foxp3+ CD25+ regulatory T-cell infiltration is associated with high-grade cutaneous squamous cell carcinoma and counterbalanced by CD8+/Foxp3+ CD25+ ratio. Br J Dermatol 172:64–73. https://doi.org/10.1111/bjd.13172
Baas M, Besancon A, Goncalves T et al (2016) TGFbeta-dependent expression of PD-1 and PD-L1 controls CD8(+) T cell anergy in transplant tolerance. Elife 5:e08133. https://doi.org/10.7554/eLife.08133
Goudie DR, D’Alessandro M, Merriman B et al (2011) Multiple self-healing squamous epithelioma is caused by a disease-specific spectrum of mutations in TGFBR1. Nat Genet 43:365–369. https://doi.org/10.1038/ng.780
Lacouture ME, Morris JC, Lawrence DP et al (2015) Cutaneous keratoacanthomas/squamous cell carcinomas associated with neutralization of transforming growth factor beta by the monoclonal antibody fresolimumab (GC1008). Cancer Immunol Immunother 64:437–446. https://doi.org/10.1007/s00262-015-1653-0
Acknowledgements
We like to thank Dr. Damir Krunic from the Imaging Core Facility of the German Cancer Research Center (Deutsches Krebsforschungszentrum, DKFZ, Germany) as well as Helmut Hübers for technical support with image analysis. This research was supported by the tissue bank of the NCT (Heidelberg, Germany) by staining the skin tumor samples.
Funding
This work was supported by Grants from the Deutsche Forschungsgemeinschaft (German Research Council) [Förderprojekt Nr. FE 1282/2-1 (Moritz Felcht) and GRK2099/RTG2099 “Hallmarks of Skin Cancer” (to Viktor Umansky, Jochen Utikal, Hellmut G. Augustin, Sergij Goerdt, Cyrill Géraud and Moritz Felcht)] and partially supported by the Federal Ministry for Research and Education (Bundesmininsterium für Bildung und Forschung, BMBF) (FKZ 02NUK036A to Petra Boukamp).
Author information
Authors and Affiliations
Contributions
Study conception and design were done by Corinne Bauer, Ashik Ahmed Abdul Pari, Viktor Umansky, Petra Boukamp, Cyrill Géraud and Moritz Felcht. Corinne Bauer, Ashik Ahmed Abdul Pari, Cyrill Géraud and Moritz Felcht are responsible for the integrity of acquired data. Statistical analysis was performed by Corinne Bauer, Ashik Ahmed Abdul Pari, Cyrill Géraud and Moritz Felcht. Corinne Bauer, Ashik Ahmed Abdul Pari, Viktor Umansky, Petra Boukamp, Cyrill Géraud, Moritz Felcht prepared the initial manuscript. Corinne Bauer, Ashik Ahmed Abdul Pari, Viktor Umansky, Jochen Utikal, Petra Boukamp, Hellmut G. Augustin, Sergij Goerdt, Cyrill Géraud and Moritz Felcht made substantial contributions to data analysis and interpretation of results, rewriting of the manuscript, review and approval.
Corresponding author
Ethics declarations
Conflict of interest
Moritz Felcht received travel and congress participation funding by TEVA company, honoraria as an advisory board member of AbbVie Ltd. and lecture fees by Periderm GmbH and Mibe Vertrieb GmbH. All other authors declare no conflict of interest.
Ethical standards
The study was performed with archived paraffin-embedded tissue samples. The study was approved by the ethical committee II of Heidelberg University (2014-835R-MA).
Informed consent
Informed consent by individual patients cannot be given, as the study only included paraffin-embedded archived tissue. With the approval of the ethical committee informed consent was not required as all patient data were anonymized.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bauer, C., Abdul Pari, A.A., Umansky, V. et al. T-lymphocyte profiles differ between keratoacanthomas and invasive squamous cell carcinomas of the human skin. Cancer Immunol Immunother 67, 1147–1157 (2018). https://doi.org/10.1007/s00262-018-2171-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-018-2171-7