Abstract
Background
Chemokine (C-X-C motif) ligand 13 (CXCL13/BLC/BCA-1) is a cytokine from C-X-C chemokine family, which is selectively chemotactic for B cells. Previous research has demonstrated that high CXCL13 expression is correlated to poor prognosis in various cancers. However, the association between CXCL13 expression and gastric cancer is still unclear.
Methods
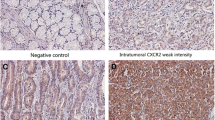
Intratumoral CXCL13 expression was evaluated by immunohistochemistry using a semi-quantitative method (modified H-score) in a testing set of 214 and a validation set of 227 randomly selected gastric cancer patients resected in 2008 in one institution. The median value was used as the cut-off point. We performed correlative analysis of CXCL-13 expression with clinicopathological variables, Kaplan–Meier analysis for association with overall survival (OS), and multivariate modeling.
Results
High CXCL13 expression was associated with larger tumor diameter and shorter OS. By multivariate analysis, CXCL13 expression was associated with OS independently from clinicopathological factors. Within the T2–4 stage patients group, low CXCL13 expression was associated with longer survival, especially in the subgroup of patients (57.6%) who received adjuvant chemotherapy.
Conclusions
Intratumoral CXCL13 expression appears as an independent prognostic marker for patients after gastric cancer resection. In addition, CXCL13 expression may serve as a predictive biomarker of response to postoperative adjuvant chemotherapy in these patients.



Similar content being viewed by others
Abbreviations
- Akt:
-
Protein kinase B, PKB
- BCA-1:
-
B cell attracting chemokine 1
- CXCL13:
-
Chemokine (C-X-C motif) ligand 13
- CXCR5:
-
Chemokine (C-X-C motif) receptor 5
- IOD:
-
Integrated optical density
- MMP:
-
Matrix metalloproteinase
- PI3-K:
-
Phosphatidylinositol 3-kinase
- TAMs:
-
Tumor-associated macrophages
- TMA:
-
Tissue microarray
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA Cancer J Clin 61(2):69
Parkin DM, Pisani P, Ferlay J (1999) Global cancer statistics. CA Cancer J Clin 49(1):33
Hu Y, Huang C, Sun Y, Su X, Cao H, Hu J, Xue Y, Suo J, Tao K, He X (2016) Morbidity and mortality of laparoscopic versus open D2 distal gastrectomy for advanced gastric cancer: a randomized controlled trial. J Clin Oncol 34(12):1350
Balkwill FR (2012) The chemokine system and cancer. J Pathol 226(2):148–157
Sarvaiya PJ, Guo D, Ulasov I, Gabikian P, Lesniak MS (2013) Chemokines in tumor progression and metastasis. Oncotarget 4(12):2171–2185
Franciszkiewicz K, Boissonnas A, Boutet M, Combadière C, Mami-Chouaib F (2012) Role of chemokines and chemokine receptors in shaping the effector phase of the antitumor immune response. Cancer Res 72(24):6325–6332
Raman D, Sobolikdelmaire T, Richmond A (2011) Chemokines in health and disease. Exp Cell Res 317(5):575–589
Gunn MD, Ngo VN, Ansel KM, Ekland EH, Cyster JG, Williams LT (1998) A B-cell-homing chemokine made in lymphoid follicles activates Burkitt’s lymphoma receptor-1. Nature 391(6669):799
Biswas S, Sengupta S, Roy CS, Jana S, Mandal G, Mandal PK, Saha N, Malhotra V, Gupta A, Kuprash DV (2016) CXCL13–CXCR5 co-expression regulates epithelial to mesenchymal transition of breast cancer cells during lymph node metastasis. Breast Cancer Res Treat 155:615. doi:10.1007/s10549-016-3713-3
Qi XW, Xia SH, Yin Y, Jin LF, Pu Y, Hua D, Wu HR (2014) Expression features of CXCR5 and its ligand, CXCL13 associated with poor prognosis of advanced colorectal cancer. Eur Rev Med Pharmacol Sci 18(13):1916
Singh S, Singh R, Sharma PK, Singh UP, Rai SN, Chung LW, Cooper CR, Novakovic KR, Grizzle WE, Jr LJ (2009) Serum CXCL13 positively correlates with prostatic disease, prostate-specific antigen and mediates prostate cancer cell invasion, integrin clustering and cell adhesion. Cancer Lett 283(1):29–35
Duan Z, Gao J, Zhang L, Liang H, Huang X, Xu Q, Zhang Y, Shen T, Lu F (2015) Phenotype and function of CXCR5 + CD45RA − CD4 + T cells were altered in HBV-related hepatocellular carcinoma and elevated serum CXCL13 predicted better prognosis. Oncotarget 6(42):44239–44253
Sambandam Y, Sundaram K, Liu A, Kirkwood KL, Ries WL, Reddy SV (2012) CXCL13 activation of c-Myc induces RANK ligand expression in stromal/preosteoblast cells in the oral squamous cell carcinoma tumor-bone microenvironment. Oncogene 32(1):97
Singh R, Gupta P, Kloecker GH, Singh S, Jr LJ (2014) Expression and clinical significance of CXCR5/CXCL13 in human non-small cell lung carcinoma. Int J Oncol 45(6):2232–2240
Kim SJ, Ryu KJ, Hong M, Ko YH, Kim WS (2015) The serum CXCL13 level is associated with the Glasgow Prognostic Score in extranodal NK/T cell lymphoma patients. J Hematol Oncol 8(1):49
Grosso FD, Coco S, Scaruffi P, Stigliani S, Valdora F, Benelli R, Salvi S, Boccardo S, Truini M, Croce M (2011) Role of CXCL13-CXCR5 crosstalk between malignant neuroblastoma cells and schwannian stromal cells in neuroblastic tumors. Mol Cancer Res 9(7):815
Ding Y, Shen J, Zhang G, Chen X, Wu JM, Chen W (2015) CD40 controls CXCR5-induced recruitment of myeloid-derived suppressor cells to gastric cancer. Oncotarget 6(36):38901–38911
Hofman VJ, Moreilhon C, Brest PD et al (2007) Gene expression profiling in human gastric mucosa infected with Helicobacter pylori. Mod Pathol 20(9):974–989
Galamb O, Gyõrffy B, Sipos F et al (2008) Helicobacter pylori and antrum erosion-specific gene expression patterns: the discriminative role of CXCL13 and VCAM1 transcripts. Helicobacter 13(2):112–126
Van Cutsem E, Sagaert X, Topal B, Haustermans K, Prenen H (2016) Gastric cancer. Lancet 388(10060):2654–2664
Ebert LM, Schaerli P, Moser B (2005) Chemokine-mediated control of T cell traffic in lymphoid and peripheral tissues. Mol Immunol 42(7):799
Carlsen HS, Baekkevold ES, Morton HC, Haraldsen G, Brandtzaeg P (2004) Monocyte-like and mature macrophages produce CXCL13 (B cell-attracting chemokine 1) in inflammatory lesions with lymphoid neogenesis. Blood 104(10):3021–3027
Bürkle A, Niedermeier M, Schmittgräff A, Wierda WG, Keating MJ, Burger JA (2007) Overexpression of the CXCR5 chemokine receptor, and its ligand, CXCL13 in B-cell chronic lymphocytic leukemia. Blood 110(110):3316–3325
Fallarino F, Grohmann U, You S, Mcgrath BC, Cavener DR, Vacca C, Orabona C, Bianchi R, Belladonna ML, Volpi C (2006) The combined effects of tryptophan starvation and tryptophan catabolites down-regulate T cell receptor zeta-chain and induce a regulatory phenotype in naive T cells. J Immunol 176(11):6752
Katz JB, Muller AJ, Prendergast GC (2008) Indoleamine 2,3-dioxygenase in T cell tolerance and tumoral immune escape. Immunol Rev 222(1):206
Widney DP, Olafsen T, Wu AM, Kitchen CM, Said JW, Smith JB, Peña G, Magpantay LI, Penichet ML, Martinez-Maza O (2013) Levels of murine, but not human, CXCL13 are greatly elevated in NOD-SCID mice bearing the AIDS-associated Burkitt lymphoma cell line, 2F7. PLoS One 8(8):e72414
Astigiano S, Morandi B, Costa R, Mastracci L, D’Agostino A, Ratto GB, Melioli G, Frumento G (2005) Eosinophil granulocytes account for indoleamine 2,3-dioxygenase-mediated immune escape in human non-small cell lung cancer. Neoplasia 7(4):390–396
Lint PV, Libert C (2007) Chemokine and cytokine processing by matrix metalloproteinases and its effect on leukocyte migration and inflammation. J Leukoc Biol 82(82):1375–1381
Zhu Z, Zhang X, Guo H, Fu L, Pan G, Sun Y (2015) CXCL13-CXCR5 axis promotes the growth and invasion of colon cancer cells via PI3K/AKT pathway. Mol Cell Biochem 400(1):287–295
Ohtsu A, Shah MA, Van CE et al (2011) Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a randomized, double-blind, placebo-controlled phase III study. J Clin Oncol 29(30):3968–3976
Selvakumaran M, Yao KS, Feldman MD, O’Dwyer PJ (2008) Antitumor effect of the angiogenesis inhibitor bevacizumab is dependent on susceptibility of tumors to hypoxia-induced apoptosis. Biochem Pharmacol 75(3):627
Ammirante M, Shalapour S, Kang Y, Jamieson CA, Karin M (2014) Tissue injury and hypoxia promote malignant progression of prostate cancer by inducing CXCL13 expression in tumor myofibroblasts. PNAS 111(41):14776
Durand C, Westendorf J, Tse K, Gold M (2006) The Rap GTPases mediate CXCL13- and sphingosine1-phosphate-induced chemotaxis, adhesion, and Pyk2 tyrosine phosphorylation in B lymphocytes. Eur J Immunol 36(8):2235–2249
Song G, Ouyang G, Bao S (2005) The activation of AKT/PKB signaling pathway and cell survival. J Cell Mol Med 9(1):59
Page C, Lin HJ, Jin Y, Castle VP, Nunez G, Huang M, Lin J (2000) Overexpression of Akt/AKT can modulate chemotherapy-induced apoptosis. Anticancer Res 20(1A):407
Brognard J, Clark AS, Ni Y, Dennis PA (2001) Akt/protein kinase B is constitutively active in non-small cell lung cancer cells and promotes cellular survival and resistance to chemotherapy and radiation. Cancer Res 61(10):3986
Acknowledgements
This study was funded by Grants from National Natural Science Foundation of China (81471621, 81472227, 81501999, 81671628 and 31770851) and Shanghai Sailing Program (17YF1402200). All these study sponsors have no roles in the study design, in the collection, analysis, and interpretation of data.
Author information
Authors and Affiliations
Contributions
Y. Wei, C. Lin and H. Li: acquisition of data, analysis and interpretation of data, statistical analysis and drafting of the manuscript. Z. Xu, J. Wang, R. Li, H. Liu and H. Zhang: technical and material support. H. He and J. Xu: study concept and design, analysis and interpretation of data, drafting of the manuscript, obtained funding and study supervision. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Wei, Y., Lin, C., Li, H. et al. CXCL13 expression is prognostic and predictive for postoperative adjuvant chemotherapy benefit in patients with gastric cancer. Cancer Immunol Immunother 67, 261–269 (2018). https://doi.org/10.1007/s00262-017-2083-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-017-2083-y