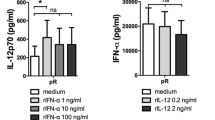
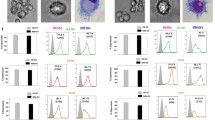
Abstract
Therapeutic tumor vaccination based on dendritic cells (DC) is safe; however, its efficacy is low. Among the reasons for only a subset of patients benefitting from DC-based immunotherapy is an insufficient potency of in vitro generated classical DCs (cDCs), made by treating monocytes with GM-CSF + IL-4 + maturation factors. Recent studies demonstrated that CD137L (4-1BBL, TNFSF9) signaling differentiates human monocytes to a highly potent novel type of DC (CD137L-DCs) which have an inflammatory phenotype and are closely related to in vivo DCs. Here, we show that CD137L-DCs induce potent CD8+ T-cell responses against Epstein–Barr virus (EBV) and Hepatitis B virus (HBV), and that T cells primed by CD137L-DCs more effectively lyse EBV+ and HBV+ target cells. The chemokine profile of CD137L-DCs identifies them as inflammatory DCs, and they polarize CD8+ T cells to a Tc1 phenotype. Expression of exhaustion markers is reduced on T cells activated by CD137L-DCs. Furthermore, these T cells are metabolically more active and have a higher capacity to utilize glucose. CD137L-induced monocyte to DC differentiation leads to the formation of AIM2 inflammasome, with IL-1beta contributing to CD137L-DCs possessing a stronger T cell activation ability. CD137L-DCs are effective in crosspresentation. PGE2 as a maturation factor is required for enhancing migration of CD137L-DCs but does not significantly reduce their potency. This study shows that CD137L-DCs have a superior ability to activate T cells and to induce potent Tc1 responses against the cancer-causing viruses EBV and HBV which suggest CD137L-DCs as promising candidates for DC-based tumor immunotherapy.





Similar content being viewed by others
Abbreviations
- CD137L:
-
CD137 ligand
- CD137L-DC:
-
CD137 ligand-generated dendritic cell
- cDC:
-
Classical DC
- DC:
-
Dendritic cell
- EBNA:
-
Epstein–Barr virus nuclear antigen
- EBV:
-
Epstein–Barr virus
- ECAR:
-
Extracellular acidification rate
- HBsAG:
-
HBV surface antigen
- HBV:
-
Hepatitis B virus
- LMP-1:
-
Latent membrane protein
- NUS:
-
National University of Singapore
- OCR:
-
Oxygen consumption rate
- PGE2:
-
Prostaglandin E2
References
Mellman I, Steinman RM (2001) Dendritic cells: specialized and regulated antigen processing machines. Cell 106:255–258
Palucka K, Banchereau J (2013) Dendritic-cell-based therapeutic cancer vaccines. Immunity 39:38–48. https://doi.org/10.1016/j.immuni.2013.07.004
Bol KF, Schreibelt G, Gerritsen WR, de Vries IJ, Figdor CG (2016) Dendritic Cell-based immunotherapy: state of the art and beyond. Clin Cancer Res 22:1897–1906. https://doi.org/10.1158/1078-0432.CCR-15-1399
Anguille S, Smits EL, Lion E, van Tendeloo VF, Berneman ZN (2014) Clinical use of dendritic cells for cancer therapy. Lancet Oncol 15:e257–267. https://doi.org/10.1016/S1470-2045(13)70585-0
Louis CU, Straathof K, Bollard CM, Ennamuri S, Gerken C, Lopez TT, Huls MH, Sheehan A, Wu MF, Liu H, Gee A, Brenner MK, Rooney CM et al (2010) Adoptive transfer of EBV-specific T cells results in sustained clinical responses in patients with locoregional nasopharyngeal carcinoma. J Immunother 33:983–990. https://doi.org/10.1097/CJI.0b013e3181f3cbf4
Smith C, Tsang J, Beagley L, Chua D, Lee V, Li V, Moss DJ, Coman W, Chan KH, Nicholls J, Kwong D, Khanna R (2012) Effective treatment of metastatic forms of Epstein-Barr virus-associated nasopharyngeal carcinoma with a novel adenovirus-based adoptive immunotherapy. Cancer Res 72:1116–1125. https://doi.org/10.1158/0008-5472.can-11-3399
Lin CL, Lo WF, Lee TH, Ren Y, Hwang SL, Cheng YF, Chen CL, Chang YS, Lee SP, Rickinson AB, Tam PK (2002) Immunization with Epstein-Barr Virus (EBV) peptide-pulsed dendritic cells induces functional CD8+ T-cell immunity and may lead to tumor regression in patients with EBV-positive nasopharyngeal carcinoma. Cancer Res 62:6952–6958
Qasim W, Brunetto M, Gehring AJ, Xue SA, Schurich A, Khakpoor A, Zhan H, Ciccorossi P, Gilmour K, Cavallone D, Moriconi F, Farzhenah F, Mazzoni A et al (2014) Immunotherapy of HCC metastases with autologous T cell receptor redirected T cells, targeting HBsAg in a liver transplant patient. J Hepatol 62:486–491. https://doi.org/10.1016/j.jhep.2014.10.001
Kwajah MMS, Schwarz H (2010) CD137 ligand signaling induces human monocyte to dendritic cell differentiation. Eur J Immunol 40:1938–1949. https://doi.org/10.1002/eji.200940105
Harfuddin Z, Kwajah S, Chong Nyi Sim A, Macary PA, Schwarz H (2013) CD137L-stimulated dendritic cells are more potent than conventional dendritic cells at eliciting cytotoxic T-cell responses. Oncoimmunology 2:e26859. https://doi.org/10.4161/onci.26859
Harfuddin Z, Dharmadhikari B, Wong SC, Duan K, Poidinger M, Kwajah S, Schwarz H (2016) Transcriptional and functional characterization of CD137L-dendritic cells identifies a novel dendritic cell phenotype. Sci Rep 6:29712. https://doi.org/10.1038/srep29712
Shao Z, Schwarz H (2011) CD137 ligand, a member of the tumor necrosis factor family, regulates immune responses via reverse signal transduction. J Leukoc Biol 89:21–29. https://doi.org/10.1189/jlb.0510315
Drenkard D, Becke FM, Langstein J, Spruss T, Kunz-Schughart LA, Tan TE, Lim YC, Schwarz H (2007) CD137 is expressed on blood vessel walls at sites of inflammation and enhances monocyte migratory activity. FASEB J 21:456–463. https://doi.org/10.1096/fj.05-4739com
Broll K, Richter G, Pauly S, Hofstaedter F, Schwarz H (2001) CD137 expression in tumor vessel walls. High correlation with malignant tumors. Am J Clin Pathol 115:543–549. https://doi.org/10.1309/6U88-357U-UKJ5-YPT3
Quek BZ, Lim YC, Lin JH, Tan TE, Chan J, Biswas A, Schwarz H (2010) CD137 enhances monocyte-ICAM-1 interactions in an E-selectin-dependent manner under flow conditions. Mol Immunol 47:1839–1847. https://doi.org/10.1016/j.molimm.2009.11.010
Jiang D, Chen Y, Schwarz H (2008) CD137 induces proliferation of murine hematopoietic progenitor cells and differentiation to macrophages. J Immunol 181:3923–3932
Jiang D, Yue PS, Drenkard D, Schwarz H (2008) Induction of proliferation and monocytic differentiation of human CD34 + cells by CD137 ligand signaling. Stem Cells 26:2372–2381. https://doi.org/10.1634/stemcells.2008-0158
Jiang D, Schwarz H (2010) Regulation of granulocyte and macrophage populations of murine bone marrow cells by G-CSF and CD137 protein. PLoS One 5:e15565. https://doi.org/10.1371/journal.pone.0015565
Chauvin JM, Larrieu P, Sarrabayrouse G, Prevost-Blondel A, Lengagne R, Desfrancois J, Labarriere N, Jotereau F (2012) HLA anchor optimization of the melan-A-HLA-A2 epitope within a long peptide is required for efficient cross-priming of human tumor-reactive T cells. J Immunol 188:2102–2110. https://doi.org/10.4049/jimmunol.1101807
Jurgens LA, Khanna R, Weber J, Orentas RJ (2006) Transduction of primary lymphocytes with Epstein-Barr virus (EBV) latent membrane protein-specific T-cell receptor induces lysis of virus-infected cells: A novel strategy for the treatment of Hodgkin’s disease and nasopharyngeal carcinoma. J Clin Immunol 26:22–32. https://doi.org/10.1007/s10875-006-6532-1
Gehring AJ, Xue SA, Ho ZZ, Teoh D, Ruedl C, Chia A, Koh S, Lim SG, Maini MK, Stauss H, Bertoletti A (2011) Engineering virus-specific T cells that target HBV infected hepatocytes and hepatocellular carcinoma cell lines. J Hepatol 55:103–110. https://doi.org/10.1016/j.jhep.2010.10.025
Jonuleit H, Kuhn U, Muller G, Steinbrink K, Paragnik L, Schmitt E, Knop J, Enk AH (1997) Pro-inflammatory cytokines and prostaglandins induce maturation of potent immunostimulatory dendritic cells under fetal calf serum-free conditions. Eur J Immunol 27:3135–3142. https://doi.org/10.1002/eji.1830271209
Dinarello CA (2009) Immunological and inflammatory functions of the interleukin-1 family. Annu Rev Immunol 27:519–550. https://doi.org/10.1146/annurev.immunol.021908.132612
Lebre MC, Burwell T, Vieira PL, Lora J, Coyle AJ, Kapsenberg ML, Clausen BE, De Jong EC (2005) Differential expression of inflammatory chemokines by Th1- and Th2-cell promoting dendritic cells: a role for different mature dendritic cell populations in attracting appropriate effector cells to peripheral sites of inflammation. Immunol Cell Biol 83:525–535. https://doi.org/10.1111/j.1440-1711.2005.01365.x
Garg AD, Vandenberk L, Koks C, Verschuere T, Boon L, Van Gool SW, Agostinis P (2016) Dendritic cell vaccines based on immunogenic cell death elicit danger signals and T cell-driven rejection of high-grade glioma. Sci Transl Med 8:328ra27. https://doi.org/10.1126/scitranslmed.aae0105
Wherry EJ (2011) T cell exhaustion. Nat Immunol 12:492–499
Macintyre AN, Gerriets VA, Nichols AG, Michalek RD, Rudolph MC, Deoliveira D, Anderson SM, Abel ED, Chen BJ, Hale LP, Rathmell JC (2014) The glucose transporter Glut1 is selectively essential for CD4 T cell activation and effector function. Cell Metab 20:61–72. https://doi.org/10.1016/j.cmet.2014.05.004
Sena LA, Li S, Jairaman A, Prakriya M, Ezponda T, Hildeman DA, Wang CR, Schumacker PT, Licht JD, Perlman H, Bryce PJ, Chandel NS (2013) Mitochondria are required for antigen-specific T cell activation through reactive oxygen species signaling. Immunity 38:225–236. https://doi.org/10.1016/j.immuni.2012.10.020
Pearce EL, Poffenberger MC, Chang C-H, Jones RG (2013) Fueling immunity: insights into metabolism and lymphocyte function. Science 342:1242454. https://doi.org/10.1126/science.1242454
Gerriets VA, Rathmell JC (2012) Metabolic pathways in T cell fate and function. Trends Immunol 33:168–173. https://doi.org/10.1016/j.it.2012.01.010
Jacobs SR, Herman CE, MacIver NJ, Wofford JA, Wieman HL, Hammen JJ, Rathmell JC (2008) Glucose uptake is limiting in T cell activation and requires CD28-mediated Akt-dependent and independent pathways. J Immunol 180:4476–4486
Van den Bergh JMJ, Smits E, Berneman ZN, Hutten TJA, De Reu H, Van Tendeloo VFI, Dolstra H, Lion E, Hobo W (2017) Monocyte-derived dendritic cells with silenced PD-1 ligands and transpresenting interleukin-15 stimulate strong tumor-reactive T-cell expansion. Cancer Immunol Res 5:710–715. https://doi.org/10.1158/2326-6066.CIR-16-0336
Van den Bergh JMJ, Smits ELJM., Versteven M, De Reu H, Berneman ZN, Van Tendeloo VFI, Lion E (2017) Characterization of interleukin-15-transpresenting dendritic cells for clinical use. J Immunol Res 2017:1975902. https://doi.org/10.1155/2017/1975902
Mitchell DA, Batich KA, Gunn MD, Huang M-N, Sanchez-Perez L, Nair SK, Congdon KL, Reap EA, Archer GE, Desjardins A, Friedman AH, Friedman HS, Herndon Ii JE et al (2015) Tetanus toxoid and CCL3 improve dendritic cell vaccines in mice and glioblastoma patients. Nature 519:366–369. https://doi.org/10.1038/nature14320
Carreno BM, Magrini V, Becker-Hapak M, Kaabinejadian S, Hundal J, Petti AA, Ly A, Lie W-R, Hildebrand WH, Mardis ER, Linette GP (2015) A dendritic cell vaccine increases the breadth and diversity of melanoma neoantigen-specific T cells. Science 348:803–808. https://doi.org/10.1126/science.aaa3828
Rosenblatt J, Stone RM, Uhl L, Neuberg D, Joyce R, Levine JD, Arnason J, McMasters M, Luptakova K, Jain S, Zwicker JI, Hamdan A, Boussiotis V et al (2016) Individualized vaccination of AML patients in remission is associated with induction of antileukemia immunity and prolonged remissions. Sci Transl Med 8:368ra171. https://doi.org/10.1126/scitranslmed.aag1298
Dovedi SJ, Lipowska-Bhalla G, Beers SA, Cheadle EJ, Mu L, Glennie MJ, Illidge TM, Honeychurch J (2016) Antitumor efficacy of radiation plus immunotherapy depends upon dendritic cell activation of effector CD8+ T Cells. Cancer Immunol Res 4:621–630. https://doi.org/10.1158/2326-6066.CIR-15-0253
Bukczynski J, Wen T, Watts TH (2003) Costimulation of human CD28- T cells by 4-1BB ligand. Eur J Immunol 33:446–454. https://doi.org/10.1002/immu.200310020
Bertram EM, Dawicki W, Sedgmen B, Bramson JL, Lynch DH, Watts TH (2004) A switch in costimulation from CD28 to 4-1BB during primary versus secondary CD8 T cell response to influenza in vivo. J Immunol 172:981–988
Dharmadhikari B, Wu M, Abdullah NS, Rajendran S, Ishak ND, Nickles E, Harfuddin Z, Schwarz H (2016) CD137 and CD137L signals are main drivers of type 1, cell-mediated immune responses. Oncoimmunology 5:e1113367. https://doi.org/10.1080/2162402X.2015.1113367
Campana D, Schwarz H, Imai C (2014) 4-1BB chimeric antigen receptors. Cancer J 20:134–140. https://doi.org/10.1097/PPO.0000000000000028
Setareh M, Schwarz H, Lotz M (1995) A mRNA variant encoding a soluble form of 4-1BB, a member of the murine NGF/TNF receptor family. Gene 164:311–315
Michel J, Langstein J, Hofstadter F, Schwarz H (1998) A soluble form of CD137 (ILA/4-1BB),a member of the TNF receptor family, is released by activated lymphocytes and is detectable in sera of patients with rheumatoid arthritis. Eur J Immunol 28:290–295. https://doi.org/10.1002/(Sici)1521–4141(199801)28:01<290::Aid-Immu290>3.0.Co;2-S
Shao Z, Sun F, Koh DR, Schwarz H (2008) Characterisation of soluble murine CD137 and its association with systemic lupus. Mol Immunol 45:3990–3999. https://doi.org/10.1016/j.molimm.2008.05.028
Furtner M, Straub RH, Kruger S, Schwarz H (2005) Levels of soluble CD137 are enhanced in sera of leukemia and lymphoma patients and are strongly associated with chronic lymphocytic leukemia. Leukemia 19:883–885. https://doi.org/10.1038/sj.leu.2403675
Acknowledgements
We thank the Life Sciences Institute flow cytometry core facility for excellent assistance, and members of our laboratory for critical proofreading of the manuscript.
Funding
This research was supported by a Grant (NMRC/BnB/018b/2015) from the National Medical Research Council, Singapore.
Author information
Authors and Affiliations
Contributions
Conception and design: Bhushan Dharmadhikari, Zulkarnain Harfuddin, and Herbert Schwarz. Development of methodology: Bhushan Dharmadhikari, Emily Nickles, and Zulkarnain Harfuddin. Acquisition of data: Bhushan Dharmadhikari, Emily Nickles, Zulkarnain Harfuddin, and Nur Diana Binte Ishak. Analysis and interpretation of data: Bhushan Dharmadhikari, Emily Nickles, Zulkarnain Harfuddin, Nur Diana Binte Ishak, Antonio Bertoletti, and Herbert Schwarz. Writing, review, and/or revision of the manuscript: Bhushan Dharmadhikari, Emily Nickles, Zulkarnain Harfuddin, Qun Zeng, Antonio Bertoletti, and Herbert Schwarz. Administrative, technical, or material support: Bhushan Dharmadhikari, Emily Nickles, Zulkarnain Harfuddin, Nur Diana Binte Ishak, Qun Zeng, and Antonio Bertoletti.
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors. PBMC and monocytes were isolated from buffy coats of healthy volunteers, which were obtained from the Blood Donation Center of the National University Hospital, Singapore, after obtaining donors’ consent and approval by the NUS IRB (# B-15-320E). Cell lines used were newly purchased from ATCC (Manassas, VA, USA).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dharmadhikari, B., Nickles, E., Harfuddin, Z. et al. CD137L dendritic cells induce potent response against cancer-associated viruses and polarize human CD8+ T cells to Tc1 phenotype. Cancer Immunol Immunother 67, 893–905 (2018). https://doi.org/10.1007/s00262-018-2144-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-018-2144-x